 Found 236 hits with Last Name = 'bell' and Initial = 'mg'
Found 236 hits with Last Name = 'bell' and Initial = 'mg' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM50228078
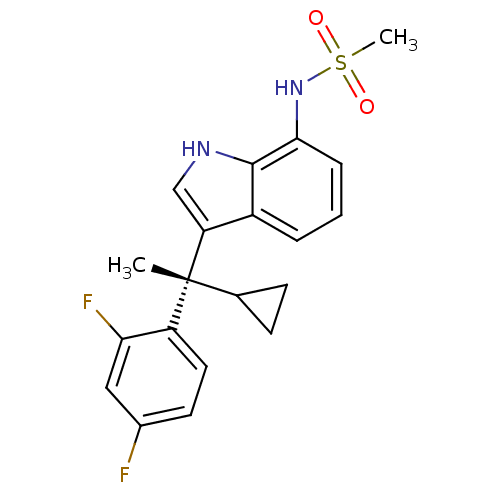
((S)-N-(3-(1-cyclopropyl-1-(2,4-difluorophenyl)ethy...)Show SMILES C[C@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1F Show InChI InChI=1S/C20H20F2N2O2S/c1-20(12-6-7-12,15-9-8-13(21)10-17(15)22)16-11-23-19-14(16)4-3-5-18(19)24-27(2,25)26/h3-5,8-12,23-24H,6-7H2,1-2H3/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.494 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human mineralocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM50228079
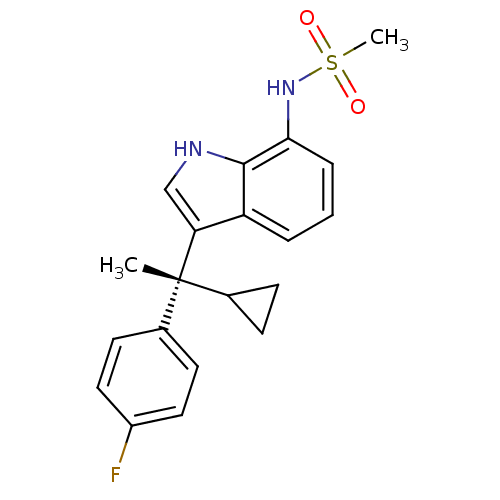
((R)-N-(3-(1-cyclopropyl-1-(4-fluorophenyl)ethyl)-1...)Show SMILES C[C@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1 Show InChI InChI=1S/C20H21FN2O2S/c1-20(13-6-7-13,14-8-10-15(21)11-9-14)17-12-22-19-16(17)4-3-5-18(19)23-26(2,24)25/h3-5,8-13,22-23H,6-7H2,1-2H3/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.56 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human mineralocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM50228076
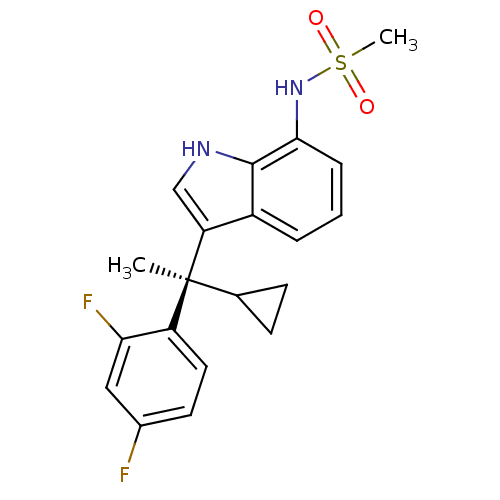
((R)-N-(3-(1-cyclopropyl-1-(2,4-difluorophenyl)ethy...)Show SMILES C[C@@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1F Show InChI InChI=1S/C20H20F2N2O2S/c1-20(12-6-7-12,15-9-8-13(21)10-17(15)22)16-11-23-19-14(16)4-3-5-18(19)24-27(2,25)26/h3-5,8-12,23-24H,6-7H2,1-2H3/t20-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human mineralocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM50228080
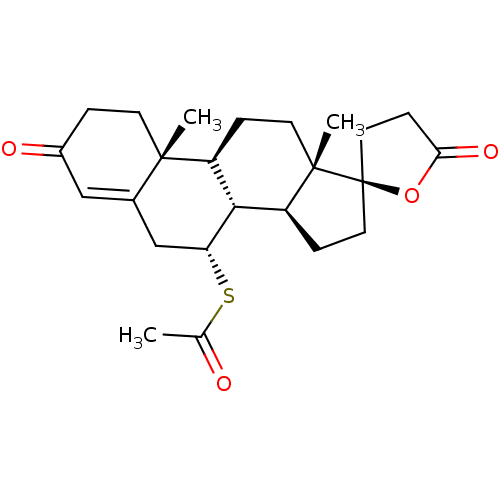
(2',15'-dimethyl-5,5'-dioxo-(9'R)-spiro[tetrahydrof...)Show SMILES CC(=O)S[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@H]2CC[C@@]3(C)[C@@H](CC[C@@]33CCC(=O)O3)[C@H]12 |r,t:6| Show InChI InChI=1S/C24H32O4S/c1-14(25)29-19-13-15-12-16(26)4-8-22(15,2)17-5-9-23(3)18(21(17)19)6-10-24(23)11-7-20(27)28-24/h12,17-19,21H,4-11,13H2,1-3H3/t17-,18-,19+,21+,22-,23-,24+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 2.32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human mineralocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50129720
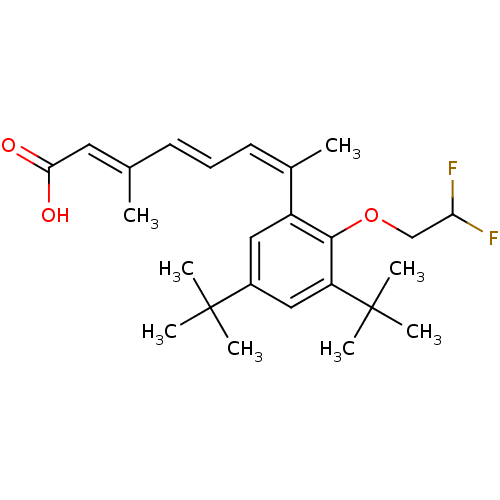
((2E,4E,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C25H34F2O3/c1-16(12-22(28)29)10-9-11-17(2)19-13-18(24(3,4)5)14-20(25(6,7)8)23(19)30-15-21(26)27/h9-14,21H,15H2,1-8H3,(H,28,29)/b10-9+,16-12+,17-11- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM50494356
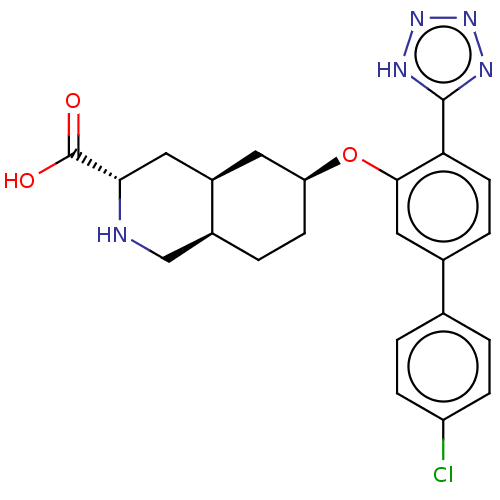
(CHEMBL3088070)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(ccc1-c1nnn[nH]1)-c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C23H24ClN5O3.ClH/c24-17-5-1-13(2-6-17)14-4-8-19(22-26-28-29-27-22)21(11-14)32-18-7-3-15-12-25-20(23(30)31)10-16(15)9-18;/h1-2,4-6,8,11,15-16,18,20,25H,3,7,9-10,12H2,(H,30,31)(H,26,27,28,29);1H/t15-,16+,18-,20-;/m0./s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]AMPA from homomeric recombinant GluA2 receptor (unknown origin) |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132579

((2E,4E,6Z)-7-(2-Butoxy-3,5-di-tert-butyl-phenyl)-3...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(/C)=C/C(O)=O Show InChI InChI=1S/C27H40O3/c1-10-11-15-30-25-22(20(3)14-12-13-19(2)16-24(28)29)17-21(26(4,5)6)18-23(25)27(7,8)9/h12-14,16-18H,10-11,15H2,1-9H3,(H,28,29)/b13-12+,19-16+,20-14- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132585
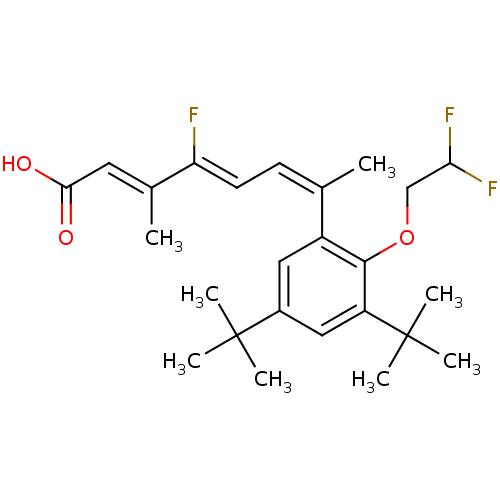
((2E,4Z,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(=C/C(O)=O)\C(\F)=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C Show InChI InChI=1S/C25H33F3O3/c1-15(9-10-20(26)16(2)11-22(29)30)18-12-17(24(3,4)5)13-19(25(6,7)8)23(18)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b15-9-,16-11+,20-10- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 4.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM50228081

((S)-N-(3-(1-cyclopropyl-1-(4-fluorophenyl)ethyl)-1...)Show SMILES C[C@@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1 Show InChI InChI=1S/C20H21FN2O2S/c1-20(13-6-7-13,14-8-10-15(21)11-9-14)17-12-22-19-16(17)4-3-5-18(19)23-26(2,24)25/h3-5,8-13,22-23H,6-7H2,1-2H3/t20-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 7.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human mineralocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132580
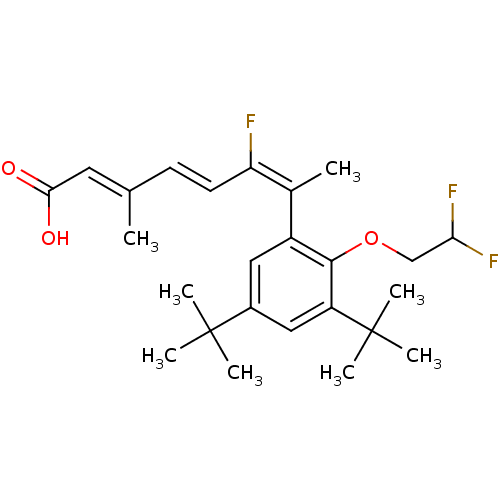
((2E,4E,6E)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C(\F)=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C25H33F3O3/c1-15(11-22(29)30)9-10-20(26)16(2)18-12-17(24(3,4)5)13-19(25(6,7)8)23(18)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b10-9+,15-11+,20-16+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 7.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132581
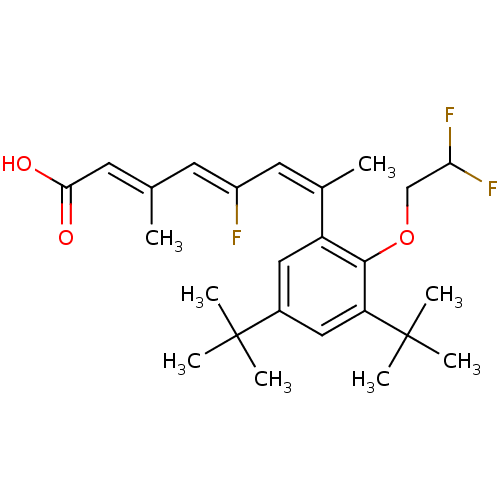
((2E,4Z,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(=C/C(O)=O)\C=C(/F)\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C Show InChI InChI=1S/C25H33F3O3/c1-15(10-22(29)30)9-18(26)11-16(2)19-12-17(24(3,4)5)13-20(25(6,7)8)23(19)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b15-10+,16-11-,18-9- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50228078

((S)-N-(3-(1-cyclopropyl-1-(2,4-difluorophenyl)ethy...)Show SMILES C[C@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1F Show InChI InChI=1S/C20H20F2N2O2S/c1-20(12-6-7-12,15-9-8-13(21)10-17(15)22)16-11-23-19-14(16)4-3-5-18(19)24-27(2,25)26/h3-5,8-12,23-24H,6-7H2,1-2H3/t20-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human glucocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50129728

((2E,4E,6Z)-7-(3,5-Di-tert-butyl-2-ethoxy-phenyl)-3...)Show SMILES CCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(/C)=C/C(O)=O Show InChI InChI=1S/C25H36O3/c1-10-28-23-20(18(3)13-11-12-17(2)14-22(26)27)15-19(24(4,5)6)16-21(23)25(7,8)9/h11-16H,10H2,1-9H3,(H,26,27)/b12-11+,17-14+,18-13- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132584
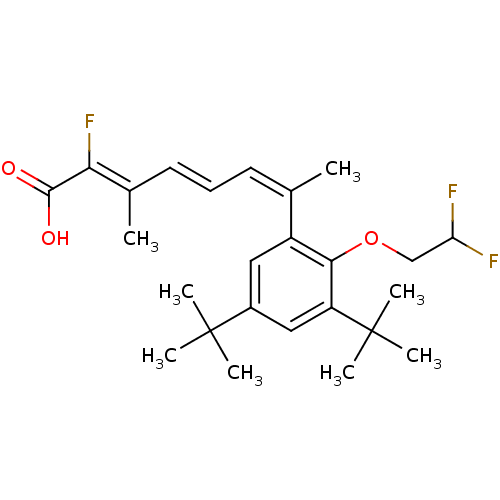
((2Z,4E,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C(\F)C(O)=O Show InChI InChI=1S/C25H33F3O3/c1-15(10-9-11-16(2)21(28)23(29)30)18-12-17(24(3,4)5)13-19(25(6,7)8)22(18)31-14-20(26)27/h9-13,20H,14H2,1-8H3,(H,29,30)/b11-9+,15-10-,21-16- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Antagonist activity against Retinoic acid receptor RXR-alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494356

(CHEMBL3088070)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(ccc1-c1nnn[nH]1)-c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C23H24ClN5O3.ClH/c24-17-5-1-13(2-6-17)14-4-8-19(22-26-28-29-27-22)21(11-14)32-18-7-3-15-12-25-20(23(30)31)10-16(15)9-18;/h1-2,4-6,8,11,15-16,18,20,25H,3,7,9-10,12H2,(H,30,31)(H,26,27,28,29);1H/t15-,16+,18-,20-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50228080

(2',15'-dimethyl-5,5'-dioxo-(9'R)-spiro[tetrahydrof...)Show SMILES CC(=O)S[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@H]2CC[C@@]3(C)[C@@H](CC[C@@]33CCC(=O)O3)[C@H]12 |r,t:6| Show InChI InChI=1S/C24H32O4S/c1-14(25)29-19-13-15-12-16(26)4-8-22(15,2)17-5-9-23(3)18(21(17)19)6-10-24(23)11-7-20(27)28-24/h12,17-19,21H,4-11,13H2,1-3H3/t17-,18-,19+,21+,22-,23-,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 32.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human glucocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50228080

(2',15'-dimethyl-5,5'-dioxo-(9'R)-spiro[tetrahydrof...)Show SMILES CC(=O)S[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@H]2CC[C@@]3(C)[C@@H](CC[C@@]33CCC(=O)O3)[C@H]12 |r,t:6| Show InChI InChI=1S/C24H32O4S/c1-14(25)29-19-13-15-12-16(26)4-8-22(15,2)17-5-9-23(3)18(21(17)19)6-10-24(23)11-7-20(27)28-24/h12,17-19,21H,4-11,13H2,1-3H3/t17-,18-,19+,21+,22-,23-,24+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 39.4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human androgen receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132583
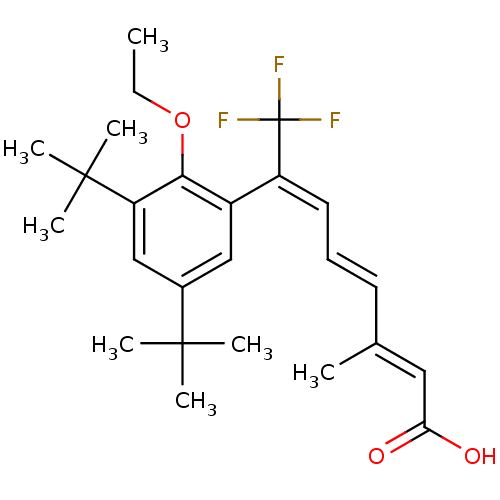
((2E,4E,6E)-7-(3,5-Di-tert-butyl-2-ethoxy-phenyl)-8...)Show SMILES CCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(=C/C=C/C(/C)=C/C(O)=O)\C(F)(F)F Show InChI InChI=1S/C25H33F3O3/c1-9-31-22-18(14-17(23(3,4)5)15-20(22)24(6,7)8)19(25(26,27)28)12-10-11-16(2)13-21(29)30/h10-15H,9H2,1-8H3,(H,29,30)/b11-10+,16-13+,19-12+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494342

(CHEMBL3088072)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(ccc1-c1nnn[nH]1)-c1cccs1 |r| Show InChI InChI=1S/C21H23N5O3S.ClH/c27-21(28)17-9-14-8-15(5-3-13(14)11-22-17)29-18-10-12(19-2-1-7-30-19)4-6-16(18)20-23-25-26-24-20;/h1-2,4,6-7,10,13-15,17,22H,3,5,8-9,11H2,(H,27,28)(H,23,24,25,26);1H/t13-,14+,15-,17-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494350
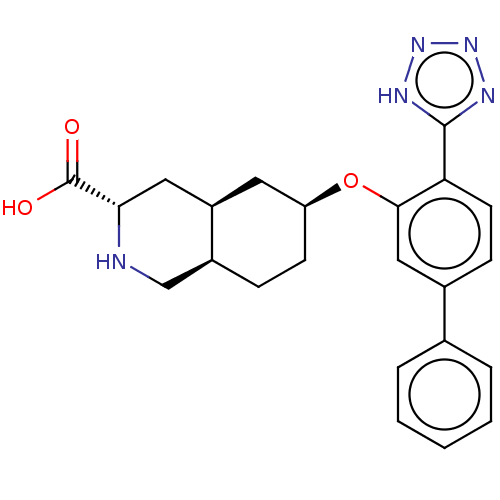
(CHEMBL3088064)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(ccc1-c1nnn[nH]1)-c1ccccc1 |r| Show InChI InChI=1S/C23H25N5O3.ClH/c29-23(30)20-11-17-10-18(8-6-16(17)13-24-20)31-21-12-15(14-4-2-1-3-5-14)7-9-19(21)22-25-27-28-26-22;/h1-5,7,9,12,16-18,20,24H,6,8,10-11,13H2,(H,29,30)(H,25,26,27,28);1H/t16-,17+,18-,20-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM50228077
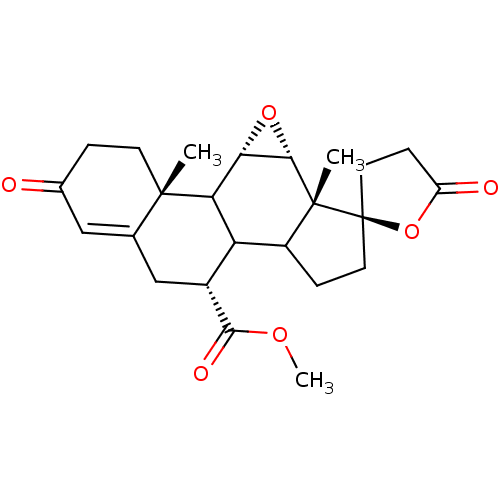
(CHEMBL237122 | epierenone)Show SMILES COC(=O)[C@@H]1CC2=CC(=O)CC[C@]2(C)C2[C@@H]3O[C@@H]3[C@@]3(C)C(CC[C@@]33CCC(=O)O3)C12 |t:6| Show InChI InChI=1S/C24H30O6/c1-22-7-4-13(25)10-12(22)11-14(21(27)28-3)17-15-5-8-24(9-6-16(26)30-24)23(15,2)20-19(29-20)18(17)22/h10,14-15,17-20H,4-9,11H2,1-3H3/t14-,15?,17?,18?,19+,20+,22+,23-,24-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 124 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human mineralocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Progesterone receptor
(Homo sapiens (Human)) | BDBM50228079

((R)-N-(3-(1-cyclopropyl-1-(4-fluorophenyl)ethyl)-1...)Show SMILES C[C@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1 Show InChI InChI=1S/C20H21FN2O2S/c1-20(13-6-7-13,14-8-10-15(21)11-9-14)17-12-22-19-16(17)4-3-5-18(19)23-26(2,24)25/h3-5,8-13,22-23H,6-7H2,1-2H3/t20-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 137 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human progesterone receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494365

(CHEMBL3085837)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(Cc2ccccc2)ccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C24H27N5O3.ClH/c30-24(31)21-13-18-12-19(8-7-17(18)14-25-21)32-22-11-16(10-15-4-2-1-3-5-15)6-9-20(22)23-26-28-29-27-23;/h1-6,9,11,17-19,21,25H,7-8,10,12-14H2,(H,30,31)(H,26,27,28,29);1H/t17-,18+,19-,21-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494355
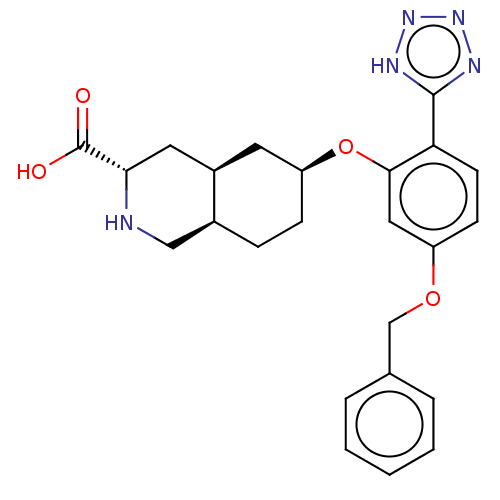
(CHEMBL3085836)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(OCc2ccccc2)ccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C24H27N5O4.ClH/c30-24(31)21-11-17-10-19(7-6-16(17)13-25-21)33-22-12-18(32-14-15-4-2-1-3-5-15)8-9-20(22)23-26-28-29-27-23;/h1-5,8-9,12,16-17,19,21,25H,6-7,10-11,13-14H2,(H,30,31)(H,26,27,28,29);1H/t16-,17+,19-,21-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Progesterone receptor
(Homo sapiens (Human)) | BDBM50228078

((S)-N-(3-(1-cyclopropyl-1-(2,4-difluorophenyl)ethy...)Show SMILES C[C@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1F Show InChI InChI=1S/C20H20F2N2O2S/c1-20(12-6-7-12,15-9-8-13(21)10-17(15)22)16-11-23-19-14(16)4-3-5-18(19)24-27(2,25)26/h3-5,8-12,23-24H,6-7H2,1-2H3/t20-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 163 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human progesterone receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50228079

((R)-N-(3-(1-cyclopropyl-1-(4-fluorophenyl)ethyl)-1...)Show SMILES C[C@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1 Show InChI InChI=1S/C20H21FN2O2S/c1-20(13-6-7-13,14-8-10-15(21)11-9-14)17-12-22-19-16(17)4-3-5-18(19)23-26(2,24)25/h3-5,8-13,22-23H,6-7H2,1-2H3/t20-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 167 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human glucocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494358

(CHEMBL3088058)Show SMILES [H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Nc1cccc(Cl)c1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H21ClN6O2/c18-12-2-1-3-13(15(12)16-21-23-24-22-16)20-11-5-4-9-8-19-14(17(25)26)7-10(9)6-11/h1-3,9-11,14,19-20H,4-8H2,(H,25,26)(H,21,22,23,24)/t9-,10+,11-,14-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494364

(CHEMBL3088054)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cccc(Cl)c1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H20ClN5O3.ClH/c18-12-2-1-3-14(15(12)16-20-22-23-21-16)26-11-5-4-9-8-19-13(17(24)25)7-10(9)6-11;/h1-3,9-11,13,19H,4-8H2,(H,24,25)(H,20,21,22,23);1H/t9-,10+,11-,13-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM50207594
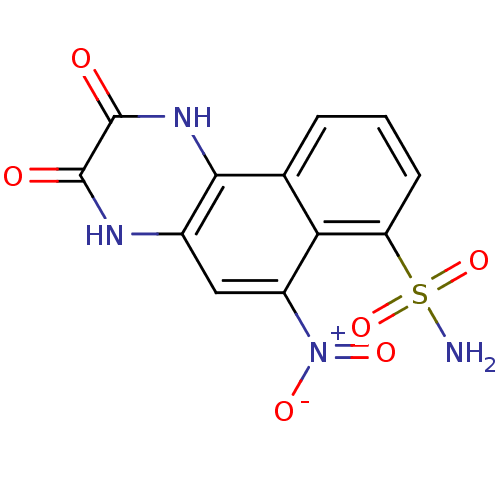
(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigación Lilly
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-AMPA binding to human GluR2 receptors expressed in HEK 293 cells |
J Med Chem 48: 4200-3 (2005)
Article DOI: 10.1021/jm0491952
BindingDB Entry DOI: 10.7270/Q2HQ3ZDM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM50207594

(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 2 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Progesterone receptor
(Homo sapiens (Human)) | BDBM50228081

((S)-N-(3-(1-cyclopropyl-1-(4-fluorophenyl)ethyl)-1...)Show SMILES C[C@@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1 Show InChI InChI=1S/C20H21FN2O2S/c1-20(13-6-7-13,14-8-10-15(21)11-9-14)17-12-22-19-16(17)4-3-5-18(19)23-26(2,24)25/h3-5,8-13,22-23H,6-7H2,1-2H3/t20-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 313 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human progesterone receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50228081

((S)-N-(3-(1-cyclopropyl-1-(4-fluorophenyl)ethyl)-1...)Show SMILES C[C@@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1 Show InChI InChI=1S/C20H21FN2O2S/c1-20(13-6-7-13,14-8-10-15(21)11-9-14)17-12-22-19-16(17)4-3-5-18(19)23-26(2,24)25/h3-5,8-13,22-23H,6-7H2,1-2H3/t20-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 387 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human glucocorticoid receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Progesterone receptor
(Homo sapiens (Human)) | BDBM50228080

(2',15'-dimethyl-5,5'-dioxo-(9'R)-spiro[tetrahydrof...)Show SMILES CC(=O)S[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@H]2CC[C@@]3(C)[C@@H](CC[C@@]33CCC(=O)O3)[C@H]12 |r,t:6| Show InChI InChI=1S/C24H32O4S/c1-14(25)29-19-13-15-12-16(26)4-8-22(15,2)17-5-9-23(3)18(21(17)19)6-10-24(23)11-7-20(27)28-24/h12,17-19,21H,4-11,13H2,1-3H3/t17-,18-,19+,21+,22-,23-,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human progesterone receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494351
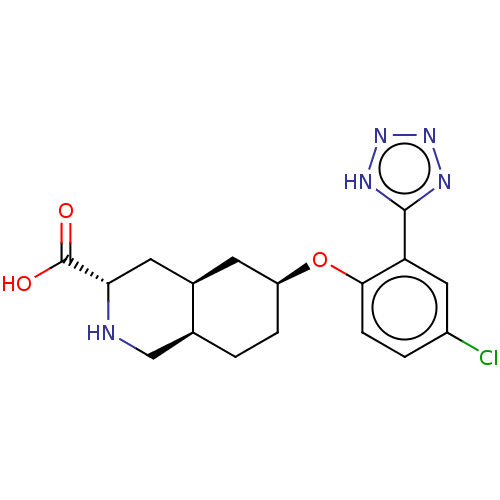
(CHEMBL3088060)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1ccc(Cl)cc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H20ClN5O3.ClH/c18-11-2-4-15(13(7-11)16-20-22-23-21-16)26-12-3-1-9-8-19-14(17(24)25)6-10(9)5-12;/h2,4,7,9-10,12,14,19H,1,3,5-6,8H2,(H,24,25)(H,20,21,22,23);1H/t9-,10+,12-,14-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM50494351

(CHEMBL3088060)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1ccc(Cl)cc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H20ClN5O3.ClH/c18-11-2-4-15(13(7-11)16-20-22-23-21-16)26-12-3-1-9-8-19-14(17(24)25)6-10(9)5-12;/h2,4,7,9-10,12,14,19H,1,3,5-6,8H2,(H,24,25)(H,20,21,22,23);1H/t9-,10+,12-,14-;/m0./s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]AMPA from homomeric recombinant GluA2 receptor (unknown origin) |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494055

(CHEMBL2440699)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(ccc1C(O)=O)-c1ccccc1 |r| Show InChI InChI=1S/C23H25NO5.ClH/c25-22(26)19-9-7-15(14-4-2-1-3-5-14)12-21(19)29-18-8-6-16-13-24-20(23(27)28)11-17(16)10-18;/h1-5,7,9,12,16-18,20,24H,6,8,10-11,13H2,(H,25,26)(H,27,28);1H/t16-,17+,18-,20-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Binding affinity to GluK1 receptor (unknown origin) |
Bioorg Med Chem Lett 23: 6459-62 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.046
BindingDB Entry DOI: 10.7270/Q2862KDB |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50228081

((S)-N-(3-(1-cyclopropyl-1-(4-fluorophenyl)ethyl)-1...)Show SMILES C[C@@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1 Show InChI InChI=1S/C20H21FN2O2S/c1-20(13-6-7-13,14-8-10-15(21)11-9-14)17-12-22-19-16(17)4-3-5-18(19)23-26(2,24)25/h3-5,8-13,22-23H,6-7H2,1-2H3/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human androgen receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494367

(CHEMBL3087685)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Sc1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H21N5O2S.ClH/c23-17(24)14-8-11-7-12(6-5-10(11)9-18-14)25-15-4-2-1-3-13(15)16-19-21-22-20-16;/h1-4,10-12,14,18H,5-9H2,(H,23,24)(H,19,20,21,22);1H/t10-,11+,12-,14-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor 4
(Homo sapiens (Human)) | BDBM50207594

(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 4 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494348
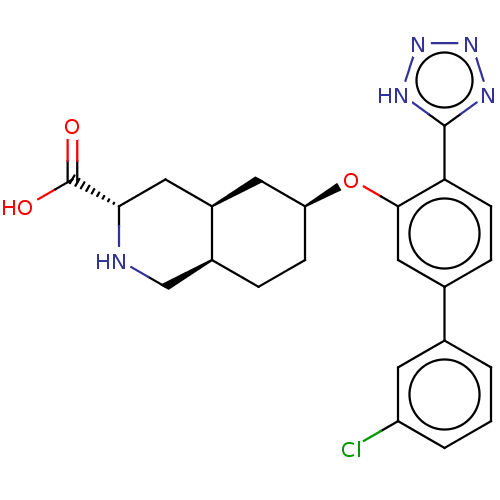
(CHEMBL3088069)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(ccc1-c1nnn[nH]1)-c1cccc(Cl)c1 |r| Show InChI InChI=1S/C23H24ClN5O3.ClH/c24-17-3-1-2-13(8-17)14-5-7-19(22-26-28-29-27-22)21(11-14)32-18-6-4-15-12-25-20(23(30)31)10-16(15)9-18;/h1-3,5,7-8,11,15-16,18,20,25H,4,6,9-10,12H2,(H,30,31)(H,26,27,28,29);1H/t15-,16+,18-,20-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50228078

((S)-N-(3-(1-cyclopropyl-1-(2,4-difluorophenyl)ethy...)Show SMILES C[C@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1F Show InChI InChI=1S/C20H20F2N2O2S/c1-20(12-6-7-12,15-9-8-13(21)10-17(15)22)16-11-23-19-14(16)4-3-5-18(19)24-27(2,25)26/h3-5,8-12,23-24H,6-7H2,1-2H3/t20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human androgen receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494068
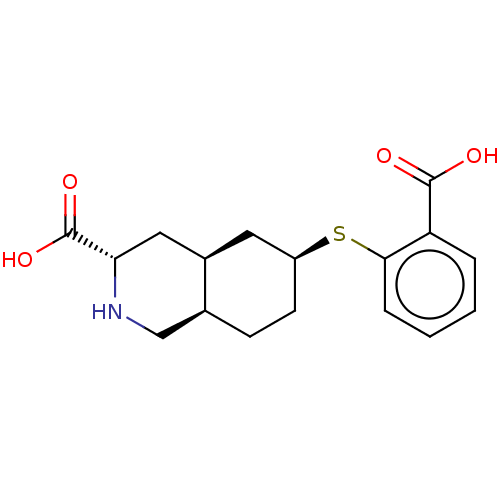
(CHEMBL2440688)Show SMILES [H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Sc1ccccc1C(O)=O |r| Show InChI InChI=1S/C17H21NO4S/c19-16(20)13-3-1-2-4-15(13)23-12-6-5-10-9-18-14(17(21)22)8-11(10)7-12/h1-4,10-12,14,18H,5-9H2,(H,19,20)(H,21,22)/t10-,11+,12-,14-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Binding affinity to GluK1 receptor (unknown origin) |
Bioorg Med Chem Lett 23: 6459-62 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.046
BindingDB Entry DOI: 10.7270/Q2862KDB |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50168964

((3S,4aR,6S,8aR)-6-[2-(1H-Tetrazol-5-yl)-phenylamin...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@H](CC[C@H]2CN1)Nc1ccccc1-c1nnn[nH]1 Show InChI InChI=1S/C17H22N6O2/c24-17(25)15-8-11-7-12(6-5-10(11)9-18-15)19-14-4-2-1-3-13(14)16-20-22-23-21-16/h1-4,10-12,15,18-19H,5-9H2,(H,24,25)(H,20,21,22,23)/t10-,11+,12-,15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50228079

((R)-N-(3-(1-cyclopropyl-1-(4-fluorophenyl)ethyl)-1...)Show SMILES C[C@](C1CC1)(c1c[nH]c2c(NS(C)(=O)=O)cccc12)c1ccc(F)cc1 Show InChI InChI=1S/C20H21FN2O2S/c1-20(13-6-7-13,14-8-10-15(21)11-9-14)17-12-22-19-16(17)4-3-5-18(19)23-26(2,24)25/h3-5,8-13,22-23H,6-7H2,1-2H3/t20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 849 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human androgen receptor |
J Med Chem 50: 6443-5 (2007)
Article DOI: 10.1021/jm701186z
BindingDB Entry DOI: 10.7270/Q23B5ZVP |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494352
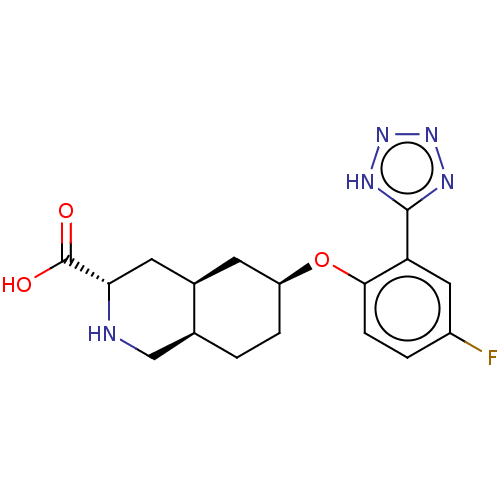
(CHEMBL3088059)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1ccc(F)cc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H20FN5O3.ClH/c18-11-2-4-15(13(7-11)16-20-22-23-21-16)26-12-3-1-9-8-19-14(17(24)25)6-10(9)5-12;/h2,4,7,9-10,12,14,19H,1,3,5-6,8H2,(H,24,25)(H,20,21,22,23);1H/t9-,10+,12-,14-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494346
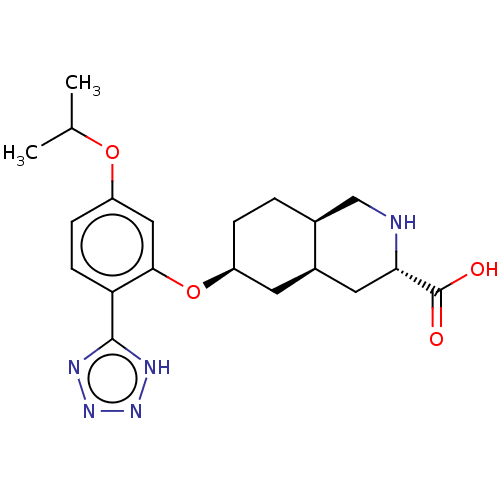
(CHEMBL3088074)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(OC(C)C)ccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C20H27N5O4.ClH/c1-11(2)28-15-5-6-16(19-22-24-25-23-19)18(9-15)29-14-4-3-12-10-21-17(20(26)27)8-13(12)7-14;/h5-6,9,11-14,17,21H,3-4,7-8,10H2,1-2H3,(H,26,27)(H,22,23,24,25);1H/t12-,13+,14-,17-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor 3
(Homo sapiens (Human)) | BDBM50207594

(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 3 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494061
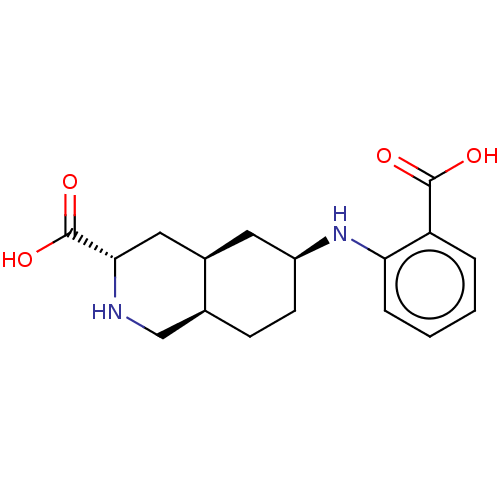
(CHEMBL2440689)Show SMILES [H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Nc1ccccc1C(O)=O |r| Show InChI InChI=1S/C17H22N2O4/c20-16(21)13-3-1-2-4-14(13)19-12-6-5-10-9-18-15(17(22)23)8-11(10)7-12/h1-4,10-12,15,18-19H,5-9H2,(H,20,21)(H,22,23)/t10-,11+,12-,15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Binding affinity to GluK1 receptor (unknown origin) |
Bioorg Med Chem Lett 23: 6459-62 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.046
BindingDB Entry DOI: 10.7270/Q2862KDB |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494347
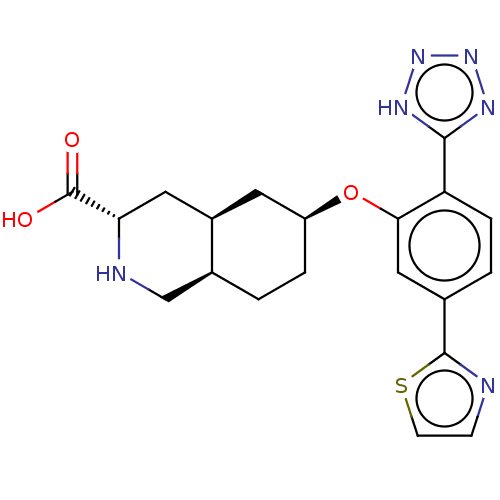
(CHEMBL3088071)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(ccc1-c1nnn[nH]1)-c1nccs1 |r| Show InChI InChI=1S/C20H22N6O3S.ClH/c27-20(28)16-8-13-7-14(3-1-12(13)10-22-16)29-17-9-11(19-21-5-6-30-19)2-4-15(17)18-23-25-26-24-18;/h2,4-6,9,12-14,16,22H,1,3,7-8,10H2,(H,27,28)(H,23,24,25,26);1H/t12-,13+,14-,16-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50494366

(CHEMBL3088068)Show SMILES Cl.[H][C@@]12CC[C@@H](C[C@]1([H])C[C@H](NC2)C(O)=O)Oc1cc(ccc1-c1nnn[nH]1)-c1ccccc1Cl |r| Show InChI InChI=1S/C23H24ClN5O3.ClH/c24-19-4-2-1-3-17(19)13-6-8-18(22-26-28-29-27-22)21(11-13)32-16-7-5-14-12-25-20(23(30)31)10-15(14)9-16;/h1-4,6,8,11,14-16,20,25H,5,7,9-10,12H2,(H,30,31)(H,26,27,28,29);1H/t14-,15+,16-,20-;/m0./s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Centro de Investigaci�n Lilly
Curated by ChEMBL
| Assay Description
Displacement of [3H]ATPA from human Gluk1 receptor |
Bioorg Med Chem Lett 23: 6463-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.045
BindingDB Entry DOI: 10.7270/Q2XS5ZBW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data