

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Muscarinic acetylcholine receptor M4 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.00780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M4 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.00980 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M1 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0215 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M5 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0266 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M5 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355612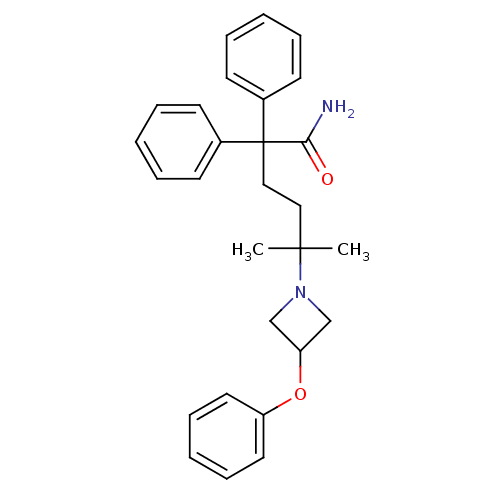 (CHEMBL1910848) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50355612 (CHEMBL1910848) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 2 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 2 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0750 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 2 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM528106 (N-{[5-methoxy-2-(1,2,3,4-tetrazol-1-yl)phenyl]meth...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355612 (CHEMBL1910848) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 2 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M4 (Homo sapiens (Human)) | BDBM50355622 (CHEMBL1910856) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.115 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M4 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50355622 (CHEMBL1910856) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.115 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M1 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355622 (CHEMBL1910856) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.128 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50355622 (CHEMBL1910856) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.137 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 2 (Homo sapiens (Human)) | BDBM50429823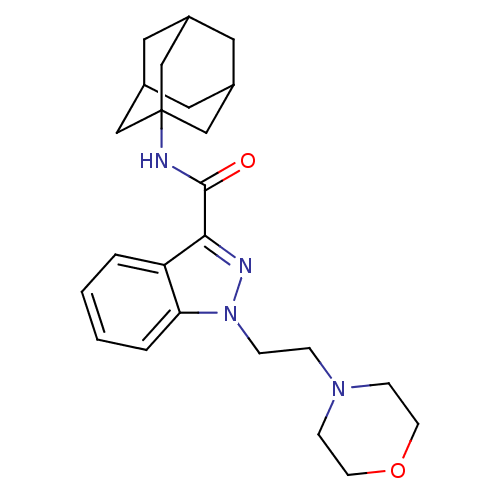 (CHEMBL2338173) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd Curated by ChEMBL | Assay Description Binding affinity to CB2 receptor (unknown origin) | Bioorg Med Chem Lett 23: 1177-81 (2013) Article DOI: 10.1016/j.bmcl.2013.01.044 BindingDB Entry DOI: 10.7270/Q2M32X3H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM528123 (N-{[5-chloro-2-(1,2,3,4-tetrazol-1-yl)phenyl]methy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355618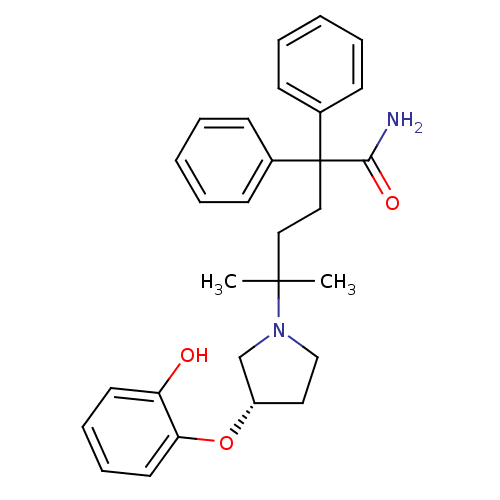 (CHEMBL1910852) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355631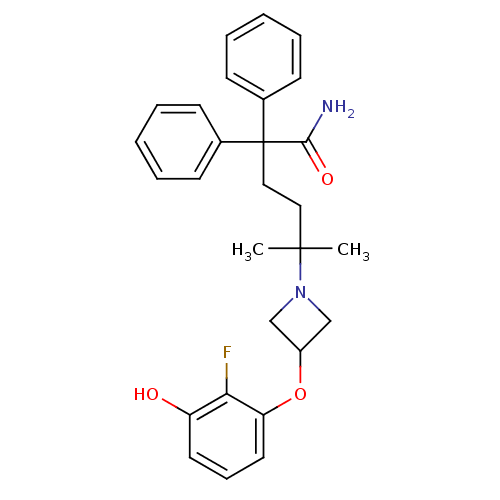 (CHEMBL1910865) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.163 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M5 (Homo sapiens (Human)) | BDBM50355622 (CHEMBL1910856) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.169 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M5 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM455297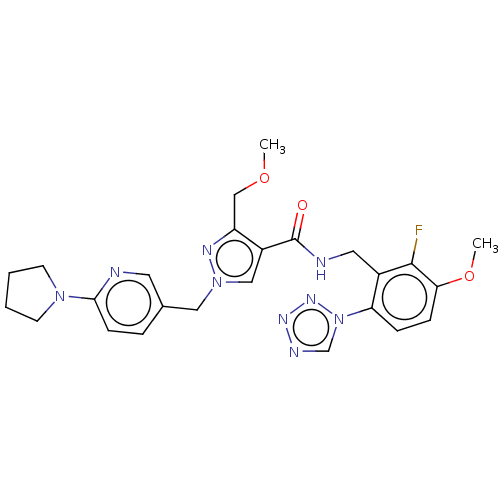 (US10730874, Compound TABLE II.2 | US11180484, Exam...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355617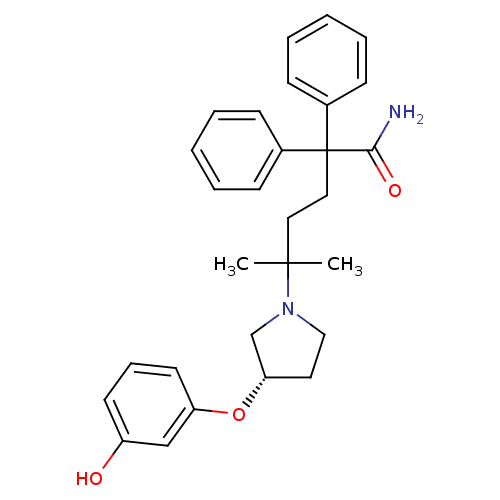 (CHEMBL1910851) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355623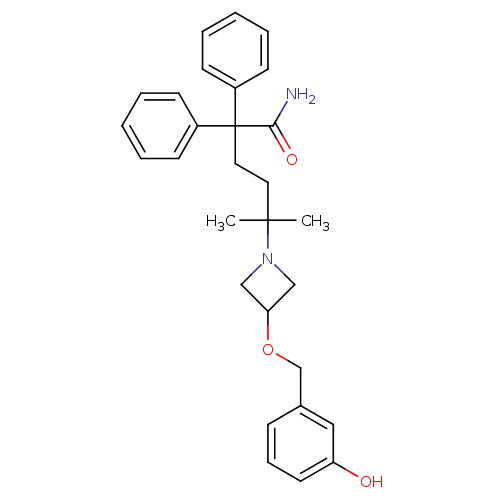 (CHEMBL1910857) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355610 (CHEMBL1237108) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355622 (CHEMBL1910856) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355616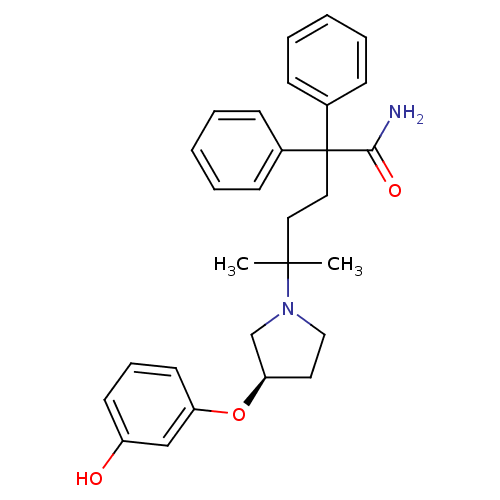 (CHEMBL1910850) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.201 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355617 (CHEMBL1910851) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355610 (CHEMBL1237108) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.213 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355628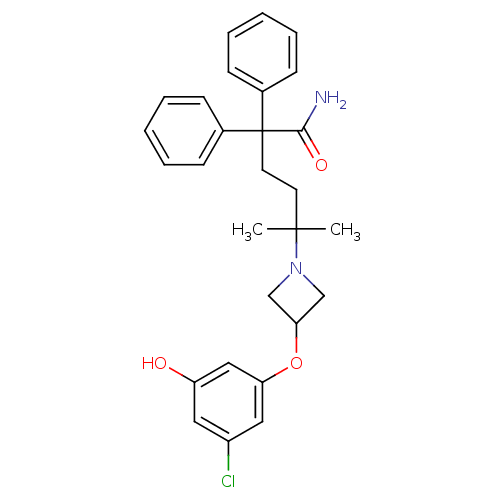 (CHEMBL1910862) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM528127 (US11180484, Example 79 | US11180484, Reference Exa...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355614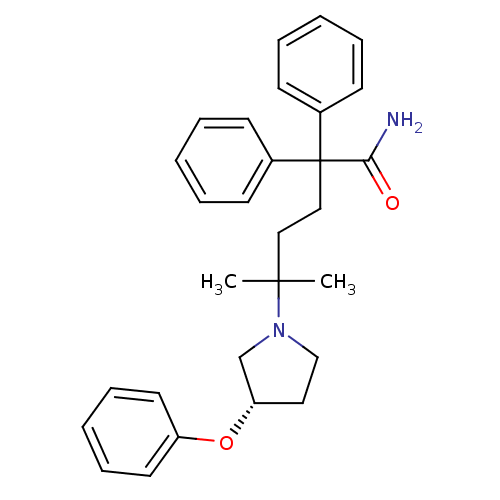 (CHEMBL1910846) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM528105 (N-{[2-fluoro-3-methoxy-6-(1,2,3,4-tetrazol-1-yl)ph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355631 (CHEMBL1910865) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355611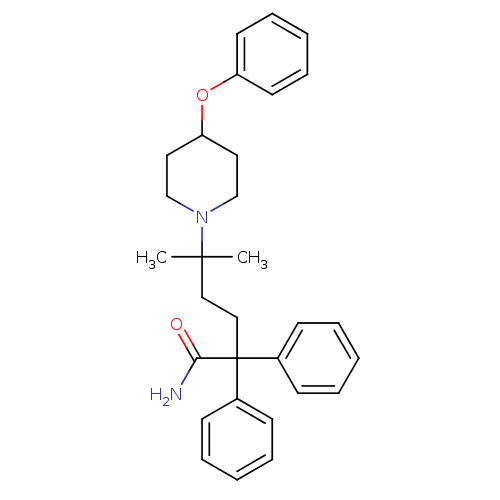 (CHEMBL1910849) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM528131 (1-{[2-(3,3-Difluoropyrrolidin-1-yl)pyrimidin-5-yl]...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355610 (CHEMBL1237108) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 2 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM528119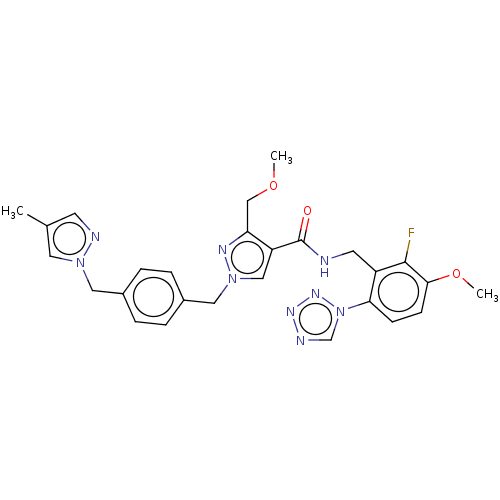 (N-{[2-fluoro-3-methoxy-6-(1,2,3,4-tetrazol-1-yl)ph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Caspase-1 (Homo sapiens (Human)) | BDBM50068972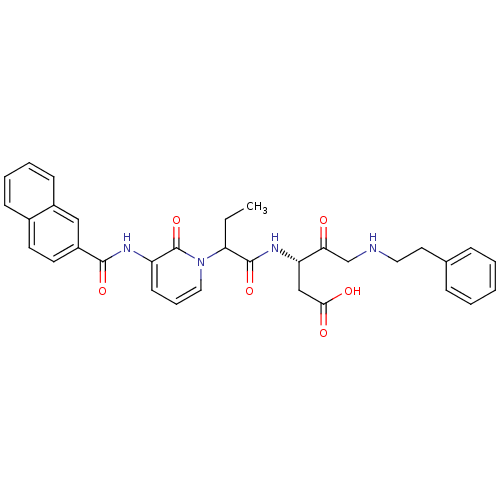 ((S)-3-(2-{3-[(Naphthalene-2-carbonyl)-amino]-2-oxo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ferring Research Institute Curated by ChEMBL | Assay Description Inhibition of human recombinant IL-1 beta converting enzyme | Bioorg Med Chem Lett 8: 959-64 (1999) BindingDB Entry DOI: 10.7270/Q2ZP458C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM528127 (US11180484, Example 79 | US11180484, Reference Exa...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M4 (Homo sapiens (Human)) | BDBM50355610 (CHEMBL1237108) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.382 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M4 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50355610 (CHEMBL1237108) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.413 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50355610 (CHEMBL1237108) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.433 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M1 receptor expressed in CHO cells after 24 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50355610 (CHEMBL1237108) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 2 hrs by filter binding assay | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50077223 (CHEMBL3416881) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]-vasopressin from human vasopressin 1a receptor expressed in HEK293 cell membranes after 1 hr by scintillation proximity assay | J Med Chem 58: 2275-89 (2015) Article DOI: 10.1021/jm501745f BindingDB Entry DOI: 10.7270/Q2JW8GK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355616 (CHEMBL1910850) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM528074 (N-{[2-Fluoro-6-(1,2,3,4-tetrazol-1-yl)phenyl]methy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasma kallikrein (Homo sapiens (Human)) | BDBM528130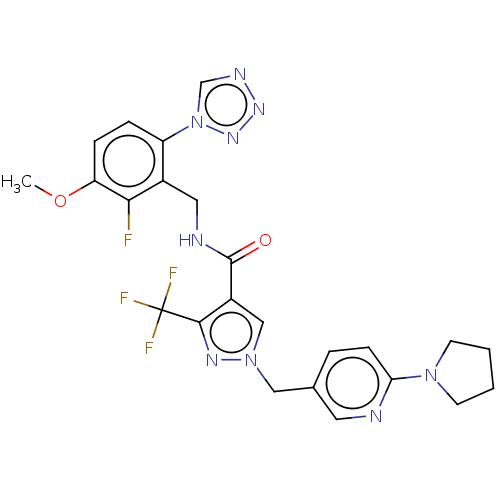 (N-{[2-fluoro-3-methoxy-6-(1,2,3,4-tetrazol-1-yl)ph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Plasma kallikrein inhibitory activity in vitro was determined using standard published methods (e.g. Johansen et al., Int. J. Tiss. Reac. 1986, 8, 18... | Citation and Details BindingDB Entry DOI: 10.7270/Q2MG7SP5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355626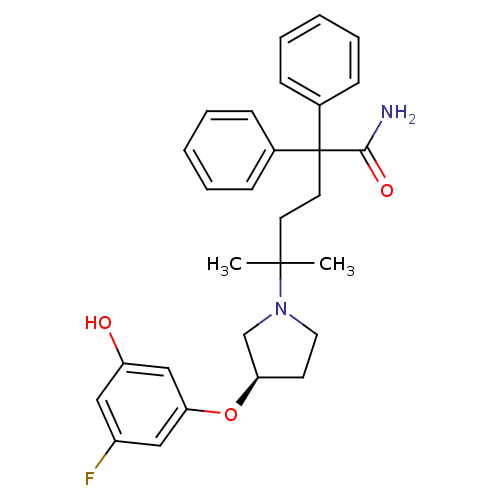 (CHEMBL1910860) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50355629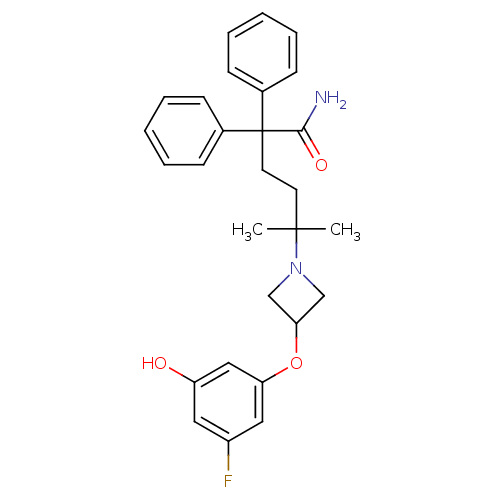 (CHEMBL1910863) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... | J Med Chem 54: 6888-904 (2011) Article DOI: 10.1021/jm200884j BindingDB Entry DOI: 10.7270/Q2CZ37JW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 8480 total ) | Next | Last >> |