 Found 429 hits with Last Name = 'reisine' and Initial = 't'
Found 429 hits with Last Name = 'reisine' and Initial = 't' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Somatostatin receptor type 2
(MOUSE) | BDBM82455
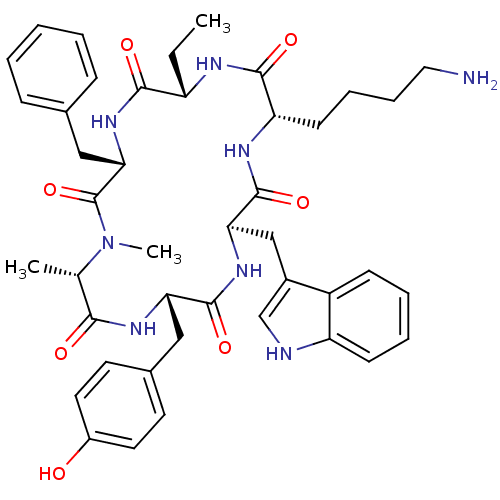
(BIM 23027 | BIM-23027 | N-Methylcyclo[L-Ala-L-Tyr-...)Show SMILES CC[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@H](C)N(C)C(=O)[C@H](Cc2ccccc2)NC1=O Show InChI InChI=1S/C43H54N8O7/c1-4-32-39(54)50-37(23-27-12-6-5-7-13-27)43(58)51(3)26(2)38(53)48-35(22-28-17-19-30(52)20-18-28)41(56)49-36(24-29-25-45-33-15-9-8-14-31(29)33)42(57)47-34(40(55)46-32)16-10-11-21-44/h5-9,12-15,17-20,25-26,32,34-37,45,52H,4,10-11,16,21-24,44H2,1-3H3,(H,46,55)(H,47,57)(H,48,53)(H,49,56)(H,50,54)/t26-,32-,34-,35-,36+,37-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM82465
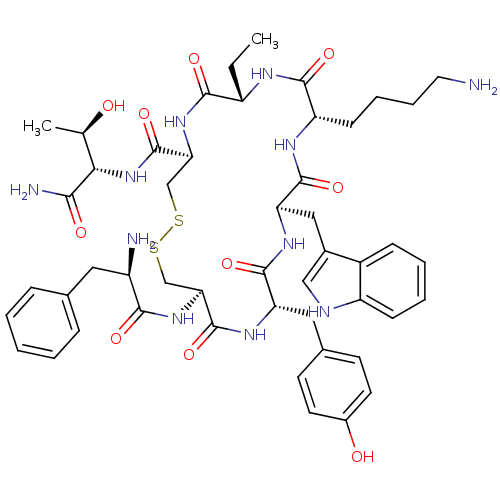
(BIM 23023 | BIM-23023 | D-Phe-L-Cys(1)-L-Tyr-D-Trp...)Show SMILES CC[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@H](CSSC[C@H](NC1=O)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)NC(=O)[C@H](N)Cc1ccccc1 Show InChI InChI=1S/C49H65N11O10S2/c1-3-34-44(65)59-40(49(70)60-41(27(2)61)42(52)63)26-72-71-25-39(58-43(64)33(51)21-28-11-5-4-6-12-28)48(69)56-37(22-29-16-18-31(62)19-17-29)46(67)57-38(23-30-24-53-35-14-8-7-13-32(30)35)47(68)55-36(45(66)54-34)15-9-10-20-50/h4-8,11-14,16-19,24,27,33-34,36-41,53,61-62H,3,9-10,15,20-23,25-26,50-51H2,1-2H3,(H2,52,63)(H,54,66)(H,55,68)(H,56,69)(H,57,67)(H,58,64)(H,59,65)(H,60,70)/t27-,33-,34+,36+,37+,38-,39+,40+,41+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(RAT) | BDBM50097783
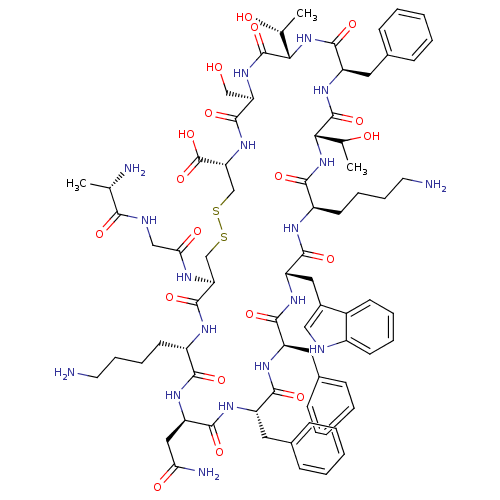
(CHEMBL438401 | D-Trp8 SST-14 | SOMATOSTATIN | SRIF...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@@H](NC(=O)[C@@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)C(C)O Show InChI InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42?,43+,50-,51+,52-,53+,54+,55-,56+,57+,58-,59+,62-,63-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 44: 385-92 (1993)
BindingDB Entry DOI: 10.7270/Q2X065KZ |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM84626
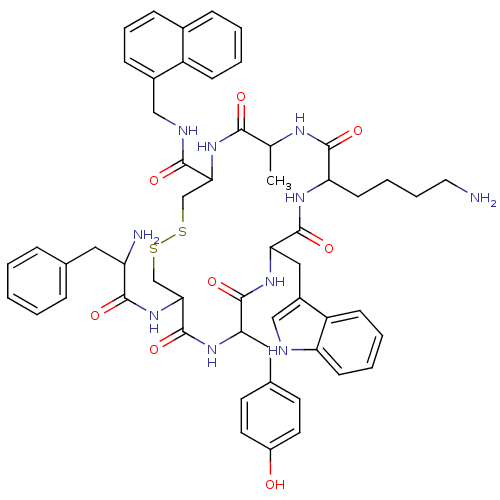
(BIM 23034)Show SMILES CC1NC(=O)C(CCCCN)NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(Cc2ccc(O)cc2)NC(=O)C(CSSCC(NC1=O)C(=O)NCc1cccc2ccccc12)NC(=O)C(N)Cc1ccccc1 Show InChI InChI=1S/C55H64N10O8S2/c1-33-49(67)64-47(51(69)59-29-37-16-11-15-36-14-5-6-17-40(36)37)31-74-75-32-48(65-50(68)42(57)26-34-12-3-2-4-13-34)55(73)62-45(27-35-21-23-39(66)24-22-35)53(71)63-46(28-38-30-58-43-19-8-7-18-41(38)43)54(72)61-44(52(70)60-33)20-9-10-25-56/h2-8,11-19,21-24,30,33,42,44-48,58,66H,9-10,20,25-29,31-32,56-57H2,1H3,(H,59,69)(H,60,70)(H,61,72)(H,62,73)(H,63,71)(H,64,67)(H,65,68) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(RAT) | BDBM82253
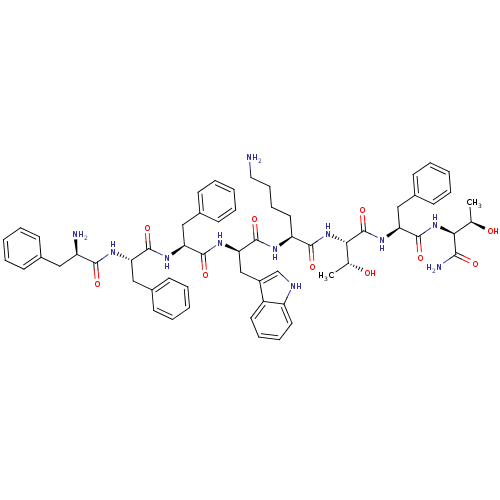
(BIM 23052 | CAS_133073-82-2)Show SMILES C[C@@H](O)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](N)Cc1ccccc1)[C@@H](C)O)C(N)=O |r| Show InChI InChI=1S/C61H75N11O10/c1-37(73)52(54(64)75)71-60(81)50(34-42-25-13-6-14-26-42)70-61(82)53(38(2)74)72-56(77)47(29-17-18-30-62)66-59(80)51(35-43-36-65-46-28-16-15-27-44(43)46)69-58(79)49(33-41-23-11-5-12-24-41)68-57(78)48(32-40-21-9-4-10-22-40)67-55(76)45(63)31-39-19-7-3-8-20-39/h3-16,19-28,36-38,45,47-53,65,73-74H,17-18,29-35,62-63H2,1-2H3,(H2,64,75)(H,66,80)(H,67,76)(H,68,78)(H,69,79)(H,70,82)(H,71,81)(H,72,77)/t37-,38-,45-,47+,48+,49+,50+,51-,52+,53+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 44: 385-92 (1993)
BindingDB Entry DOI: 10.7270/Q2X065KZ |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM84623
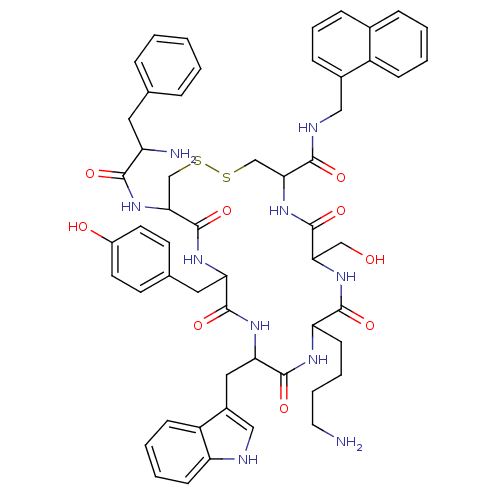
(NC4-28B)Show SMILES NCCCCC1NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(Cc2ccc(O)cc2)NC(=O)C(CSSCC(NC(=O)C(CO)NC1=O)C(=O)NCc1cccc2ccccc12)NC(=O)C(N)Cc1ccccc1 Show InChI InChI=1S/C55H64N10O9S2/c56-24-9-8-19-43-51(70)63-46(30-66)54(73)65-47(50(69)59-28-36-15-10-14-35-13-4-5-16-39(35)36)31-75-76-32-48(64-49(68)41(57)25-33-11-2-1-3-12-33)55(74)61-44(26-34-20-22-38(67)23-21-34)52(71)62-45(53(72)60-43)27-37-29-58-42-18-7-6-17-40(37)42/h1-7,10-18,20-23,29,41,43-48,58,66-67H,8-9,19,24-28,30-32,56-57H2,(H,59,69)(H,60,72)(H,61,74)(H,62,71)(H,63,70)(H,64,68)(H,65,73) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM50097783

(CHEMBL438401 | D-Trp8 SST-14 | SOMATOSTATIN | SRIF...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@@H](NC(=O)[C@@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)C(C)O Show InChI InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42?,43+,50-,51+,52-,53+,54+,55-,56+,57+,58-,59+,62-,63-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(RAT) | BDBM82463
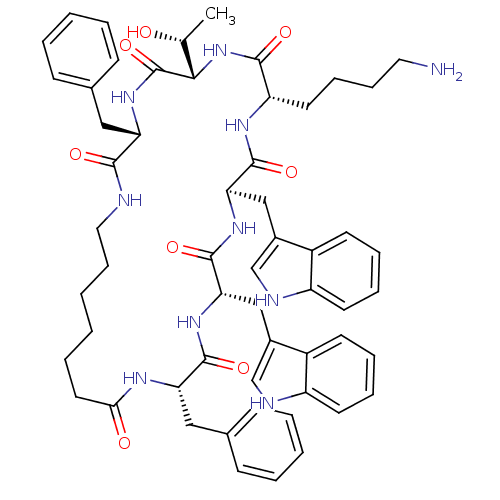
(Cyclo[L-Trp-D-Trp-L-Lys-L-Thr-L-Phe-(7-amino*hepta...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)CCCCCCNC(=O)[C@H](Cc2ccccc2)NC1=O Show InChI InChI=1S/C57H70N10O8/c1-36(68)51-57(75)66-46(30-37-18-6-4-7-19-37)52(70)59-29-17-3-2-10-27-50(69)62-47(31-38-20-8-5-9-21-38)54(72)64-49(33-40-35-61-44-25-14-12-23-42(40)44)56(74)65-48(32-39-34-60-43-24-13-11-22-41(39)43)55(73)63-45(53(71)67-51)26-15-16-28-58/h4-9,11-14,18-25,34-36,45-49,51,60-61,68H,2-3,10,15-17,26-33,58H2,1H3,(H,59,70)(H,62,69)(H,63,73)(H,64,72)(H,65,74)(H,66,75)(H,67,71)/t36-,45+,46+,47+,48-,49+,51+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 44: 385-92 (1993)
BindingDB Entry DOI: 10.7270/Q2X065KZ |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM82469
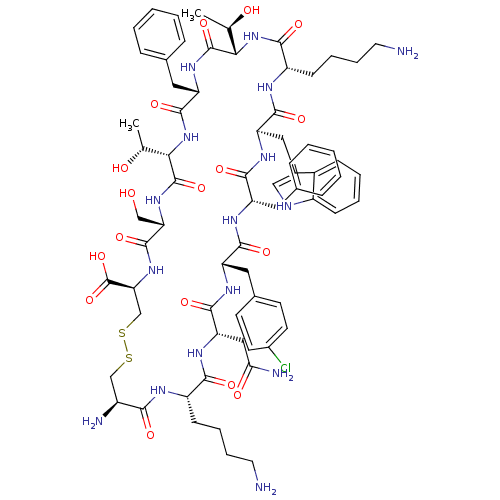
(BIM 23003 | BIM-23003 | EC5-21 | L-Cys(1)-L-Lys-L-...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(Cl)cc2)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](N)CSSC[C@H](NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC1=O)[C@@H](C)O)C(O)=O Show InChI InChI=1S/C71H95ClN16O17S2/c1-38(90)58-69(102)84-52(30-41-17-7-4-8-18-41)67(100)88-59(39(2)91)70(103)85-55(35-89)68(101)86-56(71(104)105)37-107-106-36-46(75)60(93)78-48(21-11-13-27-73)61(94)83-54(33-57(76)92)66(99)81-51(31-42-23-25-44(72)26-24-42)63(96)80-50(29-40-15-5-3-6-16-40)64(97)82-53(32-43-34-77-47-20-10-9-19-45(43)47)65(98)79-49(62(95)87-58)22-12-14-28-74/h3-10,15-20,23-26,34,38-39,46,48-56,58-59,77,89-91H,11-14,21-22,27-33,35-37,73-75H2,1-2H3,(H2,76,92)(H,78,93)(H,79,98)(H,80,96)(H,81,99)(H,82,97)(H,83,94)(H,84,102)(H,85,103)(H,86,101)(H,87,95)(H,88,100)(H,104,105)/t38-,39-,46+,48+,49+,50+,51+,52+,53-,54+,55+,56+,58+,59+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM82462
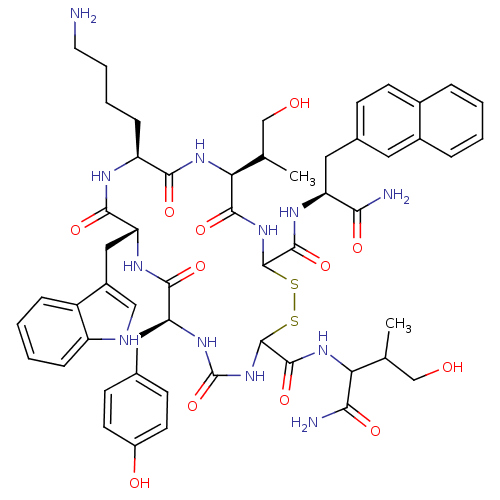
(BIM 23059 | D-Nal-c[Cys-Tyr-D-Trp-Lys-Thr-Cys]-Thr...)Show SMILES CC(CO)C(NC(=O)C1NC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@H](Cc2c[nH]c3ccccc23)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C(C)CO)C(=O)NC(SS1)C(=O)N[C@@H](Cc1ccc2ccccc2c1)C(N)=O)C(N)=O |r| Show InChI InChI=1S/C54H68N12O12S2/c1-28(26-67)42(45(57)71)63-51(77)53-66-54(78)62-40(22-30-15-18-35(69)19-16-30)47(73)61-41(24-34-25-58-37-12-6-5-11-36(34)37)48(74)59-38(13-7-8-20-55)46(72)64-43(29(2)27-68)49(75)65-52(79-80-53)50(76)60-39(44(56)70)23-31-14-17-32-9-3-4-10-33(32)21-31/h3-6,9-12,14-19,21,25,28-29,38-43,52-53,58,67-69H,7-8,13,20,22-24,26-27,55H2,1-2H3,(H2,56,70)(H2,57,71)(H,59,74)(H,60,76)(H,61,73)(H,63,77)(H,64,72)(H,65,75)(H2,62,66,78)/t28?,29?,38-,39-,40-,41+,42?,43-,52?,53?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM84630
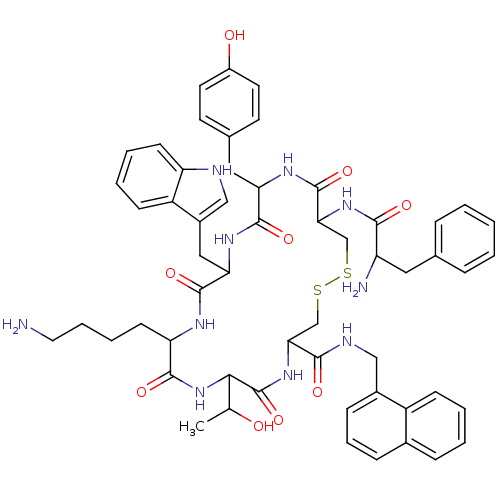
(BIM 23060)Show SMILES CC(O)C1NC(=O)C(CCCCN)NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(Cc2ccc(O)cc2)NC(=O)C(CSSCC(NC1=O)C(=O)NCc1cccc2ccccc12)NC(=O)C(N)Cc1ccccc1 Show InChI InChI=1S/C56H66N10O9S2/c1-33(67)49-56(75)65-47(51(70)60-29-37-16-11-15-36-14-5-6-17-40(36)37)31-76-77-32-48(64-50(69)42(58)26-34-12-3-2-4-13-34)55(74)62-45(27-35-21-23-39(68)24-22-35)53(72)63-46(28-38-30-59-43-19-8-7-18-41(38)43)54(73)61-44(52(71)66-49)20-9-10-25-57/h2-8,11-19,21-24,30,33,42,44-49,59,67-68H,9-10,20,25-29,31-32,57-58H2,1H3,(H,60,70)(H,61,73)(H,62,74)(H,63,72)(H,64,69)(H,65,75)(H,66,71) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50001714
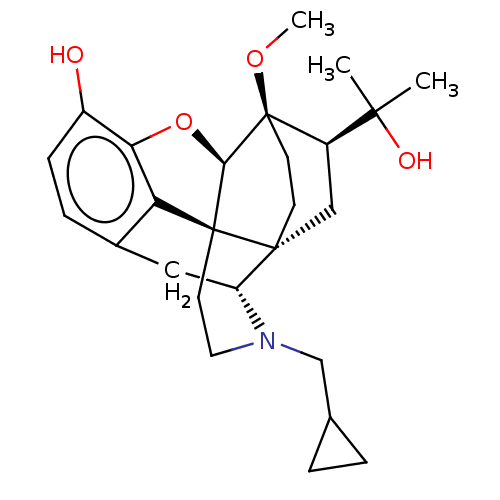
(2-[3-cyclopropylmethyl-11-hydroxy-15-methoxy-(1S,2...)Show SMILES CO[C@]12CC[C@@]3(C[C@@H]1C(C)(C)O)[C@H]1Cc4ccc(O)c5O[C@@H]2[C@]3(CCN1CC1CC1)c45 |THB:8:7:22.21:4.3| Show InChI InChI=1S/C26H35NO4/c1-23(2,29)18-13-24-8-9-26(18,30-3)22-25(24)10-11-27(14-15-4-5-15)19(24)12-16-6-7-17(28)21(31-22)20(16)25/h6-7,15,18-19,22,28-29H,4-5,8-14H2,1-3H3/t18-,19-,22-,24-,25+,26-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM82552
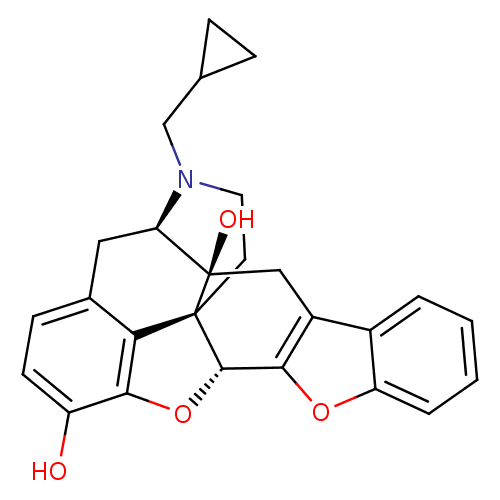
(CAS_111555-58-9 | NTB | naltrindolebenzofuran)Show SMILES Oc1ccc2C[C@H]3N(CC4CC4)CC[C@@]45[C@@H](Oc1c24)c1oc2ccccc2c1C[C@@]35O |r| Show InChI InChI=1S/C26H25NO4/c28-18-8-7-15-11-20-26(29)12-17-16-3-1-2-4-19(16)30-22(17)24-25(26,21(15)23(18)31-24)9-10-27(20)13-14-5-6-14/h1-4,7-8,14,20,24,28-29H,5-6,9-13H2/t20-,24+,25+,26-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50039022
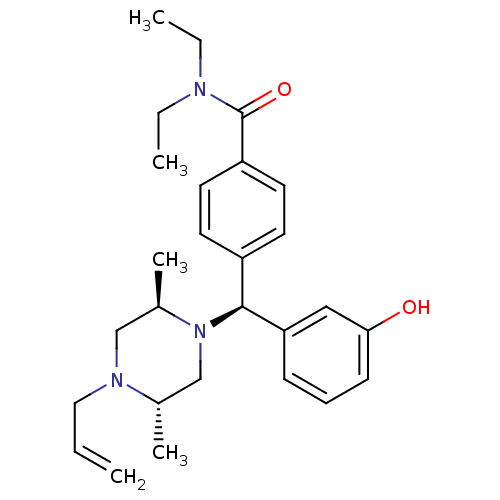
(4-[(S)-((2R,5S)-4-Allyl-2,5-dimethyl-piperazin-1-y...)Show SMILES CCN(CC)C(=O)c1ccc(cc1)[C@H](N1C[C@H](C)N(CC=C)C[C@H]1C)c1cccc(O)c1 Show InChI InChI=1S/C27H37N3O2/c1-6-16-29-18-21(5)30(19-20(29)4)26(24-10-9-11-25(31)17-24)22-12-14-23(15-13-22)27(32)28(7-2)8-3/h6,9-15,17,20-21,26,31H,1,7-8,16,18-19H2,2-5H3/t20-,21+,26-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 3
(MOUSE) | BDBM84629
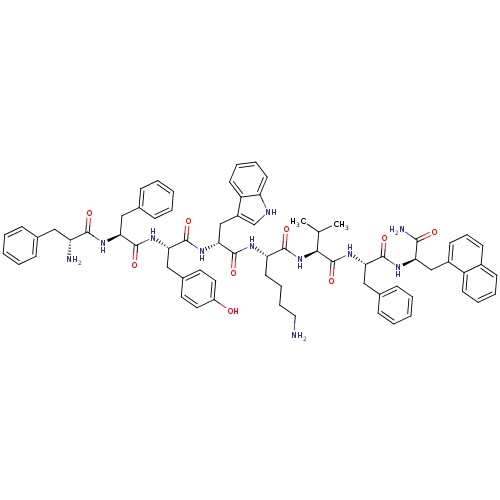
(BIM 23056)Show SMILES CC(C)[C@H](NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](N)Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@H](Cc1cccc2ccccc12)C(N)=O Show InChI InChI=1S/C71H81N11O9/c1-44(2)63(71(91)81-61(39-47-23-10-5-11-24-47)67(87)77-58(64(74)84)41-50-27-18-26-49-25-12-13-28-53(49)50)82-66(86)57(31-16-17-36-72)76-70(90)62(42-51-43-75-56-30-15-14-29-54(51)56)80-69(89)60(40-48-32-34-52(83)35-33-48)79-68(88)59(38-46-21-8-4-9-22-46)78-65(85)55(73)37-45-19-6-3-7-20-45/h3-15,18-30,32-35,43-44,55,57-63,75,83H,16-17,31,36-42,72-73H2,1-2H3,(H2,74,84)(H,76,90)(H,77,87)(H,78,85)(H,79,88)(H,80,89)(H,81,91)(H,82,86)/t55-,57+,58-,59+,60+,61+,62-,63+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM82551
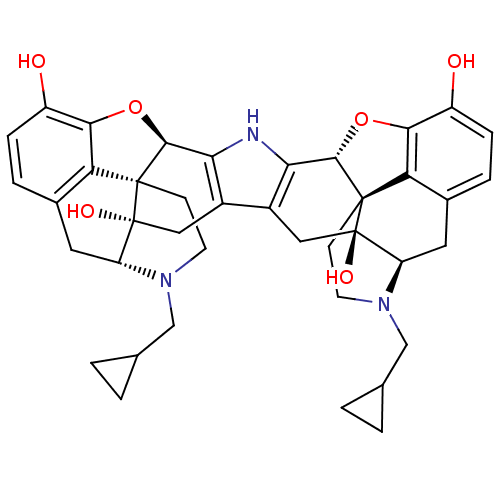
(C18130 | CAS_105618-26-6 | NOR-BNI (HCI)2 | NORBNI)Show SMILES Oc1ccc2C[C@H]3N(CC4CC4)CC[C@@]45[C@@H](Oc1c24)c1[nH]c2[C@@H]4Oc6c7c(C[C@H]8N(CC9CC9)CC[C@@]47[C@@]8(O)Cc2c1C[C@@]35O)ccc6O |r| Show InChI InChI=1S/C40H43N3O6/c44-25-7-5-21-13-27-39(46)15-23-24-16-40(47)28-14-22-6-8-26(45)34-30(22)38(40,10-12-43(28)18-20-3-4-20)36(49-34)32(24)41-31(23)35-37(39,29(21)33(25)48-35)9-11-42(27)17-19-1-2-19/h5-8,19-20,27-28,35-36,41,44-47H,1-4,9-18H2/t27-,28-,35+,36+,37+,38+,39-,40-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM21864
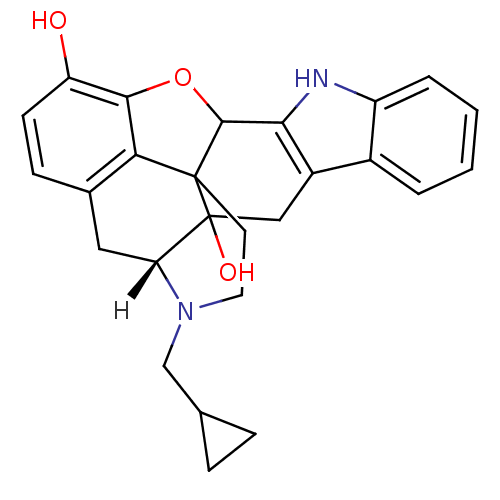
((21R)-22-(cyclopropylmethyl)-14-oxa-11,22-diazahep...)Show SMILES [H][C@@]12Cc3ccc(O)c4OC5c6[nH]c7ccccc7c6CC1(O)C5(CCN2CC1CC1)c34 |THB:27:26:21:31.2.3| Show InChI InChI=1S/C26H26N2O3/c29-19-8-7-15-11-20-26(30)12-17-16-3-1-2-4-18(16)27-22(17)24-25(26,21(15)23(19)31-24)9-10-28(20)13-14-5-6-14/h1-4,7-8,14,20,24,27,29-30H,5-6,9-13H2/t20-,24?,25?,26?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM83436
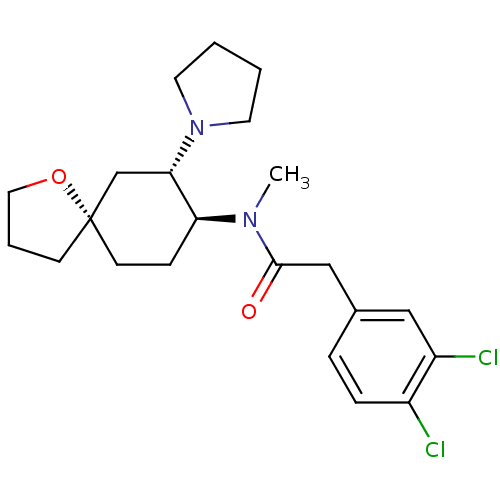
(2-(3,4-dichlorophenyl)-N-methyl-N-[(5R,7S,8S)-7-(1...)Show SMILES CN([C@H]1CC[C@@]2(CCCO2)C[C@@H]1N1CCCC1)C(=O)Cc1ccc(Cl)c(Cl)c1 Show InChI InChI=1S/C22H30Cl2N2O2/c1-25(21(27)14-16-5-6-17(23)18(24)13-16)19-7-9-22(8-4-12-28-22)15-20(19)26-10-2-3-11-26/h5-6,13,19-20H,2-4,7-12,14-15H2,1H3/t19-,20-,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 3
(MOUSE) | BDBM82459
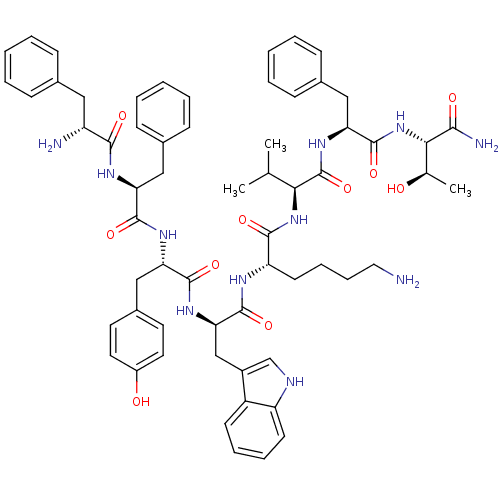
(BIM 23058 | BIM-23058 | D-Phe-L-Phe-L-Tyr-D-Trp-L-...)Show SMILES CC(C)[C@H](NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](N)Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H]([C@@H](C)O)C(N)=O Show InChI InChI=1S/C62H77N11O10/c1-37(2)53(62(83)71-51(33-41-21-11-6-12-22-41)61(82)73-54(38(3)74)55(65)76)72-57(78)48(25-15-16-30-63)67-60(81)52(35-43-36-66-47-24-14-13-23-45(43)47)70-59(80)50(34-42-26-28-44(75)29-27-42)69-58(79)49(32-40-19-9-5-10-20-40)68-56(77)46(64)31-39-17-7-4-8-18-39/h4-14,17-24,26-29,36-38,46,48-54,66,74-75H,15-16,25,30-35,63-64H2,1-3H3,(H2,65,76)(H,67,81)(H,68,77)(H,69,79)(H,70,80)(H,71,83)(H,72,78)(H,73,82)/t38-,46-,48+,49+,50+,51+,52-,53+,54+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM82555

(CAS_4424 | NSC_4424 | Naloxonazine)Show SMILES Oc1ccc2CC3N(CC=C)CCC45C(Oc1c24)C(CCC35O)=NN=C1CCC2(O)C3Cc4ccc(O)c5OC1C2(CCN3CC=C)c45 |w:24.28,18.21,TLB:27:28:43.42.41:31.32.47,22:21:5.4.17:12.11.7,20:21:5.4.17:12.11.7,THB:29:28:43.42.41:31.32.47| Show InChI InChI=1S/C38H42N4O6/c1-3-15-41-17-13-35-29-21-5-7-25(43)31(29)47-33(35)23(9-11-37(35,45)27(41)19-21)39-40-24-10-12-38(46)28-20-22-6-8-26(44)32-30(22)36(38,34(24)48-32)14-18-42(28)16-4-2/h3-8,27-28,33-34,43-46H,1-2,9-20H2 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 3
(MOUSE) | BDBM50097783

(CHEMBL438401 | D-Trp8 SST-14 | SOMATOSTATIN | SRIF...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@@H](NC(=O)[C@@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)C(C)O Show InChI InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42?,43+,50-,51+,52-,53+,54+,55-,56+,57+,58-,59+,62-,63-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50001714

(2-[3-cyclopropylmethyl-11-hydroxy-15-methoxy-(1S,2...)Show SMILES CO[C@]12CC[C@@]3(C[C@@H]1C(C)(C)O)[C@H]1Cc4ccc(O)c5O[C@@H]2[C@]3(CCN1CC1CC1)c45 |THB:8:7:22.21:4.3| Show InChI InChI=1S/C26H35NO4/c1-23(2,29)18-13-24-8-9-26(18,30-3)22-25(24)10-11-27(14-15-4-5-15)19(24)12-16-6-7-17(28)21(31-22)20(16)25/h6-7,15,18-19,22,28-29H,4-5,8-14H2,1-3H3/t18-,19-,22-,24-,25+,26-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 3
(MOUSE) | BDBM50254239

(somatostatin-28)Show SMILES CSCC[C@H](NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CO)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@H]1CSSC[C@H](NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC1=O)[C@@H](C)O)[C@@H](C)O)C(O)=O |r,wU:42.43,28.31,13.12,4.4,66.67,86.87,106.108,115.116,194.201,172.177,149.152,134.136,124.126,120.224,wD:47.48,34.39,20.25,8.8,54.55,62.64,77.78,97.98,202.209,183.189,158.161,145.218,211.220,130.221,214.223,(-22.23,-7.83,;-23.64,-7.23,;-24.87,-8.16,;-26.29,-7.56,;-27.51,-8.49,;-28.93,-7.9,;-29.12,-6.37,;-27.9,-5.44,;-30.54,-5.78,;-31.77,-6.71,;-30.73,-4.25,;-32.15,-3.66,;-33.4,-4.59,;-32.34,-2.13,;-31.06,-1.26,;-31.8,.25,;-33.45,.03,;-33.78,-1.63,;-35.09,-2.39,;-35.19,-3.93,;-36.39,-1.55,;-37.77,-2.23,;-37.83,-3.76,;-39.2,-4.44,;-36.56,-4.61,;-36.28,-.01,;-37.58,.85,;-38.94,.16,;-37.46,2.37,;-36.09,3.05,;-34.83,2.21,;-38.75,3.22,;-38.65,4.76,;-37.27,5.45,;-39.93,5.6,;-41.31,4.92,;-41.42,3.4,;-40.12,2.54,;-42.8,2.71,;-39.84,7.14,;-41.12,8,;-42.5,7.31,;-41.02,9.53,;-39.65,10.21,;-42.31,10.39,;-42.21,11.93,;-40.83,12.61,;-43.49,12.76,;-43.38,14.3,;-44.87,12.08,;-44.96,10.56,;-27.32,-10.02,;-28.56,-10.95,;-25.9,-10.61,;-25.71,-12.14,;-26.93,-13.07,;-24.29,-12.75,;-23.09,-11.81,;-24.1,-14.26,;-25.25,-15.51,;-24.43,-16.97,;-22.76,-16.63,;-22.84,-15.08,;-21.53,-14.32,;-21.53,-12.78,;-20.18,-15.08,;-18.84,-14.32,;-18.84,-12.78,;-17.53,-12.02,;-17.53,-10.47,;-16.2,-9.71,;-16.2,-8.17,;-17.52,-7.41,;-14.86,-7.41,;-17.53,-15.08,;-17.53,-16.63,;-16.18,-14.32,;-14.85,-15.09,;-14.85,-16.63,;-13.54,-17.39,;-13.54,-18.94,;-14.87,-19.72,;-12.18,-19.71,;-13.54,-14.32,;-13.54,-12.78,;-12.19,-15.09,;-10.86,-14.33,;-10.86,-12.79,;-9.51,-12.02,;-9.51,-10.48,;-8.18,-9.72,;-8.18,-8.18,;-9.52,-7.42,;-6.84,-7.41,;-9.51,-15.11,;-9.51,-16.63,;-8.2,-14.33,;-6.85,-15.11,;-6.85,-16.64,;-5.52,-17.42,;-5.52,-18.94,;-4.2,-19.72,;-4.2,-21.27,;-5.52,-14.33,;-5.52,-12.79,;-4.19,-15.11,;-2.85,-14.33,;-2.85,-12.81,;-1.53,-15.11,;-1.53,-16.64,;-.2,-14.34,;1.14,-15.12,;2.46,-14.34,;2.46,-12.82,;3.81,-15.12,;5.13,-14.34,;5.14,-3.92,;6.49,-3.13,;50.46,-3.19,;50.41,-14.4,;49.07,-15.18,;47.74,-14.4,;46.4,-15.18,;46.4,-16.7,;45.07,-14.39,;45.07,-12.87,;46.4,-12.09,;43.75,-15.17,;42.41,-14.39,;42.41,-12.87,;41.08,-15.17,;39.75,-14.39,;38.42,-15.15,;38.42,-16.7,;37.09,-14.39,;37.09,-12.85,;38.42,-12.08,;39.76,-12.87,;41.08,-12.09,;41.09,-10.55,;39.76,-9.78,;38.42,-10.54,;35.75,-15.15,;34.42,-14.38,;34.42,-12.84,;33.1,-15.15,;31.76,-14.38,;30.44,-15.14,;30.44,-16.69,;29.09,-14.38,;29.09,-12.84,;30.44,-12.08,;30.44,-10.54,;31.76,-9.77,;31.76,-8.23,;27.77,-15.14,;26.44,-14.38,;26.44,-12.84,;25.1,-15.14,;25.1,-16.68,;26.44,-17.45,;27.84,-16.82,;28.87,-17.98,;28.09,-19.3,;28.57,-20.76,;27.53,-21.91,;26.03,-21.59,;25.57,-20.12,;26.59,-18.98,;23.78,-14.38,;22.44,-15.14,;22.44,-16.68,;21.11,-14.37,;21.11,-12.83,;22.44,-12.07,;23.78,-12.83,;25.1,-12.07,;25.1,-10.53,;23.78,-9.77,;22.44,-10.53,;19.79,-15.13,;18.44,-14.37,;18.44,-12.83,;17.12,-15.13,;17.12,-16.68,;18.44,-17.44,;19.76,-16.68,;21.1,-17.45,;21.09,-18.99,;19.75,-19.75,;18.44,-18.98,;15.78,-14.37,;14.45,-15.13,;14.45,-16.67,;13.11,-14.37,;13.11,-12.83,;14.45,-12.04,;15.78,-12.83,;14.45,-10.52,;11.79,-15.13,;10.46,-14.36,;10.46,-12.82,;9.12,-15.12,;9.12,-16.67,;10.46,-17.43,;10.46,-18.98,;11.78,-19.74,;11.78,-21.28,;7.79,-14.36,;6.47,-15.12,;6.47,-16.67,;33.1,-16.69,;31.76,-17.46,;34.42,-17.45,;41.08,-16.7,;39.75,-17.48,;42.41,-17.48,;49.07,-16.71,;47.74,-17.49,;50.41,-17.49,)| Show InChI InChI=1S/C137H207N41O39S3/c1-69(154-113(194)82(37-19-22-47-138)159-115(196)85(40-25-50-149-136(145)146)160-118(199)87(44-45-106(188)189)163-116(197)86(41-26-51-150-137(147)148)164-130(211)101-43-27-52-177(101)133(214)72(4)156-114(195)88(46-54-218-7)158-110(191)71(3)155-129(210)100-42-28-53-178(100)134(215)95(61-104(144)186)171-126(207)96(65-180)172-124(205)93(59-102(142)184)165-111(192)70(2)153-112(193)80(141)64-179)109(190)152-63-105(187)157-98-67-219-220-68-99(135(216)217)174-127(208)97(66-181)173-132(213)108(74(6)183)176-125(206)91(57-77-33-15-10-16-34-77)170-131(212)107(73(5)182)175-119(200)84(39-21-24-49-140)161-122(203)92(58-78-62-151-81-36-18-17-35-79(78)81)168-121(202)90(56-76-31-13-9-14-32-76)166-120(201)89(55-75-29-11-8-12-30-75)167-123(204)94(60-103(143)185)169-117(198)83(162-128(98)209)38-20-23-48-139/h8-18,29-36,62,69-74,80,82-101,107-108,151,179-183H,19-28,37-61,63-68,138-141H2,1-7H3,(H2,142,184)(H2,143,185)(H2,144,186)(H,152,190)(H,153,193)(H,154,194)(H,155,210)(H,156,195)(H,157,187)(H,158,191)(H,159,196)(H,160,199)(H,161,203)(H,162,209)(H,163,197)(H,164,211)(H,165,192)(H,166,201)(H,167,204)(H,168,202)(H,169,198)(H,170,212)(H,171,207)(H,172,205)(H,173,213)(H,174,208)(H,175,200)(H,176,206)(H,188,189)(H,216,217)(H4,145,146,149)(H4,147,148,150)/t69-,70-,71-,72-,73+,74+,80-,82-,83-,84-,85-,86-,87-,88-,89-,90-,91-,92-,93-,94-,95-,96-,97-,98-,99-,100-,101-,107-,108-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 1
(Homo sapiens (Human)) | BDBM50254239

(somatostatin-28)Show SMILES CSCC[C@H](NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CO)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@H]1CSSC[C@H](NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC1=O)[C@@H](C)O)[C@@H](C)O)C(O)=O |r,wU:42.43,28.31,13.12,4.4,66.67,86.87,106.108,115.116,194.201,172.177,149.152,134.136,124.126,120.224,wD:47.48,34.39,20.25,8.8,54.55,62.64,77.78,97.98,202.209,183.189,158.161,145.218,211.220,130.221,214.223,(-22.23,-7.83,;-23.64,-7.23,;-24.87,-8.16,;-26.29,-7.56,;-27.51,-8.49,;-28.93,-7.9,;-29.12,-6.37,;-27.9,-5.44,;-30.54,-5.78,;-31.77,-6.71,;-30.73,-4.25,;-32.15,-3.66,;-33.4,-4.59,;-32.34,-2.13,;-31.06,-1.26,;-31.8,.25,;-33.45,.03,;-33.78,-1.63,;-35.09,-2.39,;-35.19,-3.93,;-36.39,-1.55,;-37.77,-2.23,;-37.83,-3.76,;-39.2,-4.44,;-36.56,-4.61,;-36.28,-.01,;-37.58,.85,;-38.94,.16,;-37.46,2.37,;-36.09,3.05,;-34.83,2.21,;-38.75,3.22,;-38.65,4.76,;-37.27,5.45,;-39.93,5.6,;-41.31,4.92,;-41.42,3.4,;-40.12,2.54,;-42.8,2.71,;-39.84,7.14,;-41.12,8,;-42.5,7.31,;-41.02,9.53,;-39.65,10.21,;-42.31,10.39,;-42.21,11.93,;-40.83,12.61,;-43.49,12.76,;-43.38,14.3,;-44.87,12.08,;-44.96,10.56,;-27.32,-10.02,;-28.56,-10.95,;-25.9,-10.61,;-25.71,-12.14,;-26.93,-13.07,;-24.29,-12.75,;-23.09,-11.81,;-24.1,-14.26,;-25.25,-15.51,;-24.43,-16.97,;-22.76,-16.63,;-22.84,-15.08,;-21.53,-14.32,;-21.53,-12.78,;-20.18,-15.08,;-18.84,-14.32,;-18.84,-12.78,;-17.53,-12.02,;-17.53,-10.47,;-16.2,-9.71,;-16.2,-8.17,;-17.52,-7.41,;-14.86,-7.41,;-17.53,-15.08,;-17.53,-16.63,;-16.18,-14.32,;-14.85,-15.09,;-14.85,-16.63,;-13.54,-17.39,;-13.54,-18.94,;-14.87,-19.72,;-12.18,-19.71,;-13.54,-14.32,;-13.54,-12.78,;-12.19,-15.09,;-10.86,-14.33,;-10.86,-12.79,;-9.51,-12.02,;-9.51,-10.48,;-8.18,-9.72,;-8.18,-8.18,;-9.52,-7.42,;-6.84,-7.41,;-9.51,-15.11,;-9.51,-16.63,;-8.2,-14.33,;-6.85,-15.11,;-6.85,-16.64,;-5.52,-17.42,;-5.52,-18.94,;-4.2,-19.72,;-4.2,-21.27,;-5.52,-14.33,;-5.52,-12.79,;-4.19,-15.11,;-2.85,-14.33,;-2.85,-12.81,;-1.53,-15.11,;-1.53,-16.64,;-.2,-14.34,;1.14,-15.12,;2.46,-14.34,;2.46,-12.82,;3.81,-15.12,;5.13,-14.34,;5.14,-3.92,;6.49,-3.13,;50.46,-3.19,;50.41,-14.4,;49.07,-15.18,;47.74,-14.4,;46.4,-15.18,;46.4,-16.7,;45.07,-14.39,;45.07,-12.87,;46.4,-12.09,;43.75,-15.17,;42.41,-14.39,;42.41,-12.87,;41.08,-15.17,;39.75,-14.39,;38.42,-15.15,;38.42,-16.7,;37.09,-14.39,;37.09,-12.85,;38.42,-12.08,;39.76,-12.87,;41.08,-12.09,;41.09,-10.55,;39.76,-9.78,;38.42,-10.54,;35.75,-15.15,;34.42,-14.38,;34.42,-12.84,;33.1,-15.15,;31.76,-14.38,;30.44,-15.14,;30.44,-16.69,;29.09,-14.38,;29.09,-12.84,;30.44,-12.08,;30.44,-10.54,;31.76,-9.77,;31.76,-8.23,;27.77,-15.14,;26.44,-14.38,;26.44,-12.84,;25.1,-15.14,;25.1,-16.68,;26.44,-17.45,;27.84,-16.82,;28.87,-17.98,;28.09,-19.3,;28.57,-20.76,;27.53,-21.91,;26.03,-21.59,;25.57,-20.12,;26.59,-18.98,;23.78,-14.38,;22.44,-15.14,;22.44,-16.68,;21.11,-14.37,;21.11,-12.83,;22.44,-12.07,;23.78,-12.83,;25.1,-12.07,;25.1,-10.53,;23.78,-9.77,;22.44,-10.53,;19.79,-15.13,;18.44,-14.37,;18.44,-12.83,;17.12,-15.13,;17.12,-16.68,;18.44,-17.44,;19.76,-16.68,;21.1,-17.45,;21.09,-18.99,;19.75,-19.75,;18.44,-18.98,;15.78,-14.37,;14.45,-15.13,;14.45,-16.67,;13.11,-14.37,;13.11,-12.83,;14.45,-12.04,;15.78,-12.83,;14.45,-10.52,;11.79,-15.13,;10.46,-14.36,;10.46,-12.82,;9.12,-15.12,;9.12,-16.67,;10.46,-17.43,;10.46,-18.98,;11.78,-19.74,;11.78,-21.28,;7.79,-14.36,;6.47,-15.12,;6.47,-16.67,;33.1,-16.69,;31.76,-17.46,;34.42,-17.45,;41.08,-16.7,;39.75,-17.48,;42.41,-17.48,;49.07,-16.71,;47.74,-17.49,;50.41,-17.49,)| Show InChI InChI=1S/C137H207N41O39S3/c1-69(154-113(194)82(37-19-22-47-138)159-115(196)85(40-25-50-149-136(145)146)160-118(199)87(44-45-106(188)189)163-116(197)86(41-26-51-150-137(147)148)164-130(211)101-43-27-52-177(101)133(214)72(4)156-114(195)88(46-54-218-7)158-110(191)71(3)155-129(210)100-42-28-53-178(100)134(215)95(61-104(144)186)171-126(207)96(65-180)172-124(205)93(59-102(142)184)165-111(192)70(2)153-112(193)80(141)64-179)109(190)152-63-105(187)157-98-67-219-220-68-99(135(216)217)174-127(208)97(66-181)173-132(213)108(74(6)183)176-125(206)91(57-77-33-15-10-16-34-77)170-131(212)107(73(5)182)175-119(200)84(39-21-24-49-140)161-122(203)92(58-78-62-151-81-36-18-17-35-79(78)81)168-121(202)90(56-76-31-13-9-14-32-76)166-120(201)89(55-75-29-11-8-12-30-75)167-123(204)94(60-103(143)185)169-117(198)83(162-128(98)209)38-20-23-48-139/h8-18,29-36,62,69-74,80,82-101,107-108,151,179-183H,19-28,37-61,63-68,138-141H2,1-7H3,(H2,142,184)(H2,143,185)(H2,144,186)(H,152,190)(H,153,193)(H,154,194)(H,155,210)(H,156,195)(H,157,187)(H,158,191)(H,159,196)(H,160,199)(H,161,203)(H,162,209)(H,163,197)(H,164,211)(H,165,192)(H,166,201)(H,167,204)(H,168,202)(H,169,198)(H,170,212)(H,171,207)(H,172,205)(H,173,213)(H,174,208)(H,175,200)(H,176,206)(H,188,189)(H,216,217)(H4,145,146,149)(H4,147,148,150)/t69-,70-,71-,72-,73+,74+,80-,82-,83-,84-,85-,86-,87-,88-,89-,90-,91-,92-,93-,94-,95-,96-,97-,98-,99-,100-,101-,107-,108-/m0/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM81766
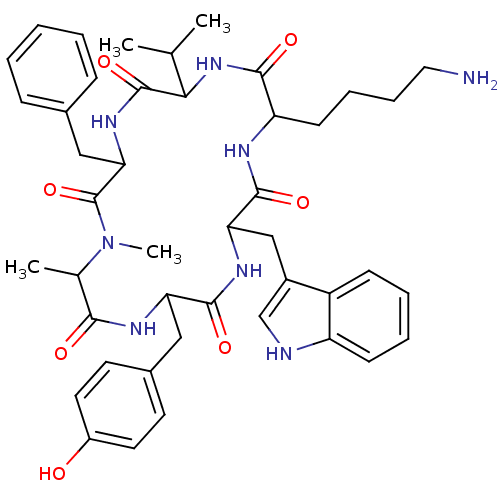
(CAS_3086456 | MK 678 | NSC_3086456)Show SMILES CC(C)C1NC(=O)C(CCCCN)NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(Cc2ccc(O)cc2)NC(=O)C(C)N(C)C(=O)C(Cc2ccccc2)NC1=O Show InChI InChI=1S/C44H56N8O7/c1-26(2)38-43(58)50-37(23-28-12-6-5-7-13-28)44(59)52(4)27(3)39(54)48-35(22-29-17-19-31(53)20-18-29)41(56)49-36(24-30-25-46-33-15-9-8-14-32(30)33)42(57)47-34(40(55)51-38)16-10-11-21-45/h5-9,12-15,17-20,25-27,34-38,46,53H,10-11,16,21-24,45H2,1-4H3,(H,47,57)(H,48,54)(H,49,56)(H,50,58)(H,51,55) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Chicago
Curated by PDSP Ki Database
| |
J Biol Chem 267: 20422-8 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6PPB |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(Homo sapiens (Human)) | BDBM81766

(CAS_3086456 | MK 678 | NSC_3086456)Show SMILES CC(C)C1NC(=O)C(CCCCN)NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(Cc2ccc(O)cc2)NC(=O)C(C)N(C)C(=O)C(Cc2ccccc2)NC1=O Show InChI InChI=1S/C44H56N8O7/c1-26(2)38-43(58)50-37(23-28-12-6-5-7-13-28)44(59)52(4)27(3)39(54)48-35(22-29-17-19-31(53)20-18-29)41(56)49-36(24-30-25-46-33-15-9-8-14-32(30)33)42(57)47-34(40(55)51-38)16-10-11-21-45/h5-9,12-15,17-20,25-27,34-38,46,53H,10-11,16,21-24,45H2,1-4H3,(H,47,57)(H,48,54)(H,49,56)(H,50,58)(H,51,55) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Endocr Rev 16: 427-42 (1995)
Article DOI: 10.1210/edrv-16-4-427
BindingDB Entry DOI: 10.7270/Q24B2ZTD |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 3
(MOUSE) | BDBM82457
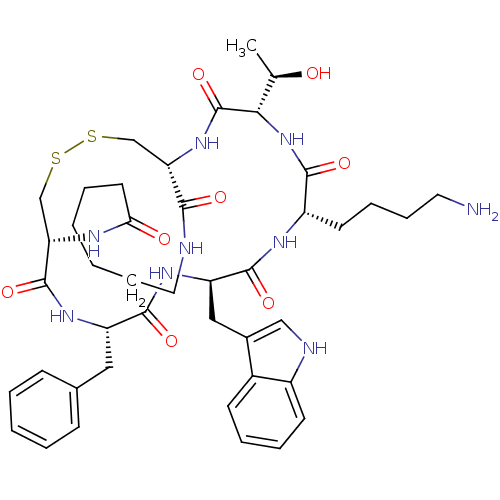
((1R,4S,7S,10R,13S,16R)-4-[(R)-1-Hydroxyethyl]-7-(4...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H]2CSSC[C@H](NC1=O)C(=O)NCCCCCCC(=O)N2 Show InChI InChI=1S/C43H59N9O8S2/c1-26(53)37-43(60)51-34-24-61-62-25-35(47-36(54)18-7-2-3-12-20-45-38(34)55)42(59)49-32(21-27-13-5-4-6-14-27)40(57)50-33(22-28-23-46-30-16-9-8-15-29(28)30)41(58)48-31(39(56)52-37)17-10-11-19-44/h4-6,8-9,13-16,23,26,31-35,37,46,53H,2-3,7,10-12,17-22,24-25,44H2,1H3,(H,45,55)(H,47,54)(H,48,58)(H,49,59)(H,50,57)(H,51,60)(H,52,56)/t26-,31+,32+,33-,34+,35+,37+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(RAT) | BDBM82462

(BIM 23059 | D-Nal-c[Cys-Tyr-D-Trp-Lys-Thr-Cys]-Thr...)Show SMILES CC(CO)C(NC(=O)C1NC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@H](Cc2c[nH]c3ccccc23)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C(C)CO)C(=O)NC(SS1)C(=O)N[C@@H](Cc1ccc2ccccc2c1)C(N)=O)C(N)=O |r| Show InChI InChI=1S/C54H68N12O12S2/c1-28(26-67)42(45(57)71)63-51(77)53-66-54(78)62-40(22-30-15-18-35(69)19-16-30)47(73)61-41(24-34-25-58-37-12-6-5-11-36(34)37)48(74)59-38(13-7-8-20-55)46(72)64-43(29(2)27-68)49(75)65-52(79-80-53)50(76)60-39(44(56)70)23-31-14-17-32-9-3-4-10-33(32)21-31/h3-6,9-12,14-19,21,25,28-29,38-43,52-53,58,67-69H,7-8,13,20,22-24,26-27,55H2,1-2H3,(H2,56,70)(H2,57,71)(H,59,74)(H,60,76)(H,61,73)(H,63,77)(H,64,72)(H,65,75)(H2,62,66,78)/t28?,29?,38-,39-,40-,41+,42?,43-,52?,53?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 44: 385-92 (1993)
BindingDB Entry DOI: 10.7270/Q2X065KZ |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50013388
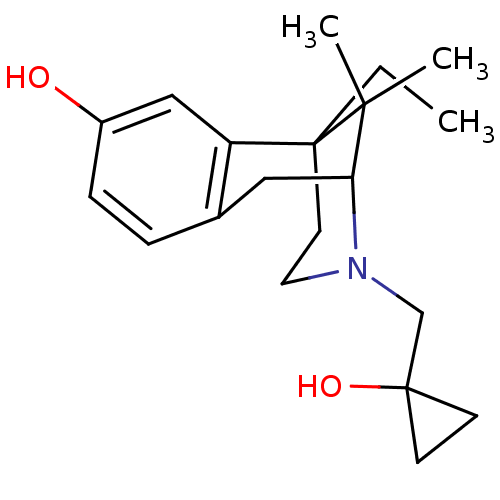
(6-Ethyl-3-(1-hydroxy-cyclopropylmethyl)-11,11-dime...)Show SMILES CCC12CCN(CC3(O)CC3)C(Cc3ccc(O)cc13)C2(C)C |TLB:6:5:20:19.13.12| Show InChI InChI=1S/C20H29NO2/c1-4-20-9-10-21(13-19(23)7-8-19)17(18(20,2)3)11-14-5-6-15(22)12-16(14)20/h5-6,12,17,22-23H,4,7-11,13H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM82554

(Beta C N A | CAS_115070 | NSC_115070)Show SMILES Oc1ccc2CC3N(CC4CC4)CCC45C(Oc1c24)C(CCC35O)N(CCCl)CCCl |TLB:21:22:7.12.13:5.4.18,THB:23:22:7.12.13:5.4.18| Show InChI InChI=1S/C24H32Cl2N2O3/c25-8-11-27(12-9-26)17-5-6-24(30)19-13-16-3-4-18(29)21-20(16)23(24,22(17)31-21)7-10-28(19)14-15-1-2-15/h3-4,15,17,19,22,29-30H,1-2,5-14H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(Homo sapiens (Human)) | BDBM50019568
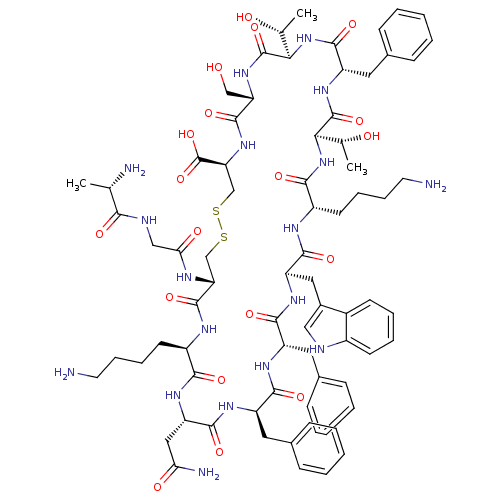
(Ala-Gly-c(Cys-Lys-Asn-Phe-Phe-Trp-Lys-Thr-Phe-Thr-...)Show SMILES C[C@@H](O)[C@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](CCCCN)NC(=O)[C@@H](CSSC[C@H](NC(=O)[C@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)[C@@H](C)O Show InChI InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42+,43+,50+,51-,52+,53-,54-,55+,56-,57-,58+,59-,62+,63+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Endocr Rev 16: 427-42 (1995)
Article DOI: 10.1210/edrv-16-4-427
BindingDB Entry DOI: 10.7270/Q24B2ZTD |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM81767
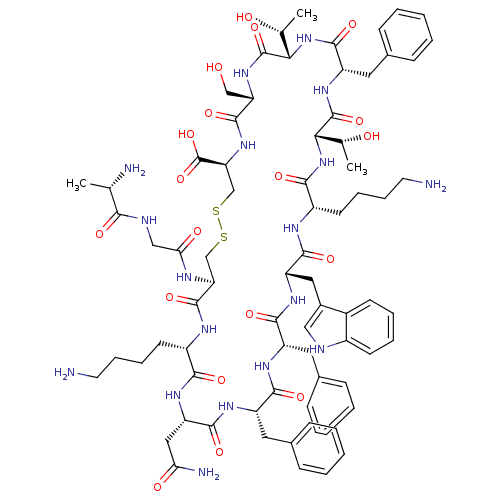
(15-28-Somatostatin-28 | CAS_38916-34-6 | CB6417646...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@H](NC(=O)[C@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)[C@@H](C)O |r| Show InChI InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42+,43+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,62-,63-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Chicago
Curated by PDSP Ki Database
| |
J Biol Chem 267: 20422-8 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6PPB |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 3
(MOUSE) | BDBM81767

(15-28-Somatostatin-28 | CAS_38916-34-6 | CB6417646...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@H](NC(=O)[C@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)[C@@H](C)O |r| Show InChI InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42+,43+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,62-,63-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM82427
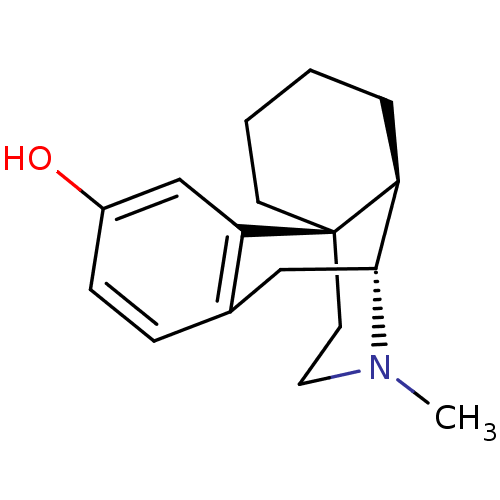
(CAS_5985-38-6 | LEVORPHANOL-tartarate)Show SMILES CN1CC[C@]23CCCC[C@H]2[C@H]1Cc1ccc(O)cc31 |r| Show InChI InChI=1S/C17H23NO/c1-18-9-8-17-7-3-2-4-14(17)16(18)10-12-5-6-13(19)11-15(12)17/h5-6,11,14,16,19H,2-4,7-10H2,1H3/t14-,16+,17+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
PC cid
PC sid
UniChem
Similars
| DrugBank
Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(RAT) | BDBM84630

(BIM 23060)Show SMILES CC(O)C1NC(=O)C(CCCCN)NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(Cc2ccc(O)cc2)NC(=O)C(CSSCC(NC1=O)C(=O)NCc1cccc2ccccc12)NC(=O)C(N)Cc1ccccc1 Show InChI InChI=1S/C56H66N10O9S2/c1-33(67)49-56(75)65-47(51(70)60-29-37-16-11-15-36-14-5-6-17-40(36)37)31-76-77-32-48(64-50(69)42(58)26-34-12-3-2-4-13-34)55(74)62-45(27-35-21-23-39(68)24-22-35)53(72)63-46(28-38-30-59-43-19-8-7-18-41(38)43)54(73)61-44(52(71)66-49)20-9-10-25-57/h2-8,11-19,21-24,30,33,42,44-49,59,67-68H,9-10,20,25-29,31-32,57-58H2,1H3,(H,60,70)(H,61,73)(H,62,74)(H,63,72)(H,64,69)(H,65,75)(H,66,71) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 44: 385-92 (1993)
BindingDB Entry DOI: 10.7270/Q2X065KZ |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 1
(Homo sapiens (Human)) | BDBM81767

(15-28-Somatostatin-28 | CAS_38916-34-6 | CB6417646...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@H](NC(=O)[C@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)[C@@H](C)O |r| Show InChI InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42+,43+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,62-,63-/m0/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(Homo sapiens (Human)) | BDBM50254239

(somatostatin-28)Show SMILES CSCC[C@H](NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CO)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@H]1CSSC[C@H](NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC1=O)[C@@H](C)O)[C@@H](C)O)C(O)=O |r,wU:42.43,28.31,13.12,4.4,66.67,86.87,106.108,115.116,194.201,172.177,149.152,134.136,124.126,120.224,wD:47.48,34.39,20.25,8.8,54.55,62.64,77.78,97.98,202.209,183.189,158.161,145.218,211.220,130.221,214.223,(-22.23,-7.83,;-23.64,-7.23,;-24.87,-8.16,;-26.29,-7.56,;-27.51,-8.49,;-28.93,-7.9,;-29.12,-6.37,;-27.9,-5.44,;-30.54,-5.78,;-31.77,-6.71,;-30.73,-4.25,;-32.15,-3.66,;-33.4,-4.59,;-32.34,-2.13,;-31.06,-1.26,;-31.8,.25,;-33.45,.03,;-33.78,-1.63,;-35.09,-2.39,;-35.19,-3.93,;-36.39,-1.55,;-37.77,-2.23,;-37.83,-3.76,;-39.2,-4.44,;-36.56,-4.61,;-36.28,-.01,;-37.58,.85,;-38.94,.16,;-37.46,2.37,;-36.09,3.05,;-34.83,2.21,;-38.75,3.22,;-38.65,4.76,;-37.27,5.45,;-39.93,5.6,;-41.31,4.92,;-41.42,3.4,;-40.12,2.54,;-42.8,2.71,;-39.84,7.14,;-41.12,8,;-42.5,7.31,;-41.02,9.53,;-39.65,10.21,;-42.31,10.39,;-42.21,11.93,;-40.83,12.61,;-43.49,12.76,;-43.38,14.3,;-44.87,12.08,;-44.96,10.56,;-27.32,-10.02,;-28.56,-10.95,;-25.9,-10.61,;-25.71,-12.14,;-26.93,-13.07,;-24.29,-12.75,;-23.09,-11.81,;-24.1,-14.26,;-25.25,-15.51,;-24.43,-16.97,;-22.76,-16.63,;-22.84,-15.08,;-21.53,-14.32,;-21.53,-12.78,;-20.18,-15.08,;-18.84,-14.32,;-18.84,-12.78,;-17.53,-12.02,;-17.53,-10.47,;-16.2,-9.71,;-16.2,-8.17,;-17.52,-7.41,;-14.86,-7.41,;-17.53,-15.08,;-17.53,-16.63,;-16.18,-14.32,;-14.85,-15.09,;-14.85,-16.63,;-13.54,-17.39,;-13.54,-18.94,;-14.87,-19.72,;-12.18,-19.71,;-13.54,-14.32,;-13.54,-12.78,;-12.19,-15.09,;-10.86,-14.33,;-10.86,-12.79,;-9.51,-12.02,;-9.51,-10.48,;-8.18,-9.72,;-8.18,-8.18,;-9.52,-7.42,;-6.84,-7.41,;-9.51,-15.11,;-9.51,-16.63,;-8.2,-14.33,;-6.85,-15.11,;-6.85,-16.64,;-5.52,-17.42,;-5.52,-18.94,;-4.2,-19.72,;-4.2,-21.27,;-5.52,-14.33,;-5.52,-12.79,;-4.19,-15.11,;-2.85,-14.33,;-2.85,-12.81,;-1.53,-15.11,;-1.53,-16.64,;-.2,-14.34,;1.14,-15.12,;2.46,-14.34,;2.46,-12.82,;3.81,-15.12,;5.13,-14.34,;5.14,-3.92,;6.49,-3.13,;50.46,-3.19,;50.41,-14.4,;49.07,-15.18,;47.74,-14.4,;46.4,-15.18,;46.4,-16.7,;45.07,-14.39,;45.07,-12.87,;46.4,-12.09,;43.75,-15.17,;42.41,-14.39,;42.41,-12.87,;41.08,-15.17,;39.75,-14.39,;38.42,-15.15,;38.42,-16.7,;37.09,-14.39,;37.09,-12.85,;38.42,-12.08,;39.76,-12.87,;41.08,-12.09,;41.09,-10.55,;39.76,-9.78,;38.42,-10.54,;35.75,-15.15,;34.42,-14.38,;34.42,-12.84,;33.1,-15.15,;31.76,-14.38,;30.44,-15.14,;30.44,-16.69,;29.09,-14.38,;29.09,-12.84,;30.44,-12.08,;30.44,-10.54,;31.76,-9.77,;31.76,-8.23,;27.77,-15.14,;26.44,-14.38,;26.44,-12.84,;25.1,-15.14,;25.1,-16.68,;26.44,-17.45,;27.84,-16.82,;28.87,-17.98,;28.09,-19.3,;28.57,-20.76,;27.53,-21.91,;26.03,-21.59,;25.57,-20.12,;26.59,-18.98,;23.78,-14.38,;22.44,-15.14,;22.44,-16.68,;21.11,-14.37,;21.11,-12.83,;22.44,-12.07,;23.78,-12.83,;25.1,-12.07,;25.1,-10.53,;23.78,-9.77,;22.44,-10.53,;19.79,-15.13,;18.44,-14.37,;18.44,-12.83,;17.12,-15.13,;17.12,-16.68,;18.44,-17.44,;19.76,-16.68,;21.1,-17.45,;21.09,-18.99,;19.75,-19.75,;18.44,-18.98,;15.78,-14.37,;14.45,-15.13,;14.45,-16.67,;13.11,-14.37,;13.11,-12.83,;14.45,-12.04,;15.78,-12.83,;14.45,-10.52,;11.79,-15.13,;10.46,-14.36,;10.46,-12.82,;9.12,-15.12,;9.12,-16.67,;10.46,-17.43,;10.46,-18.98,;11.78,-19.74,;11.78,-21.28,;7.79,-14.36,;6.47,-15.12,;6.47,-16.67,;33.1,-16.69,;31.76,-17.46,;34.42,-17.45,;41.08,-16.7,;39.75,-17.48,;42.41,-17.48,;49.07,-16.71,;47.74,-17.49,;50.41,-17.49,)| Show InChI InChI=1S/C137H207N41O39S3/c1-69(154-113(194)82(37-19-22-47-138)159-115(196)85(40-25-50-149-136(145)146)160-118(199)87(44-45-106(188)189)163-116(197)86(41-26-51-150-137(147)148)164-130(211)101-43-27-52-177(101)133(214)72(4)156-114(195)88(46-54-218-7)158-110(191)71(3)155-129(210)100-42-28-53-178(100)134(215)95(61-104(144)186)171-126(207)96(65-180)172-124(205)93(59-102(142)184)165-111(192)70(2)153-112(193)80(141)64-179)109(190)152-63-105(187)157-98-67-219-220-68-99(135(216)217)174-127(208)97(66-181)173-132(213)108(74(6)183)176-125(206)91(57-77-33-15-10-16-34-77)170-131(212)107(73(5)182)175-119(200)84(39-21-24-49-140)161-122(203)92(58-78-62-151-81-36-18-17-35-79(78)81)168-121(202)90(56-76-31-13-9-14-32-76)166-120(201)89(55-75-29-11-8-12-30-75)167-123(204)94(60-103(143)185)169-117(198)83(162-128(98)209)38-20-23-48-139/h8-18,29-36,62,69-74,80,82-101,107-108,151,179-183H,19-28,37-61,63-68,138-141H2,1-7H3,(H2,142,184)(H2,143,185)(H2,144,186)(H,152,190)(H,153,193)(H,154,194)(H,155,210)(H,156,195)(H,157,187)(H,158,191)(H,159,196)(H,160,199)(H,161,203)(H,162,209)(H,163,197)(H,164,211)(H,165,192)(H,166,201)(H,167,204)(H,168,202)(H,169,198)(H,170,212)(H,171,207)(H,172,205)(H,173,213)(H,174,208)(H,175,200)(H,176,206)(H,188,189)(H,216,217)(H4,145,146,149)(H4,147,148,150)/t69-,70-,71-,72-,73+,74+,80-,82-,83-,84-,85-,86-,87-,88-,89-,90-,91-,92-,93-,94-,95-,96-,97-,98-,99-,100-,101-,107-,108-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Endocr Rev 16: 427-42 (1995)
Article DOI: 10.1210/edrv-16-4-427
BindingDB Entry DOI: 10.7270/Q24B2ZTD |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(RAT) | BDBM82470
(3-(2-Naphtyl)-D-Ala-L-Cys(1)-L-Tyr-D-Trp-L-Lys-L-V...)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@H](CSSC[C@H](NC1=O)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)NC(=O)[C@H](N)Cc1ccc2ccccc2c1 Show InChI InChI=1S/C54H69N11O10S2/c1-29(2)45-54(75)63-44(53(74)65-46(30(3)66)47(57)68)28-77-76-27-43(62-48(69)38(56)23-32-15-18-33-10-4-5-11-34(33)22-32)52(73)60-41(24-31-16-19-36(67)20-17-31)50(71)61-42(25-35-26-58-39-13-7-6-12-37(35)39)51(72)59-40(49(70)64-45)14-8-9-21-55/h4-7,10-13,15-20,22,26,29-30,38,40-46,58,66-67H,8-9,14,21,23-25,27-28,55-56H2,1-3H3,(H2,57,68)(H,59,72)(H,60,73)(H,61,71)(H,62,69)(H,63,75)(H,64,70)(H,65,74)/t30-,38-,40+,41+,42-,43+,44+,45+,46+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 44: 385-92 (1993)
BindingDB Entry DOI: 10.7270/Q2X065KZ |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50169774
(2-(3,4-Dichloro-phenyl)-N-methyl-N-(2-pyrrolidin-1...)Show SMILES CN(C1CCCC[C@H]1N1CCCC1)C(=O)Cc1ccc(Cl)c(Cl)c1 Show InChI InChI=1S/C19H26Cl2N2O/c1-22(19(24)13-14-8-9-15(20)16(21)12-14)17-6-2-3-7-18(17)23-10-4-5-11-23/h8-9,12,17-18H,2-7,10-11,13H2,1H3/t17?,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM82425
(CAS_61677 | ETORPHINE - HCI | NSC_61677)Show SMILES CCCC(C)(O)C1CC23C=CC1(OC)C1Oc4c5c(CC2N(C)CCC315)ccc4O |c:9,TLB:16:17:8:24.23.21,7:8:24.23.21:19.18.17,9:8:24.23.21:19.18.17,THB:22:21:8:19.18.17,(7.51,1.44,;6.12,.92,;4.97,1.86,;3.58,1.33,;3.06,2.72,;4.11,-.06,;2.19,.81,;1.92,-.83,;.53,-1.78,;.54,-.47,;1.16,.67,;.87,1.89,;.87,3.38,;-.41,4.13,;-.43,.85,;-1.52,1.9,;-2.75,1.03,;-2.22,-.44,;-3.43,-1.4,;-2.64,-2.71,;-1.09,-2.63,;-1.24,-4.18,;-2.27,-5.25,;.23,-3.98,;-.13,-2.47,;-.69,-.85,;-4.92,-.98,;-5.27,.51,;-4.18,1.56,;-4.48,3.02,)| Show InChI InChI=1S/C25H33NO4/c1-5-8-22(2,28)17-14-23-9-10-25(17,29-4)21-24(23)11-12-26(3)18(23)13-15-6-7-16(27)20(30-21)19(15)24/h6-7,9-10,17-18,21,27-28H,5,8,11-14H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM94503
(2-hydroxypropane-1,2,3-tricarboxylic acid;N-[4-(me...)Show InChI InChI=1S/C22H30N2O2S/c1-3-21(25)24(19-8-5-4-6-9-19)22(18-26-2)12-15-23(16-13-22)14-11-20-10-7-17-27-20/h4-10,17H,3,11-16,18H2,1-2H3 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
PC cid
PC sid
UniChem
Similars
| DrugBank
Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM84624
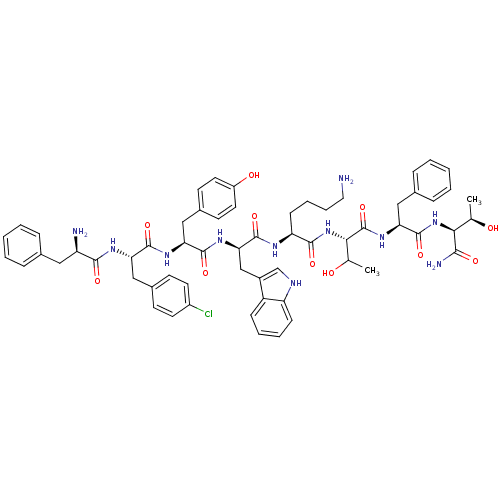
(BIM 23068)Show SMILES C[C@@H](O)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(Cl)cc1)NC(=O)[C@H](N)Cc1ccccc1)C(C)O)C(N)=O Show InChI InChI=1S/C61H74ClN11O11/c1-35(74)52(54(65)77)72-60(83)50(30-38-15-7-4-8-16-38)71-61(84)53(36(2)75)73-56(79)47(19-11-12-28-63)67-59(82)51(33-41-34-66-46-18-10-9-17-44(41)46)70-58(81)49(32-40-22-26-43(76)27-23-40)69-57(80)48(31-39-20-24-42(62)25-21-39)68-55(78)45(64)29-37-13-5-3-6-14-37/h3-10,13-18,20-27,34-36,45,47-53,66,74-76H,11-12,19,28-33,63-64H2,1-2H3,(H2,65,77)(H,67,82)(H,68,78)(H,69,80)(H,70,81)(H,71,84)(H,72,83)(H,73,79)/t35-,36?,45-,47+,48+,49+,50+,51-,52+,53+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(MOUSE) | BDBM81766

(CAS_3086456 | MK 678 | NSC_3086456)Show SMILES CC(C)C1NC(=O)C(CCCCN)NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(Cc2ccc(O)cc2)NC(=O)C(C)N(C)C(=O)C(Cc2ccccc2)NC1=O Show InChI InChI=1S/C44H56N8O7/c1-26(2)38-43(58)50-37(23-28-12-6-5-7-13-28)44(59)52(4)27(3)39(54)48-35(22-29-17-19-31(53)20-18-29)41(56)49-36(24-30-25-46-33-15-9-8-14-32(30)33)42(57)47-34(40(55)51-38)16-10-11-21-45/h5-9,12-15,17-20,25-27,34-38,46,53H,10-11,16,21-24,45H2,1-4H3,(H,47,57)(H,48,54)(H,49,56)(H,50,58)(H,51,55) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50019574
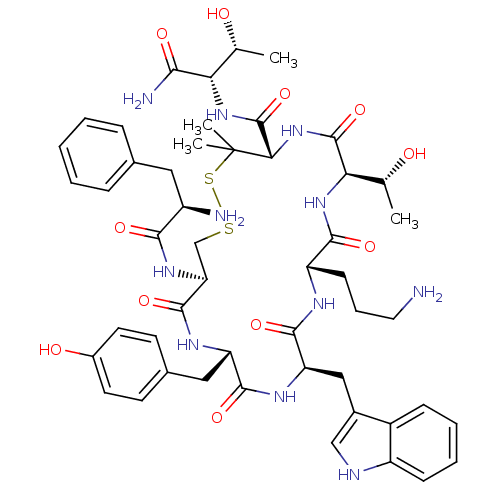
(CHEMBL406014 | CTOP | D-Phe-Cys-Tyr-D-Trp-Orn-Thr-...)Show SMILES C[C@@H](O)[C@H](NC(=O)[C@@H]1NC(=O)[C@H](NC(=O)[C@H](CCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@H](CSSC1(C)C)NC(=O)[C@H](N)Cc1ccccc1)[C@@H](C)O)C(N)=O Show InChI InChI=1S/C50H67N11O11S2/c1-26(62)39(42(53)65)59-49(72)41-50(3,4)74-73-25-38(58-43(66)33(52)21-28-11-6-5-7-12-28)47(70)56-36(22-29-16-18-31(64)19-17-29)45(68)57-37(23-30-24-54-34-14-9-8-13-32(30)34)46(69)55-35(15-10-20-51)44(67)60-40(27(2)63)48(71)61-41/h5-9,11-14,16-19,24,26-27,33,35-41,54,62-64H,10,15,20-23,25,51-52H2,1-4H3,(H2,53,65)(H,55,69)(H,56,70)(H,57,68)(H,58,66)(H,59,72)(H,60,67)(H,61,71)/t26-,27-,33-,35+,36+,37-,38+,39+,40-,41+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 45: 330-4 (1994)
Article DOI: 10.1016/j.bioorg.2015.02.008
BindingDB Entry DOI: 10.7270/Q21Z42X1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM82425

(CAS_61677 | ETORPHINE - HCI | NSC_61677)Show SMILES CCCC(C)(O)C1CC23C=CC1(OC)C1Oc4c5c(CC2N(C)CCC315)ccc4O |c:9,TLB:16:17:8:24.23.21,7:8:24.23.21:19.18.17,9:8:24.23.21:19.18.17,THB:22:21:8:19.18.17,(7.51,1.44,;6.12,.92,;4.97,1.86,;3.58,1.33,;3.06,2.72,;4.11,-.06,;2.19,.81,;1.92,-.83,;.53,-1.78,;.54,-.47,;1.16,.67,;.87,1.89,;.87,3.38,;-.41,4.13,;-.43,.85,;-1.52,1.9,;-2.75,1.03,;-2.22,-.44,;-3.43,-1.4,;-2.64,-2.71,;-1.09,-2.63,;-1.24,-4.18,;-2.27,-5.25,;.23,-3.98,;-.13,-2.47,;-.69,-.85,;-4.92,-.98,;-5.27,.51,;-4.18,1.56,;-4.48,3.02,)| Show InChI InChI=1S/C25H33NO4/c1-5-8-22(2,28)17-14-23-9-10-25(17,29-4)21-24(23)11-12-26(3)18(23)13-15-6-7-16(27)20(30-21)19(15)24/h6-7,9-10,17-18,21,27-28H,5,8,11-14H2,1-4H3 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 272: 423-8 (1995)
BindingDB Entry DOI: 10.7270/Q2TM78M9 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM21865
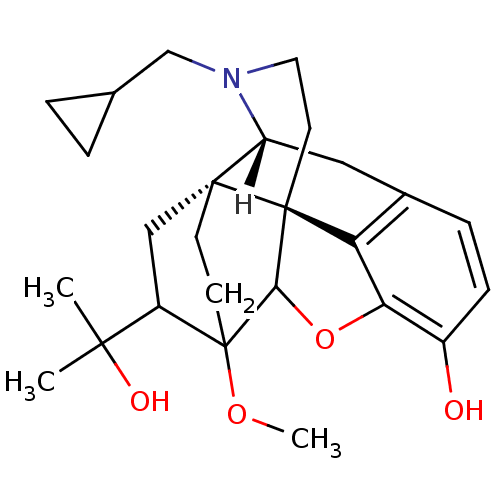
((14beta)-17-(cyclopropylmethyl)-18-(1-hydroxy-1-me...)Show SMILES [H][C@]12Cc3ccc(O)c4OC5[C@](CCN1CC1CC1)(c34)[C@@]21CCC5(OC)C(C1)C(C)(C)O |TLB:28:26:11.10:21.22,8:19:20:14.12.13,4:3:20:14.12.13| Show InChI InChI=1S/C26H35NO4/c1-23(2,29)18-13-24-8-9-26(18,30-3)22-25(24)10-11-27(14-15-4-5-15)19(24)12-16-6-7-17(28)21(31-22)20(16)25/h6-7,15,18-19,22,28-29H,4-5,8-14H2,1-3H3/t18?,19-,22?,24-,25+,26?/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 272: 423-8 (1995)
BindingDB Entry DOI: 10.7270/Q2TM78M9 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(RAT) | BDBM82465

(BIM 23023 | BIM-23023 | D-Phe-L-Cys(1)-L-Tyr-D-Trp...)Show SMILES CC[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@H](CSSC[C@H](NC1=O)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)NC(=O)[C@H](N)Cc1ccccc1 Show InChI InChI=1S/C49H65N11O10S2/c1-3-34-44(65)59-40(49(70)60-41(27(2)61)42(52)63)26-72-71-25-39(58-43(64)33(51)21-28-11-5-4-6-12-28)48(69)56-37(22-29-16-18-31(62)19-17-29)46(67)57-38(23-30-24-53-35-14-8-7-13-32(30)35)47(68)55-36(45(66)54-34)15-9-10-20-50/h4-8,11-14,16-19,24,27,33-34,36-41,53,61-62H,3,9-10,15,20-23,25-26,50-51H2,1-2H3,(H2,52,63)(H,54,66)(H,55,68)(H,56,69)(H,57,67)(H,58,64)(H,59,65)(H,60,70)/t27-,33-,34+,36+,37+,38-,39+,40+,41+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 44: 385-92 (1993)
BindingDB Entry DOI: 10.7270/Q2X065KZ |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(RAT) | BDBM84626

(BIM 23034)Show SMILES CC1NC(=O)C(CCCCN)NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(Cc2ccc(O)cc2)NC(=O)C(CSSCC(NC1=O)C(=O)NCc1cccc2ccccc12)NC(=O)C(N)Cc1ccccc1 Show InChI InChI=1S/C55H64N10O8S2/c1-33-49(67)64-47(51(69)59-29-37-16-11-15-36-14-5-6-17-40(36)37)31-74-75-32-48(65-50(68)42(57)26-34-12-3-2-4-13-34)55(73)62-45(27-35-21-23-39(66)24-22-35)53(71)63-46(28-38-30-58-43-19-8-7-18-41(38)43)54(72)61-44(52(70)60-33)20-9-10-25-56/h2-8,11-19,21-24,30,33,42,44-48,58,66H,9-10,20,25-29,31-32,56-57H2,1H3,(H,59,69)(H,60,70)(H,61,72)(H,62,73)(H,63,71)(H,64,67)(H,65,68) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 44: 385-92 (1993)
BindingDB Entry DOI: 10.7270/Q2X065KZ |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 4
(Homo sapiens (Human)) | BDBM50059090
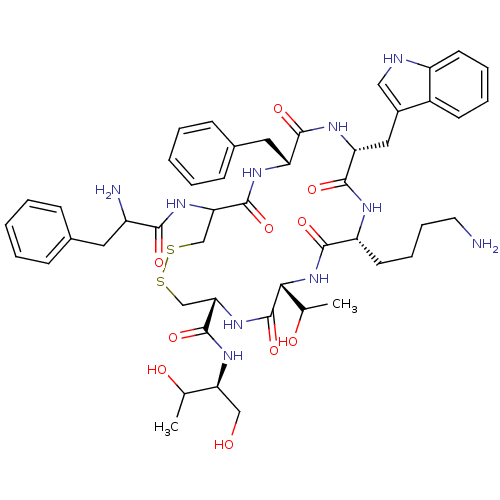
(10-(4-Amino-butyl)-19-(2-amino-3-phenyl-propionyla...)Show SMILES CC(O)[C@H](CO)NC(=O)[C@@H]1CSSCC(NC(=O)C(N)Cc2ccccc2)C(=O)N[C@H](Cc2ccccc2)C(=O)N[C@H](Cc2c[nH]c3ccccc23)C(=O)N[C@H](CCCCN)C(=O)N[C@H](C(C)O)C(=O)N1 Show InChI InChI=1S/C49H66N10O10S2/c1-28(61)39(25-60)56-48(68)41-27-71-70-26-40(57-43(63)34(51)21-30-13-5-3-6-14-30)47(67)54-37(22-31-15-7-4-8-16-31)45(65)55-38(23-32-24-52-35-18-10-9-17-33(32)35)46(66)53-36(19-11-12-20-50)44(64)59-42(29(2)62)49(69)58-41/h3-10,13-18,24,28-29,34,36-42,52,60-62H,11-12,19-23,25-27,50-51H2,1-2H3,(H,53,66)(H,54,67)(H,55,65)(H,56,68)(H,57,63)(H,58,69)(H,59,64)/t28?,29?,34?,36-,37-,38-,39+,40?,41+,42-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Endocr Rev 16: 427-42 (1995)
Article DOI: 10.1210/edrv-16-4-427
BindingDB Entry DOI: 10.7270/Q24B2ZTD |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 1
(Homo sapiens (Human)) | BDBM50097783

(CHEMBL438401 | D-Trp8 SST-14 | SOMATOSTATIN | SRIF...)Show SMILES C[C@@H](O)[C@@H]1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@@H](NC(=O)[C@@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)C(C)O Show InChI InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42?,43+,50-,51+,52-,53+,54+,55-,56+,57+,58-,59+,62-,63-/m0/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by PDSP Ki Database
| |
Mol Pharmacol 43: 838-44 (1993)
BindingDB Entry DOI: 10.7270/Q21N7ZN7 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data