 Found 628 hits with Last Name = 'lingham' and Initial = 'rb'
Found 628 hits with Last Name = 'lingham' and Initial = 'rb' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50287709

((E)-3-Carboxy-2-(16-sulfooxy-hexadecyl)-pent-2-ene...)Show SMILES OC(=O)C\C(C(O)=O)=C(\CCCCCCCCCCCCCCCCOS(O)(=O)=O)C(O)=O Show InChI InChI=1S/C22H38O10S/c23-20(24)17-19(22(27)28)18(21(25)26)15-13-11-9-7-5-3-1-2-4-6-8-10-12-14-16-32-33(29,30)31/h1-17H2,(H,23,24)(H,25,26)(H,27,28)(H,29,30,31)/b19-18+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
| Article
| 4.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibition of human recombinant Protein farnesyltransferase with respect to FPP |
Bioorg Med Chem Lett 6: 2081-2084 (1996)
Article DOI: 10.1016/0960-894X(96)00372-1
BindingDB Entry DOI: 10.7270/Q2222TQ1 |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50423687
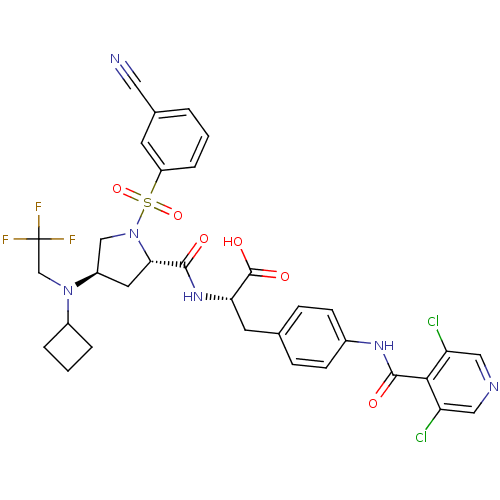
(CHEMBL569442)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N(CC(F)(F)F)C1CCC1 |r| Show InChI InChI=1S/C33H31Cl2F3N6O6S/c34-25-15-40-16-26(35)29(25)31(46)41-21-9-7-19(8-10-21)12-27(32(47)48)42-30(45)28-13-23(43(18-33(36,37)38)22-4-2-5-22)17-44(28)51(49,50)24-6-1-3-20(11-24)14-39/h1,3,6-11,15-16,22-23,27-28H,2,4-5,12-13,17-18H2,(H,41,46)(H,42,45)(H,47,48)/t23-,27+,28+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [35S]-labeled ligand from VLA4 in human Jurkat cells washed with buffer containing non activating Ca2+ and Mg2+ by competitive bindin... |
Bioorg Med Chem Lett 19: 5803-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.111
BindingDB Entry DOI: 10.7270/Q2VQ33ZJ |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50128319
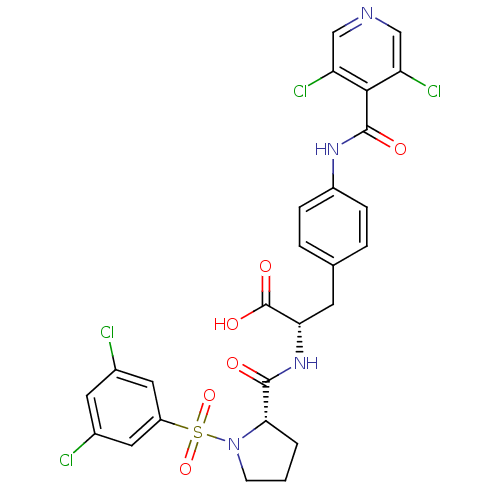
((S)-2-{[(S)-1-(3,5-Dichloro-benzenesulfonyl)-pyrro...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1CCCN1S(=O)(=O)c1cc(Cl)cc(Cl)c1 |r| Show InChI InChI=1S/C26H22Cl4N4O6S/c27-15-9-16(28)11-18(10-15)41(39,40)34-7-1-2-22(34)24(35)33-21(26(37)38)8-14-3-5-17(6-4-14)32-25(36)23-19(29)12-31-13-20(23)30/h3-6,9-13,21-22H,1-2,7-8H2,(H,32,36)(H,33,35)(H,37,38)/t21-,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of VLA-4 receptor of Jurkat cells in BURJ assay |
Bioorg Med Chem Lett 15: 4053-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.06.034
BindingDB Entry DOI: 10.7270/Q2KK9D3G |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50423695
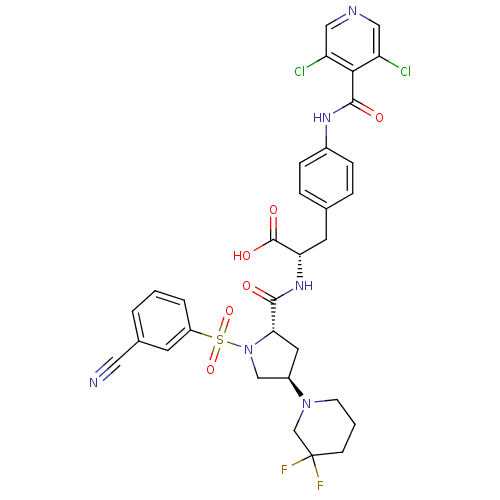
(CHEMBL566154 | MK-0617)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCCC(F)(F)C1 |r| Show InChI InChI=1S/C32H30Cl2F2N6O6S/c33-24-15-38-16-25(34)28(24)30(44)39-21-7-5-19(6-8-21)12-26(31(45)46)40-29(43)27-13-22(41-10-2-9-32(35,36)18-41)17-42(27)49(47,48)23-4-1-3-20(11-23)14-37/h1,3-8,11,15-16,22,26-27H,2,9-10,12-13,17-18H2,(H,39,44)(H,40,43)(H,45,46)/t22-,26+,27+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [35S]-labeled ligand from VLA4 in human Jurkat cells washed with buffer containing non activating Ca2+ and Mg2+ by competitive bindin... |
Bioorg Med Chem Lett 19: 5803-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.111
BindingDB Entry DOI: 10.7270/Q2VQ33ZJ |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50423695

(CHEMBL566154 | MK-0617)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCCC(F)(F)C1 |r| Show InChI InChI=1S/C32H30Cl2F2N6O6S/c33-24-15-38-16-25(34)28(24)30(44)39-21-7-5-19(6-8-21)12-26(31(45)46)40-29(43)27-13-22(41-10-2-9-32(35,36)18-41)17-42(27)49(47,48)23-4-1-3-20(11-23)14-37/h1,3-8,11,15-16,22,26-27H,2,9-10,12-13,17-18H2,(H,39,44)(H,40,43)(H,45,46)/t22-,26+,27+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [35S]-labeled ligand from VLA4 in human Jurkat cells washed with buffer containing activating Mn2+ by competitive binding assay |
Bioorg Med Chem Lett 19: 5803-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.111
BindingDB Entry DOI: 10.7270/Q2VQ33ZJ |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50423695

(CHEMBL566154 | MK-0617)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCCC(F)(F)C1 |r| Show InChI InChI=1S/C32H30Cl2F2N6O6S/c33-24-15-38-16-25(34)28(24)30(44)39-21-7-5-19(6-8-21)12-26(31(45)46)40-29(43)27-13-22(41-10-2-9-32(35,36)18-41)17-42(27)49(47,48)23-4-1-3-20(11-23)14-37/h1,3-8,11,15-16,22,26-27H,2,9-10,12-13,17-18H2,(H,39,44)(H,40,43)(H,45,46)/t22-,26+,27+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of VLA4 in human whole blood |
Bioorg Med Chem Lett 19: 5803-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.111
BindingDB Entry DOI: 10.7270/Q2VQ33ZJ |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309081
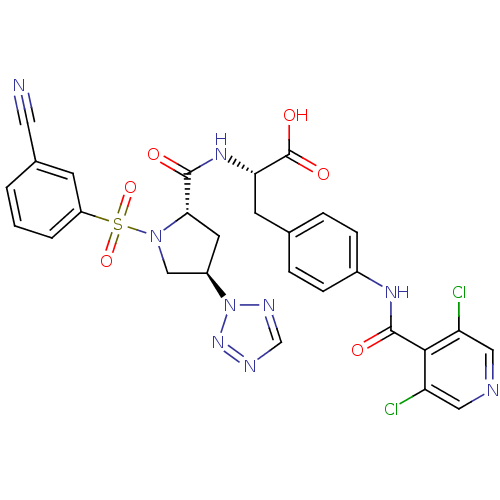
((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-(2H-tet...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)n1ncnn1 |r| Show InChI InChI=1S/C28H23Cl2N9O6S/c29-21-12-32-13-22(30)25(21)27(41)35-18-6-4-16(5-7-18)9-23(28(42)43)36-26(40)24-10-19(39-34-15-33-37-39)14-38(24)46(44,45)20-3-1-2-17(8-20)11-31/h1-8,12-13,15,19,23-24H,9-10,14H2,(H,35,41)(H,36,40)(H,42,43)/t19-,23+,24+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309084

((S)-2-((2S,4R)-4-(1H-benzo[d]imidazol-1-yl)-1-(3-c...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)n1cnc2ccccc12 |r| Show InChI InChI=1S/C34H27Cl2N7O6S/c35-25-16-38-17-26(36)31(25)33(45)40-22-10-8-20(9-11-22)13-28(34(46)47)41-32(44)30-14-23(42-19-39-27-6-1-2-7-29(27)42)18-43(30)50(48,49)24-5-3-4-21(12-24)15-37/h1-12,16-17,19,23,28,30H,13-14,18H2,(H,40,45)(H,41,44)(H,46,47)/t23-,28+,30+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50423692
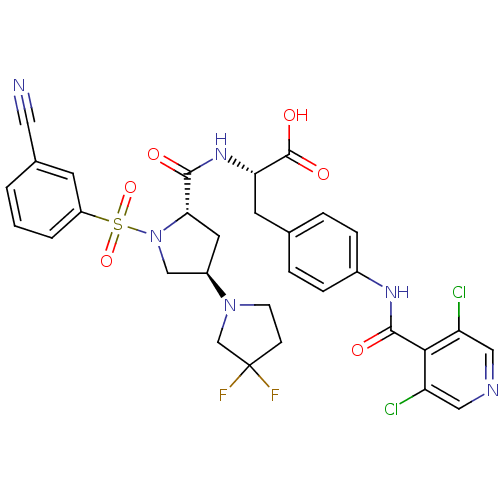
(CHEMBL569443)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCC(F)(F)C1 |r| Show InChI InChI=1S/C31H28Cl2F2N6O6S/c32-23-14-37-15-24(33)27(23)29(43)38-20-6-4-18(5-7-20)11-25(30(44)45)39-28(42)26-12-21(40-9-8-31(34,35)17-40)16-41(26)48(46,47)22-3-1-2-19(10-22)13-36/h1-7,10,14-15,21,25-26H,8-9,11-12,16-17H2,(H,38,43)(H,39,42)(H,44,45)/t21-,25+,26+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [35S]-labeled ligand from VLA4 in human Jurkat cells washed with buffer containing non activating Ca2+ and Mg2+ by competitive bindin... |
Bioorg Med Chem Lett 19: 5803-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.111
BindingDB Entry DOI: 10.7270/Q2VQ33ZJ |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309087
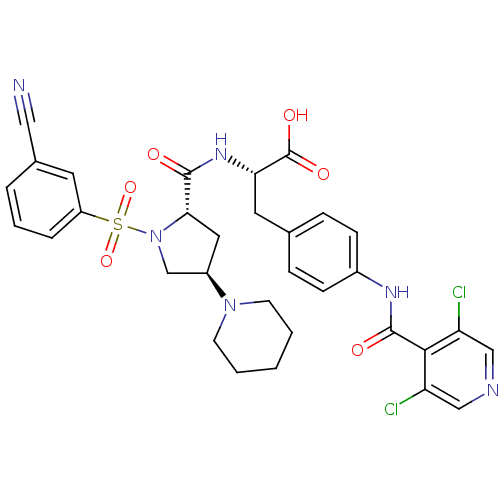
((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-(piperi...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCCCC1 |r| Show InChI InChI=1S/C32H32Cl2N6O6S/c33-25-17-36-18-26(34)29(25)31(42)37-22-9-7-20(8-10-22)14-27(32(43)44)38-30(41)28-15-23(39-11-2-1-3-12-39)19-40(28)47(45,46)24-6-4-5-21(13-24)16-35/h4-10,13,17-18,23,27-28H,1-3,11-12,14-15,19H2,(H,37,42)(H,38,41)(H,43,44)/t23-,27+,28+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309087

((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-(piperi...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCCCC1 |r| Show InChI InChI=1S/C32H32Cl2N6O6S/c33-25-17-36-18-26(34)29(25)31(42)37-22-9-7-20(8-10-22)14-27(32(43)44)38-30(41)28-15-23(39-11-2-1-3-12-39)19-40(28)47(45,46)24-6-4-5-21(13-24)16-35/h4-10,13,17-18,23,27-28H,1-3,11-12,14-15,19H2,(H,37,42)(H,38,41)(H,43,44)/t23-,27+,28+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 in presence of 90% human plasma |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50309087

((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-(piperi...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCCCC1 |r| Show InChI InChI=1S/C32H32Cl2N6O6S/c33-25-17-36-18-26(34)29(25)31(42)37-22-9-7-20(8-10-22)14-27(32(43)44)38-30(41)28-15-23(39-11-2-1-3-12-39)19-40(28)47(45,46)24-6-4-5-21(13-24)16-35/h4-10,13,17-18,23,27-28H,1-3,11-12,14-15,19H2,(H,37,42)(H,38,41)(H,43,44)/t23-,27+,28+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [35S]-labeled ligand from VLA4 in human Jurkat cells washed with buffer containing non activating Ca2+ and Mg2+ by competitive bindin... |
Bioorg Med Chem Lett 19: 5803-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.111
BindingDB Entry DOI: 10.7270/Q2VQ33ZJ |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309092
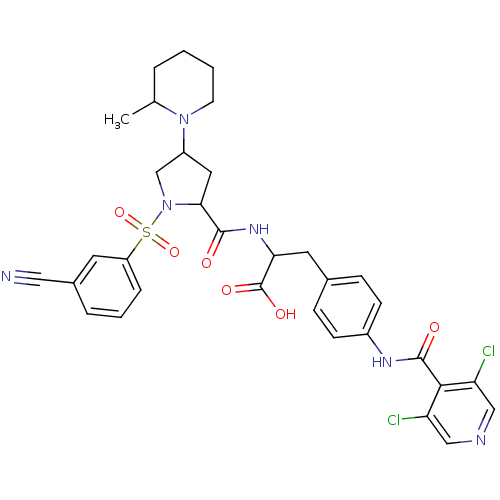
(2-(1-(1-(3-cyanophenylsulfonyl)-4-(2-methylpiperid...)Show SMILES CC1CCCCN1C1CC(N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)NC(Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O Show InChI InChI=1S/C33H34Cl2N6O6S/c1-20-5-2-3-12-40(20)24-15-29(41(19-24)48(46,47)25-7-4-6-22(13-25)16-36)31(42)39-28(33(44)45)14-21-8-10-23(11-9-21)38-32(43)30-26(34)17-37-18-27(30)35/h4,6-11,13,17-18,20,24,28-29H,2-3,5,12,14-15,19H2,1H3,(H,38,43)(H,39,42)(H,44,45) | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309092

(2-(1-(1-(3-cyanophenylsulfonyl)-4-(2-methylpiperid...)Show SMILES CC1CCCCN1C1CC(N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)NC(Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O Show InChI InChI=1S/C33H34Cl2N6O6S/c1-20-5-2-3-12-40(20)24-15-29(41(19-24)48(46,47)25-7-4-6-22(13-25)16-36)31(42)39-28(33(44)45)14-21-8-10-23(11-9-21)38-32(43)30-26(34)17-37-18-27(30)35/h4,6-11,13,17-18,20,24,28-29H,2-3,5,12,14-15,19H2,1H3,(H,38,43)(H,39,42)(H,44,45) | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 in presence of 90% human plasma |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50423690
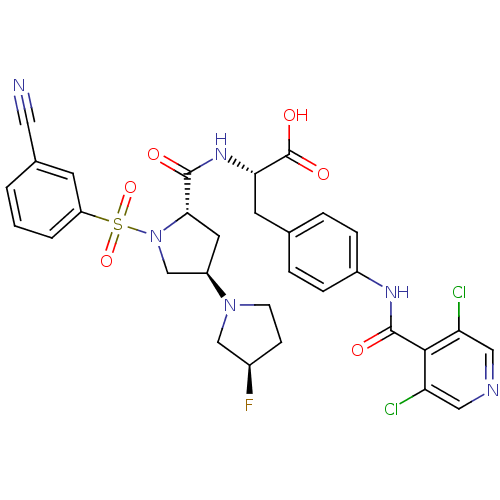
(CHEMBL569276)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CC[C@@H](F)C1 |r| Show InChI InChI=1S/C31H29Cl2FN6O6S/c32-24-14-36-15-25(33)28(24)30(42)37-21-6-4-18(5-7-21)11-26(31(43)44)38-29(41)27-12-22(39-9-8-20(34)16-39)17-40(27)47(45,46)23-3-1-2-19(10-23)13-35/h1-7,10,14-15,20,22,26-27H,8-9,11-12,16-17H2,(H,37,42)(H,38,41)(H,43,44)/t20-,22-,26+,27+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [35S]-labeled ligand from VLA4 in human Jurkat cells washed with buffer containing non activating Ca2+ and Mg2+ by competitive bindin... |
Bioorg Med Chem Lett 19: 5803-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.111
BindingDB Entry DOI: 10.7270/Q2VQ33ZJ |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309090

((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-morphol...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCOCC1 |r| Show InChI InChI=1S/C31H30Cl2N6O7S/c32-24-16-35-17-25(33)28(24)30(41)36-21-6-4-19(5-7-21)13-26(31(42)43)37-29(40)27-14-22(38-8-10-46-11-9-38)18-39(27)47(44,45)23-3-1-2-20(12-23)15-34/h1-7,12,16-17,22,26-27H,8-11,13-14,18H2,(H,36,41)(H,37,40)(H,42,43)/t22-,26+,27+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309090

((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-morphol...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCOCC1 |r| Show InChI InChI=1S/C31H30Cl2N6O7S/c32-24-16-35-17-25(33)28(24)30(41)36-21-6-4-19(5-7-21)13-26(31(42)43)37-29(40)27-14-22(38-8-10-46-11-9-38)18-39(27)47(44,45)23-3-1-2-20(12-23)15-34/h1-7,12,16-17,22,26-27H,8-11,13-14,18H2,(H,36,41)(H,37,40)(H,42,43)/t22-,26+,27+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 in presence of 90% human plasma |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309093

(2-(1-(3-cyanophenylsulfonyl)-4-(3-methylpiperidin-...)Show SMILES CC1CCCN(C1)C1CC(N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)NC(Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O Show InChI InChI=1S/C33H34Cl2N6O6S/c1-20-4-3-11-40(18-20)24-14-29(41(19-24)48(46,47)25-6-2-5-22(12-25)15-36)31(42)39-28(33(44)45)13-21-7-9-23(10-8-21)38-32(43)30-26(34)16-37-17-27(30)35/h2,5-10,12,16-17,20,24,28-29H,3-4,11,13-14,18-19H2,1H3,(H,38,43)(H,39,42)(H,44,45) | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 in presence of 90% human plasma |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309091

((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-thiomor...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCSCC1 |r| Show InChI InChI=1S/C31H30Cl2N6O6S2/c32-24-16-35-17-25(33)28(24)30(41)36-21-6-4-19(5-7-21)13-26(31(42)43)37-29(40)27-14-22(38-8-10-46-11-9-38)18-39(27)47(44,45)23-3-1-2-20(12-23)15-34/h1-7,12,16-17,22,26-27H,8-11,13-14,18H2,(H,36,41)(H,37,40)(H,42,43)/t22-,26+,27+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 in presence of 90% human plasma |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309091

((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-thiomor...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCSCC1 |r| Show InChI InChI=1S/C31H30Cl2N6O6S2/c32-24-16-35-17-25(33)28(24)30(41)36-21-6-4-19(5-7-21)13-26(31(42)43)37-29(40)27-14-22(38-8-10-46-11-9-38)18-39(27)47(44,45)23-3-1-2-20(12-23)15-34/h1-7,12,16-17,22,26-27H,8-11,13-14,18H2,(H,36,41)(H,37,40)(H,42,43)/t22-,26+,27+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309093

(2-(1-(3-cyanophenylsulfonyl)-4-(3-methylpiperidin-...)Show SMILES CC1CCCN(C1)C1CC(N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)NC(Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O Show InChI InChI=1S/C33H34Cl2N6O6S/c1-20-4-3-11-40(18-20)24-14-29(41(19-24)48(46,47)25-6-2-5-22(12-25)15-36)31(42)39-28(33(44)45)13-21-7-9-23(10-8-21)38-32(43)30-26(34)16-37-17-27(30)35/h2,5-10,12,16-17,20,24,28-29H,3-4,11,13-14,18-19H2,1H3,(H,38,43)(H,39,42)(H,44,45) | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309095
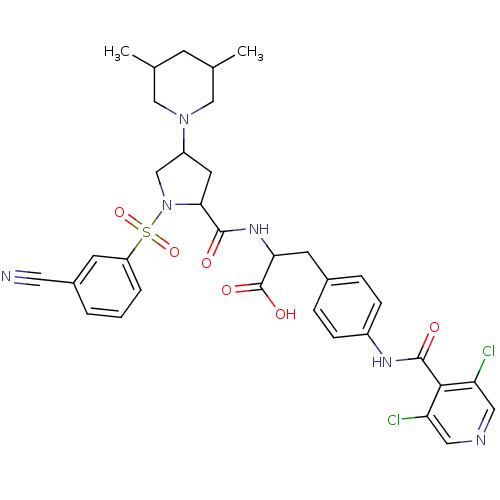
(2-(1-(3-cyanophenylsulfonyl)-4-(3,5-dimethylpiperi...)Show SMILES CC1CC(C)CN(C1)C1CC(N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)NC(Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O Show InChI InChI=1S/C34H36Cl2N6O6S/c1-20-10-21(2)18-41(17-20)25-13-30(42(19-25)49(47,48)26-5-3-4-23(11-26)14-37)32(43)40-29(34(45)46)12-22-6-8-24(9-7-22)39-33(44)31-27(35)15-38-16-28(31)36/h3-9,11,15-16,20-21,25,29-30H,10,12-13,17-19H2,1-2H3,(H,39,44)(H,40,43)(H,45,46) | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309095

(2-(1-(3-cyanophenylsulfonyl)-4-(3,5-dimethylpiperi...)Show SMILES CC1CC(C)CN(C1)C1CC(N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)NC(Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O Show InChI InChI=1S/C34H36Cl2N6O6S/c1-20-10-21(2)18-41(17-20)25-13-30(42(19-25)49(47,48)26-5-3-4-23(11-26)14-37)32(43)40-29(34(45)46)12-22-6-8-24(9-7-22)39-33(44)31-27(35)15-38-16-28(31)36/h3-9,11,15-16,20-21,25,29-30H,10,12-13,17-19H2,1-2H3,(H,39,44)(H,40,43)(H,45,46) | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 in presence of 90% human plasma |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50309085

((S)-2-((2S,4R)-4-(azetidin-1-yl)-1-(3-cyanophenyls...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCC1 |r| Show InChI InChI=1S/C30H28Cl2N6O6S/c31-23-15-34-16-24(32)27(23)29(40)35-20-7-5-18(6-8-20)12-25(30(41)42)36-28(39)26-13-21(37-9-2-10-37)17-38(26)45(43,44)22-4-1-3-19(11-22)14-33/h1,3-8,11,15-16,21,25-26H,2,9-10,12-13,17H2,(H,35,40)(H,36,39)(H,41,42)/t21-,25+,26+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [35S]-labeled ligand from VLA4 in human Jurkat cells washed with buffer containing non activating Ca2+ and Mg2+ by competitive bindin... |
Bioorg Med Chem Lett 19: 5803-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.111
BindingDB Entry DOI: 10.7270/Q2VQ33ZJ |
More data for this
Ligand-Target Pair | |
Integrin alpha-4/beta-1
(Homo sapiens (Human)) | BDBM50423686

(CHEMBL506044 | MK-0668)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)NC1CCC1 |r| Show InChI InChI=1S/C31H30Cl2N6O6S/c32-24-15-35-16-25(33)28(24)30(41)37-21-9-7-18(8-10-21)12-26(31(42)43)38-29(40)27-13-22(36-20-4-2-5-20)17-39(27)46(44,45)23-6-1-3-19(11-23)14-34/h1,3,6-11,15-16,20,22,26-27,36H,2,4-5,12-13,17H2,(H,37,41)(H,38,40)(H,42,43)/t22-,26+,27+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [35S]-labeled ligand from VLA4 in human Jurkat cells washed with buffer containing non activating Ca2+ and Mg2+ by competitive bindin... |
Bioorg Med Chem Lett 19: 5803-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.111
BindingDB Entry DOI: 10.7270/Q2VQ33ZJ |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309096

((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-(3,3-di...)Show SMILES CC1(C)CCCN(C1)[C@@H]1C[C@H](N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)N[C@@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O |r| Show InChI InChI=1S/C34H36Cl2N6O6S/c1-34(2)11-4-12-41(20-34)24-15-29(42(19-24)49(47,48)25-6-3-5-22(13-25)16-37)31(43)40-28(33(45)46)14-21-7-9-23(10-8-21)39-32(44)30-26(35)17-38-18-27(30)36/h3,5-10,13,17-18,24,28-29H,4,11-12,14-15,19-20H2,1-2H3,(H,39,44)(H,40,43)(H,45,46)/t24-,28+,29+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 in presence of 90% human plasma |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309086
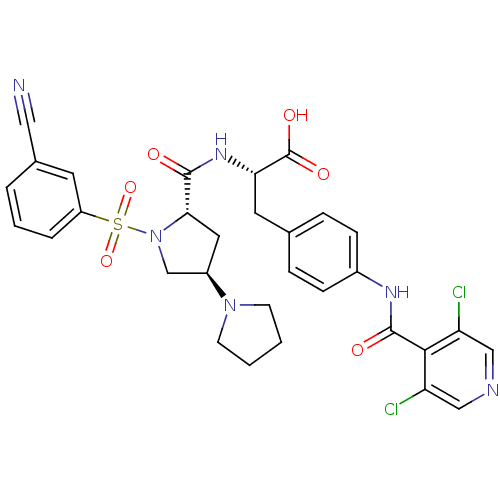
((S)-2-((3'R,5'S)-1'-(3-cyanophenylsulfonyl)-1,3'-b...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCCC1 |r| Show InChI InChI=1S/C31H30Cl2N6O6S/c32-24-16-35-17-25(33)28(24)30(41)36-21-8-6-19(7-9-21)13-26(31(42)43)37-29(40)27-14-22(38-10-1-2-11-38)18-39(27)46(44,45)23-5-3-4-20(12-23)15-34/h3-9,12,16-17,22,26-27H,1-2,10-11,13-14,18H2,(H,36,41)(H,37,40)(H,42,43)/t22-,26+,27+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 in presence of 90% human plasma |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309096

((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-(3,3-di...)Show SMILES CC1(C)CCCN(C1)[C@@H]1C[C@H](N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)N[C@@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O |r| Show InChI InChI=1S/C34H36Cl2N6O6S/c1-34(2)11-4-12-41(20-34)24-15-29(42(19-24)49(47,48)25-6-3-5-22(13-25)16-37)31(43)40-28(33(45)46)14-21-7-9-23(10-8-21)39-32(44)30-26(35)17-38-18-27(30)36/h3,5-10,13,17-18,24,28-29H,4,11-12,14-15,19-20H2,1-2H3,(H,39,44)(H,40,43)(H,45,46)/t24-,28+,29+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309094
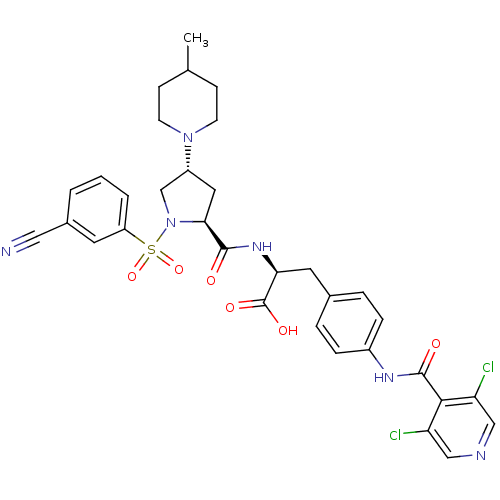
((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-(4-meth...)Show SMILES CC1CCN(CC1)[C@@H]1C[C@H](N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)N[C@@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O |r| Show InChI InChI=1S/C33H34Cl2N6O6S/c1-20-9-11-40(12-10-20)24-15-29(41(19-24)48(46,47)25-4-2-3-22(13-25)16-36)31(42)39-28(33(44)45)14-21-5-7-23(8-6-21)38-32(43)30-26(34)17-37-18-27(30)35/h2-8,13,17-18,20,24,28-29H,9-12,14-15,19H2,1H3,(H,38,43)(H,39,42)(H,44,45)/t24-,28+,29+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309094

((S)-2-((2S,4R)-1-(3-cyanophenylsulfonyl)-4-(4-meth...)Show SMILES CC1CCN(CC1)[C@@H]1C[C@H](N(C1)S(=O)(=O)c1cccc(c1)C#N)C(=O)N[C@@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)C(O)=O |r| Show InChI InChI=1S/C33H34Cl2N6O6S/c1-20-9-11-40(12-10-20)24-15-29(41(19-24)48(46,47)25-4-2-3-22(13-25)16-36)31(42)39-28(33(44)45)14-21-5-7-23(8-6-21)38-32(43)30-26(34)17-37-18-27(30)35/h2-8,13,17-18,20,24,28-29H,9-12,14-15,19H2,1H3,(H,38,43)(H,39,42)(H,44,45)/t24-,28+,29+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 in presence of 90% human plasma |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Integrin alpha-4
(Homo sapiens (Human)) | BDBM50309086

((S)-2-((3'R,5'S)-1'-(3-cyanophenylsulfonyl)-1,3'-b...)Show SMILES OC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cccc(c1)C#N)N1CCCC1 |r| Show InChI InChI=1S/C31H30Cl2N6O6S/c32-24-16-35-17-25(33)28(24)30(41)36-21-8-6-19(7-9-21)13-26(31(42)43)37-29(40)27-14-22(38-10-1-2-11-38)18-39(27)46(44,45)23-5-3-4-20(12-23)15-34/h3-9,12,16-17,22,26-27H,1-2,10-11,13-14,18H2,(H,36,41)(H,37,40)(H,42,43)/t22-,26+,27+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human VLA4 |
Bioorg Med Chem Lett 20: 1173-6 (2010)
Article DOI: 10.1016/j.bmcl.2009.12.009
BindingDB Entry DOI: 10.7270/Q2VH5NZV |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50292415

(CHEMBL504845 | Zaragozic Acid B)Show SMILES C\C=C\CCCC\C=C\CCCCC(=O)O[C@@H]1[C@@H](O)[C@]2(CCC(C)C(O)C(C)C\C=C\c3ccccc3)O[C@@]1(C(O)=O)[C@@](O)([C@H](O2)C(O)=O)C(O)=O |r| Show InChI InChI=1S/C39H54O13/c1-4-5-6-7-8-9-10-11-12-13-17-23-29(40)50-32-31(42)37(51-33(34(43)44)38(49,35(45)46)39(32,52-37)36(47)48)25-24-27(3)30(41)26(2)19-18-22-28-20-15-14-16-21-28/h4-5,10-11,14-16,18,20-22,26-27,30-33,41-42,49H,6-9,12-13,17,19,23-25H2,1-3H3,(H,43,44)(H,45,46)(H,47,48)/b5-4+,11-10+,22-18+/t26?,27?,30?,31-,32-,33-,37+,38-,39+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of rat liver squalene synthase by liqiud scintillation counting |
J Nat Prod 56: 1923-1929 (1993)
Article DOI: 10.1021/np50101a009
BindingDB Entry DOI: 10.7270/Q2M908Q1 |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385819
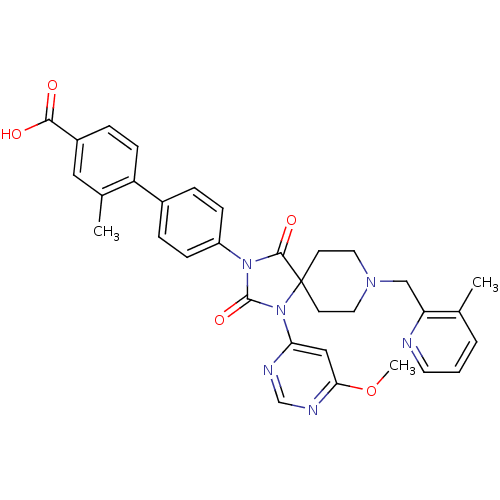
(CHEMBL2043325)Show SMILES COc1cc(ncn1)N1C(=O)N(C(=O)C11CCN(Cc2ncccc2C)CC1)c1ccc(cc1)-c1ccc(cc1C)C(O)=O Show InChI InChI=1S/C33H32N6O5/c1-21-5-4-14-34-27(21)19-37-15-12-33(13-16-37)31(42)38(32(43)39(33)28-18-29(44-3)36-20-35-28)25-9-6-23(7-10-25)26-11-8-24(30(40)41)17-22(26)2/h4-11,14,17-18,20H,12-13,15-16,19H2,1-3H3,(H,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385782
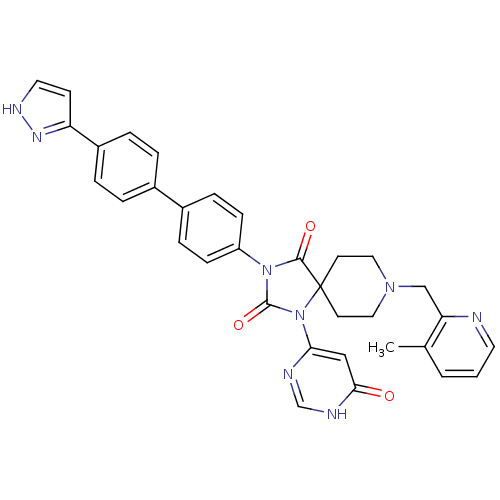
(CHEMBL2041169)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc(cc1)-c1cc[nH]n1)c1cc(=O)[nH]cn1 Show InChI InChI=1S/C33H30N8O3/c1-22-3-2-15-34-28(22)20-39-17-13-33(14-18-39)31(43)40(32(44)41(33)29-19-30(42)36-21-35-29)26-10-8-24(9-11-26)23-4-6-25(7-5-23)27-12-16-37-38-27/h2-12,15-16,19,21H,13-14,17-18,20H2,1H3,(H,37,38)(H,35,36,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385788
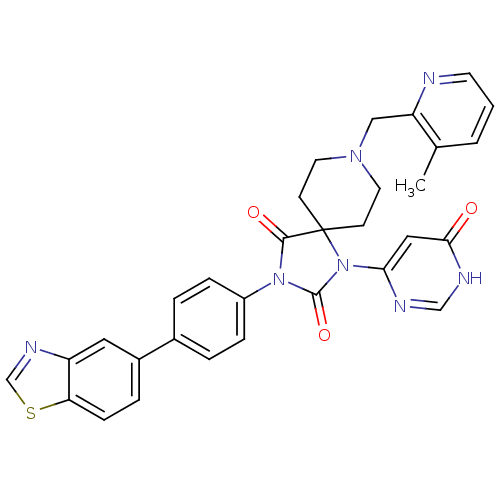
(CHEMBL2041175)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc2scnc2c1)c1cc(=O)[nH]cn1 Show InChI InChI=1S/C31H27N7O3S/c1-20-3-2-12-32-25(20)17-36-13-10-31(11-14-36)29(40)37(30(41)38(31)27-16-28(39)34-18-33-27)23-7-4-21(5-8-23)22-6-9-26-24(15-22)35-19-42-26/h2-9,12,15-16,18-19H,10-11,13-14,17H2,1H3,(H,33,34,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385796
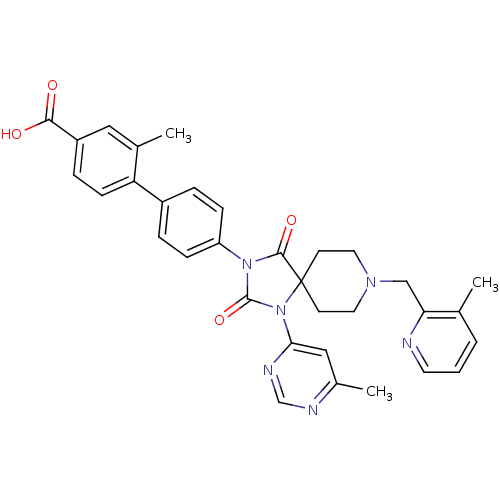
(CHEMBL2041182)Show SMILES Cc1cc(ncn1)N1C(=O)N(C(=O)C11CCN(Cc2ncccc2C)CC1)c1ccc(cc1)-c1ccc(cc1C)C(O)=O Show InChI InChI=1S/C33H32N6O4/c1-21-5-4-14-34-28(21)19-37-15-12-33(13-16-37)31(42)38(32(43)39(33)29-18-23(3)35-20-36-29)26-9-6-24(7-10-26)27-11-8-25(30(40)41)17-22(27)2/h4-11,14,17-18,20H,12-13,15-16,19H2,1-3H3,(H,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385830
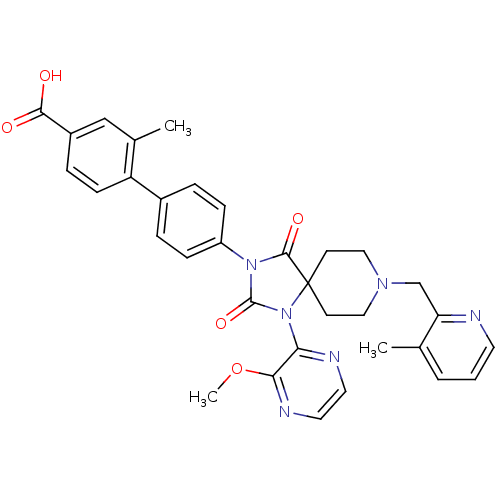
(CHEMBL2041185)Show SMILES COc1nccnc1N1C(=O)N(C(=O)C11CCN(Cc2ncccc2C)CC1)c1ccc(cc1)-c1ccc(cc1C)C(O)=O Show InChI InChI=1S/C33H32N6O5/c1-21-5-4-14-34-27(21)20-37-17-12-33(13-18-37)31(42)38(32(43)39(33)28-29(44-3)36-16-15-35-28)25-9-6-23(7-10-25)26-11-8-24(30(40)41)19-22(26)2/h4-11,14-16,19H,12-13,17-18,20H2,1-3H3,(H,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385799
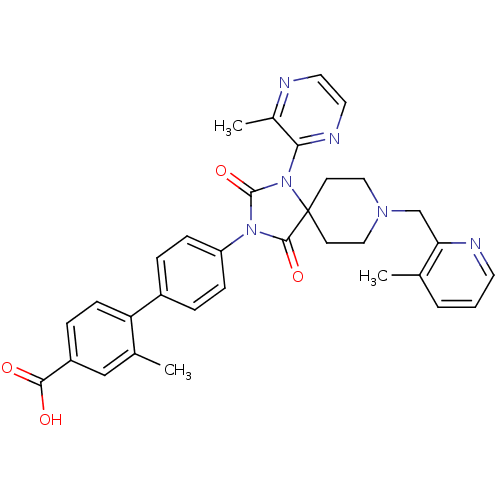
(CHEMBL2041186)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc(cc1C)C(O)=O)c1nccnc1C Show InChI InChI=1S/C33H32N6O4/c1-21-5-4-14-35-28(21)20-37-17-12-33(13-18-37)31(42)38(32(43)39(33)29-23(3)34-15-16-36-29)26-9-6-24(7-10-26)27-11-8-25(30(40)41)19-22(27)2/h4-11,14-16,19H,12-13,17-18,20H2,1-3H3,(H,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385803
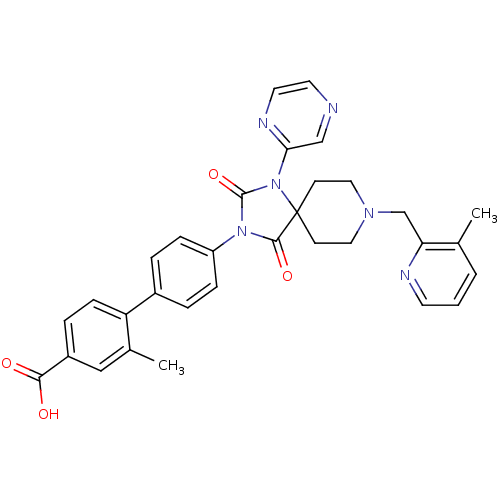
(CHEMBL2041190)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc(cc1C)C(O)=O)c1cnccn1 Show InChI InChI=1S/C32H30N6O4/c1-21-4-3-13-34-27(21)20-36-16-11-32(12-17-36)30(41)37(31(42)38(32)28-19-33-14-15-35-28)25-8-5-23(6-9-25)26-10-7-24(29(39)40)18-22(26)2/h3-10,13-15,18-19H,11-12,16-17,20H2,1-2H3,(H,39,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385805
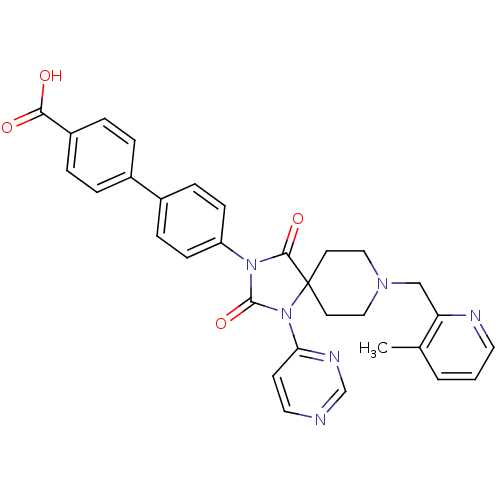
(CHEMBL2041192)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc(cc1)C(O)=O)c1ccncn1 Show InChI InChI=1S/C31H28N6O4/c1-21-3-2-15-33-26(21)19-35-17-13-31(14-18-35)29(40)36(30(41)37(31)27-12-16-32-20-34-27)25-10-8-23(9-11-25)22-4-6-24(7-5-22)28(38)39/h2-12,15-16,20H,13-14,17-19H2,1H3,(H,38,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385806
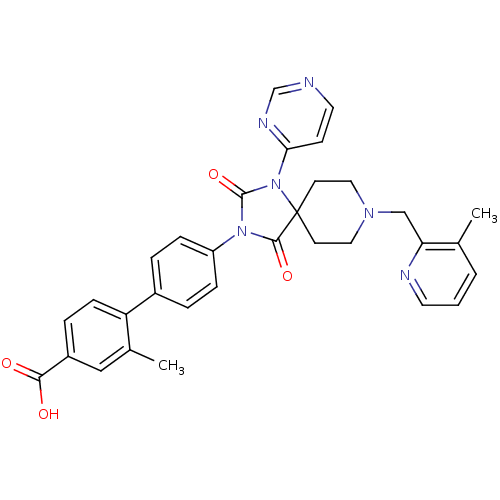
(CHEMBL2041193)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc(cc1C)C(O)=O)c1ccncn1 Show InChI InChI=1S/C32H30N6O4/c1-21-4-3-14-34-27(21)19-36-16-12-32(13-17-36)30(41)37(31(42)38(32)28-11-15-33-20-35-28)25-8-5-23(6-9-25)26-10-7-24(29(39)40)18-22(26)2/h3-11,14-15,18,20H,12-13,16-17,19H2,1-2H3,(H,39,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Prolyl hydroxylase EGLN2
(Homo sapiens (Human)) | BDBM50385814
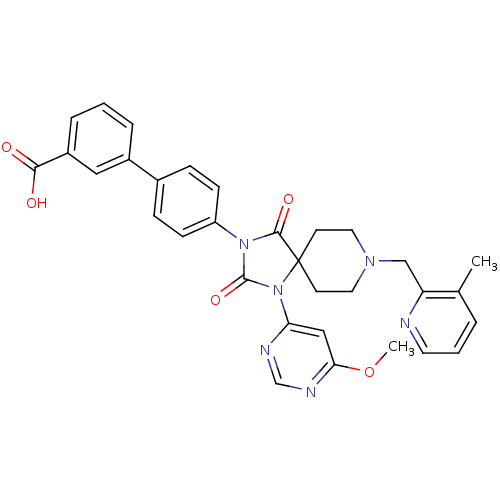
(CHEMBL2043169)Show SMILES COc1cc(ncn1)N1C(=O)N(C(=O)C11CCN(Cc2ncccc2C)CC1)c1ccc(cc1)-c1cccc(c1)C(O)=O Show InChI InChI=1S/C32H30N6O5/c1-21-5-4-14-33-26(21)19-36-15-12-32(13-16-36)30(41)37(31(42)38(32)27-18-28(43-2)35-20-34-27)25-10-8-22(9-11-25)23-6-3-7-24(17-23)29(39)40/h3-11,14,17-18,20H,12-13,15-16,19H2,1-2H3,(H,39,40) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of PHD1 |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Prolyl hydroxylase EGLN2
(Homo sapiens (Human)) | BDBM50385788

(CHEMBL2041175)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc2scnc2c1)c1cc(=O)[nH]cn1 Show InChI InChI=1S/C31H27N7O3S/c1-20-3-2-12-32-25(20)17-36-13-10-31(11-14-36)29(40)37(30(41)38(31)27-16-28(39)34-18-33-27)23-7-4-21(5-8-23)22-6-9-26-24(15-22)35-19-42-26/h2-9,12,15-16,18-19H,10-11,13-14,17H2,1H3,(H,33,34,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of PHD1 |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Egl nine homolog 1
(Homo sapiens (Human)) | BDBM50385798
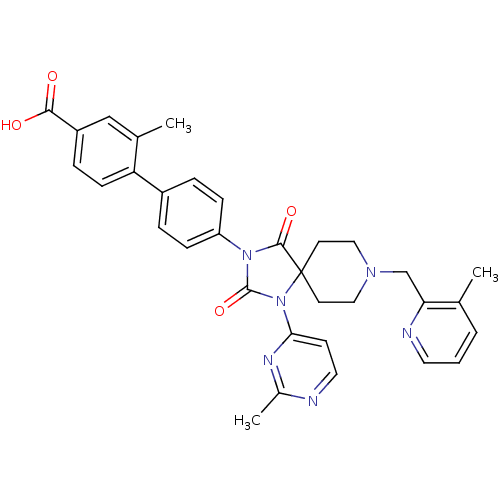
(CHEMBL2041184)Show SMILES Cc1nccc(n1)N1C(=O)N(C(=O)C11CCN(Cc2ncccc2C)CC1)c1ccc(cc1)-c1ccc(cc1C)C(O)=O Show InChI InChI=1S/C33H32N6O4/c1-21-5-4-15-35-28(21)20-37-17-13-33(14-18-37)31(42)38(32(43)39(33)29-12-16-34-23(3)36-29)26-9-6-24(7-10-26)27-11-8-25(30(40)41)19-22(27)2/h4-12,15-16,19H,13-14,17-18,20H2,1-3H3,(H,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of FLAG-tagged PHD2 expressed in baculovirus infected insect sf9 cells using biotinyl-DLDLEMLAPYIPMDDDFQL as substrate preincubated with c... |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Prolyl hydroxylase EGLN2
(Homo sapiens (Human)) | BDBM50385819

(CHEMBL2043325)Show SMILES COc1cc(ncn1)N1C(=O)N(C(=O)C11CCN(Cc2ncccc2C)CC1)c1ccc(cc1)-c1ccc(cc1C)C(O)=O Show InChI InChI=1S/C33H32N6O5/c1-21-5-4-14-34-27(21)19-37-15-12-33(13-16-37)31(42)38(32(43)39(33)28-18-29(44-3)36-20-35-28)25-9-6-23(7-10-25)26-11-8-24(30(40)41)17-22(26)2/h4-11,14,17-18,20H,12-13,15-16,19H2,1-3H3,(H,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of PHD1 |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Prolyl hydroxylase EGLN2
(Homo sapiens (Human)) | BDBM50385782

(CHEMBL2041169)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc(cc1)-c1cc[nH]n1)c1cc(=O)[nH]cn1 Show InChI InChI=1S/C33H30N8O3/c1-22-3-2-15-34-28(22)20-39-17-13-33(14-18-39)31(43)40(32(44)41(33)29-19-30(42)36-21-35-29)26-10-8-24(9-11-26)23-4-6-25(7-5-23)27-12-16-37-38-27/h2-12,15-16,19,21H,13-14,17-18,20H2,1H3,(H,37,38)(H,35,36,42) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of PHD1 |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Prolyl hydroxylase EGLN2
(Homo sapiens (Human)) | BDBM50385796

(CHEMBL2041182)Show SMILES Cc1cc(ncn1)N1C(=O)N(C(=O)C11CCN(Cc2ncccc2C)CC1)c1ccc(cc1)-c1ccc(cc1C)C(O)=O Show InChI InChI=1S/C33H32N6O4/c1-21-5-4-14-34-28(21)19-37-15-12-33(13-16-37)31(42)38(32(43)39(33)29-18-23(3)35-20-36-29)26-9-6-24(7-10-26)27-11-8-25(30(40)41)17-22(27)2/h4-11,14,17-18,20H,12-13,15-16,19H2,1-3H3,(H,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of PHD1 |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Prolyl hydroxylase EGLN2
(Homo sapiens (Human)) | BDBM50385830

(CHEMBL2041185)Show SMILES COc1nccnc1N1C(=O)N(C(=O)C11CCN(Cc2ncccc2C)CC1)c1ccc(cc1)-c1ccc(cc1C)C(O)=O Show InChI InChI=1S/C33H32N6O5/c1-21-5-4-14-34-27(21)20-37-17-12-33(13-18-37)31(42)38(32(43)39(33)28-29(44-3)36-16-15-35-28)25-9-6-23(7-10-25)26-11-8-24(30(40)41)19-22(26)2/h4-11,14-16,19H,12-13,17-18,20H2,1-3H3,(H,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of PHD1 |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Prolyl hydroxylase EGLN2
(Homo sapiens (Human)) | BDBM50385803

(CHEMBL2041190)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc(cc1C)C(O)=O)c1cnccn1 Show InChI InChI=1S/C32H30N6O4/c1-21-4-3-13-34-27(21)20-36-16-11-32(12-17-36)30(41)37(31(42)38(32)28-19-33-14-15-35-28)25-8-5-23(6-9-25)26-10-7-24(29(39)40)18-22(26)2/h3-10,13-15,18-19H,11-12,16-17,20H2,1-2H3,(H,39,40) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of PHD1 |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |
Prolyl hydroxylase EGLN2
(Homo sapiens (Human)) | BDBM50385806

(CHEMBL2041193)Show SMILES Cc1cccnc1CN1CCC2(CC1)N(C(=O)N(C2=O)c1ccc(cc1)-c1ccc(cc1C)C(O)=O)c1ccncn1 Show InChI InChI=1S/C32H30N6O4/c1-21-4-3-14-34-27(21)19-36-16-12-32(13-17-36)30(41)37(31(42)38(32)28-11-15-33-20-35-28)25-8-5-23(6-9-25)26-10-7-24(29(39)40)18-22(26)2/h3-11,14-15,18,20H,12-13,16-17,19H2,1-2H3,(H,39,40) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of PHD1 |
J Med Chem 55: 2945-59 (2012)
Article DOI: 10.1021/jm201542d
BindingDB Entry DOI: 10.7270/Q27945QW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data