 Found 4849 hits with Last Name = 'pai' and Initial = 'g'
Found 4849 hits with Last Name = 'pai' and Initial = 'g' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170092

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4ccc(Cl)cc4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H22ClN5OS/c19-13-1-3-14(4-2-13)26-9-12-7-24(8-15(12)25)6-11-5-21-17-16(11)22-10-23-18(17)20/h1-5,10,12,15,18,21,25H,6-9,20H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170082

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show InChI InChI=1S/C14H23N5OS/c1-2-21-7-10-5-19(6-11(10)20)4-9-3-16-13-12(9)17-8-18-14(13)15/h3,8,10-11,14,16,20H,2,4-7,15H2,1H3,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170089
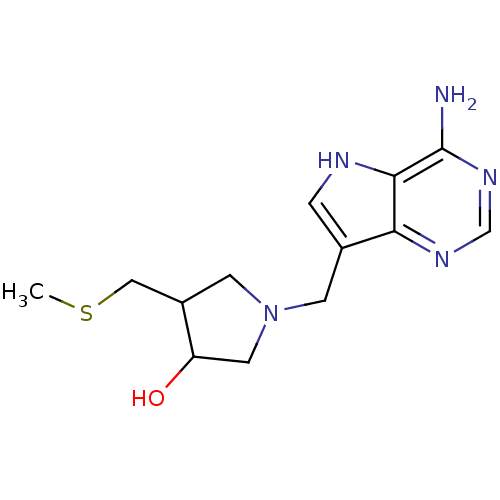
(1-(4-Amino-5H-pyrrolo[3,2-d]pyrimidin-7-ylmethyl)-...)Show InChI InChI=1S/C13H19N5OS/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,15,19H,3-6H2,1H3,(H2,14,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170086
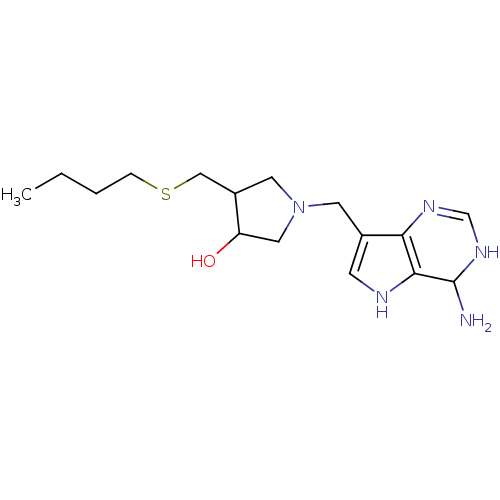
(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES CCCCSCC1CN(Cc2c[nH]c3C(N)NC=Nc23)CC1O |c:17| Show InChI InChI=1S/C16H27N5OS/c1-2-3-4-23-9-12-7-21(8-13(12)22)6-11-5-18-15-14(11)19-10-20-16(15)17/h5,10,12-13,16,18,22H,2-4,6-9,17H2,1H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170095

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES CCCSCC1CN(Cc2c[nH]c3C(N)NC=Nc23)CC1O |c:16| Show InChI InChI=1S/C15H25N5OS/c1-2-3-22-8-11-6-20(7-12(11)21)5-10-4-17-14-13(10)18-9-19-15(14)16/h4,9,11-12,15,17,21H,2-3,5-8,16H2,1H3,(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170091

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4ccncc4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C17H22N6OS/c18-17-16-15(21-10-22-17)11(5-20-16)6-23-7-12(14(24)8-23)9-25-13-1-3-19-4-2-13/h1-5,10,12,14,17,20,24H,6-9,18H2,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170098

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4ccc(F)cc4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H22FN5OS/c19-13-1-3-14(4-2-13)26-9-12-7-24(8-15(12)25)6-11-5-21-17-16(11)22-10-23-18(17)20/h1-5,10,12,15,18,21,25H,6-9,20H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170083

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4ccccc4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H23N5OS/c19-18-17-16(21-11-22-18)12(6-20-17)7-23-8-13(15(24)9-23)10-25-14-4-2-1-3-5-14/h1-6,11,13,15,18,20,24H,7-10,19H2,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170097

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES CC(C)SCC1CN(Cc2c[nH]c3C(N)NC=Nc23)CC1O |c:16| Show InChI InChI=1S/C15H25N5OS/c1-9(2)22-7-11-5-20(6-12(11)21)4-10-3-17-14-13(10)18-8-19-15(14)16/h3,8-9,11-12,15,17,21H,4-7,16H2,1-2H3,(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170088

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4cccc(Cl)c4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H22ClN5OS/c19-13-2-1-3-14(4-13)26-9-12-7-24(8-15(12)25)6-11-5-21-17-16(11)22-10-23-18(17)20/h1-5,10,12,15,18,21,25H,6-9,20H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170086

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES CCCCSCC1CN(Cc2c[nH]c3C(N)NC=Nc23)CC1O |c:17| Show InChI InChI=1S/C16H27N5OS/c1-2-3-4-23-9-12-7-21(8-13(12)22)6-11-5-18-15-14(11)19-10-20-16(15)17/h5,10,12-13,16,18,22H,2-4,6-9,17H2,1H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170092

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4ccc(Cl)cc4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H22ClN5OS/c19-13-1-3-14(4-2-13)26-9-12-7-24(8-15(12)25)6-11-5-21-17-16(11)22-10-23-18(17)20/h1-5,10,12,15,18,21,25H,6-9,20H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170094
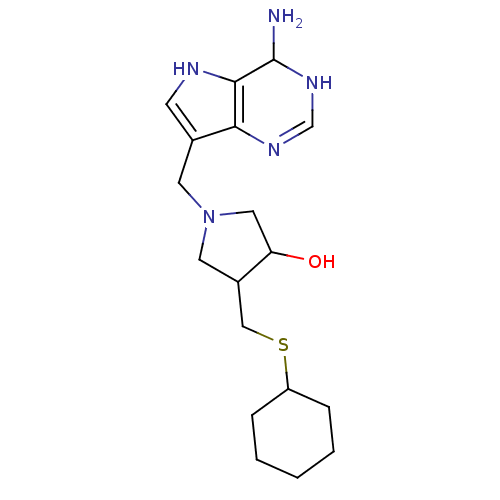
(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSC4CCCCC4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H29N5OS/c19-18-17-16(21-11-22-18)12(6-20-17)7-23-8-13(15(24)9-23)10-25-14-4-2-1-3-5-14/h6,11,13-15,18,20,24H,1-5,7-10,19H2,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170082

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show InChI InChI=1S/C14H23N5OS/c1-2-21-7-10-5-19(6-11(10)20)4-9-3-16-13-12(9)17-8-18-14(13)15/h3,8,10-11,14,16,20H,2,4-7,15H2,1H3,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM36497
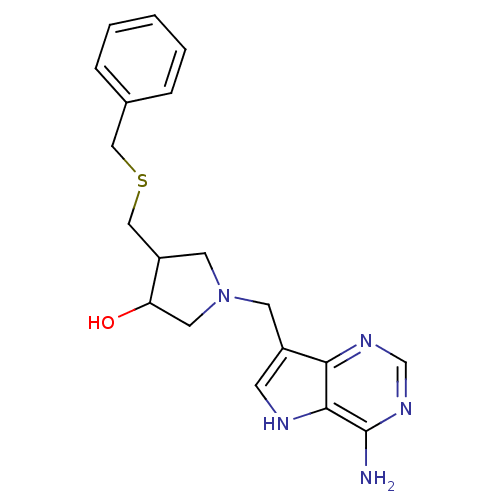
(CHEMBL191436 | DADMe-ImmA-Bn)Show SMILES Nc1ncnc2c(CN3CC(O)C(CSCc4ccccc4)C3)c[nH]c12 Show InChI InChI=1S/C19H23N5OS/c20-19-18-17(22-12-23-19)14(6-21-18)7-24-8-15(16(25)9-24)11-26-10-13-4-2-1-3-5-13/h1-6,12,15-16,21,25H,7-11H2,(H2,20,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50349850

(CHEMBL1738787)Show InChI InChI=1S/C11H13ClN6O3S/c12-9-16-10(14-5-6-19)18-11(17-9)15-7-1-3-8(4-2-7)22(13,20)21/h1-4,19H,5-6H2,(H2,13,20,21)(H2,14,15,16,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 9 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170094

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSC4CCCCC4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H29N5OS/c19-18-17-16(21-11-22-18)12(6-20-17)7-23-8-13(15(24)9-23)10-25-14-4-2-1-3-5-14/h6,11,13-15,18,20,24H,1-5,7-10,19H2,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.760 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170098

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4ccc(F)cc4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H22FN5OS/c19-13-1-3-14(4-2-13)26-9-12-7-24(8-15(12)25)6-11-5-21-17-16(11)22-10-23-18(17)20/h1-5,10,12,15,18,21,25H,6-9,20H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170097

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES CC(C)SCC1CN(Cc2c[nH]c3C(N)NC=Nc23)CC1O |c:16| Show InChI InChI=1S/C15H25N5OS/c1-9(2)22-7-11-5-20(6-12(11)21)4-10-3-17-14-13(10)18-8-19-15(14)16/h3,8-9,11-12,15,17,21H,4-7,16H2,1-2H3,(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170093
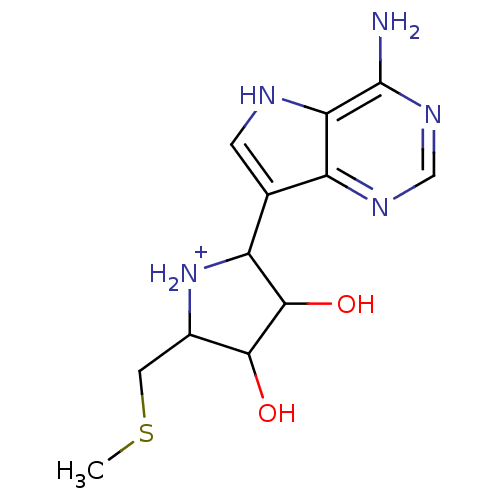
(2-(4-Amino-5H-pyrrolo[3,2-d]pyrimidin-7-yl)-3,4-di...)Show InChI InChI=1S/C12H17N5O2S/c1-20-3-6-10(18)11(19)8(17-6)5-2-14-9-7(5)15-4-16-12(9)13/h2,4,6,8,10-11,14,17-19H,3H2,1H3,(H2,13,15,16)/p+1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50349858
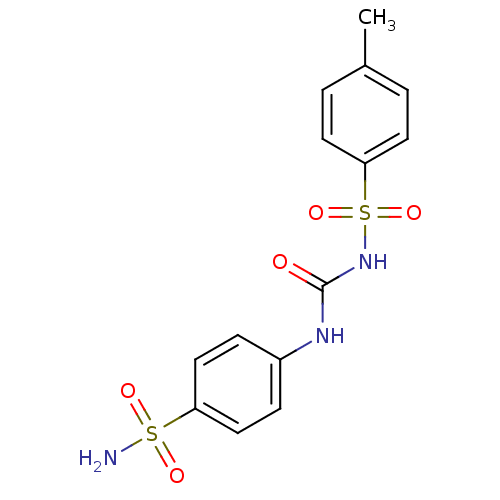
(CHEMBL78755)Show SMILES Cc1ccc(cc1)S(=O)(=O)NC(=O)Nc1ccc(cc1)S(N)(=O)=O Show InChI InChI=1S/C14H15N3O5S2/c1-10-2-6-13(7-3-10)24(21,22)17-14(18)16-11-4-8-12(9-5-11)23(15,19)20/h2-9H,1H3,(H2,15,19,20)(H2,16,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 9 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170095

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES CCCSCC1CN(Cc2c[nH]c3C(N)NC=Nc23)CC1O |c:16| Show InChI InChI=1S/C15H25N5OS/c1-2-3-22-8-11-6-20(7-12(11)21)5-10-4-17-14-13(10)18-9-19-15(14)16/h4,9,11-12,15,17,21H,2-3,5-8,16H2,1H3,(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM36497

(CHEMBL191436 | DADMe-ImmA-Bn)Show SMILES Nc1ncnc2c(CN3CC(O)C(CSCc4ccccc4)C3)c[nH]c12 Show InChI InChI=1S/C19H23N5OS/c20-19-18-17(22-12-23-19)14(6-21-18)7-24-8-15(16(25)9-24)11-26-10-13-4-2-1-3-5-13/h1-6,12,15-16,21,25H,7-11H2,(H2,20,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM50349858

(CHEMBL78755)Show SMILES Cc1ccc(cc1)S(=O)(=O)NC(=O)Nc1ccc(cc1)S(N)(=O)=O Show InChI InChI=1S/C14H15N3O5S2/c1-10-2-6-13(7-3-10)24(21,22)17-14(18)16-11-4-8-12(9-5-11)23(15,19)20/h2-9H,1H3,(H2,15,19,20)(H2,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 12 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170083

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4ccccc4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H23N5OS/c19-18-17-16(21-11-22-18)12(6-20-17)7-23-8-13(15(24)9-23)10-25-14-4-2-1-3-5-14/h1-6,11,13,15,18,20,24H,7-10,19H2,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM50349850

(CHEMBL1738787)Show InChI InChI=1S/C11H13ClN6O3S/c12-9-16-10(14-5-6-19)18-11(17-9)15-7-1-3-8(4-2-7)22(13,20)21/h1-4,19H,5-6H2,(H2,13,20,21)(H2,14,15,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 12 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170089

(1-(4-Amino-5H-pyrrolo[3,2-d]pyrimidin-7-ylmethyl)-...)Show InChI InChI=1S/C13H19N5OS/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,15,19H,3-6H2,1H3,(H2,14,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170091

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4ccncc4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C17H22N6OS/c18-17-16-15(21-10-22-17)11(5-20-16)6-23-7-12(14(24)8-23)9-25-13-1-3-19-4-2-13/h1-5,10,12,14,17,20,24H,6-9,18H2,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170088

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(CSc4cccc(Cl)c4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C18H22ClN5OS/c19-13-2-1-3-14(4-13)26-9-12-7-24(8-15(12)25)6-11-5-21-17-16(11)22-10-23-18(17)20/h1-5,10,12,15,18,21,25H,6-9,20H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170084

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show InChI InChI=1S/C14H23N5O/c1-2-3-9-5-19(7-11(9)20)6-10-4-16-13-12(10)17-8-18-14(13)15/h4,8-9,11,14,16,20H,2-3,5-7,15H2,1H3,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50349858

(CHEMBL78755)Show SMILES Cc1ccc(cc1)S(=O)(=O)NC(=O)Nc1ccc(cc1)S(N)(=O)=O Show InChI InChI=1S/C14H15N3O5S2/c1-10-2-6-13(7-3-10)24(21,22)17-14(18)16-11-4-8-12(9-5-11)23(15,19)20/h2-9H,1H3,(H2,15,19,20)(H2,16,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant carbonic anhydrase 1 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170087
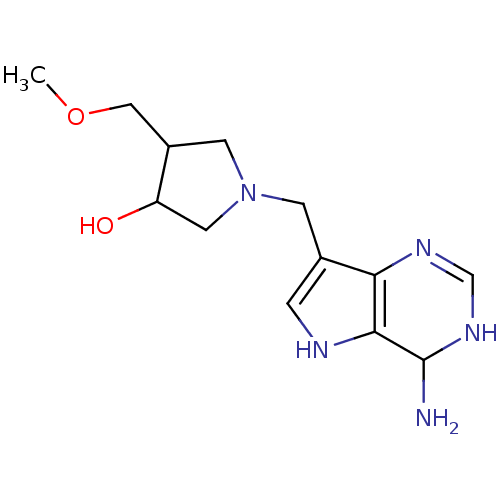
(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show InChI InChI=1S/C13H21N5O2/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,13,15,19H,3-6,14H2,1H3,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50349858

(CHEMBL78755)Show SMILES Cc1ccc(cc1)S(=O)(=O)NC(=O)Nc1ccc(cc1)S(N)(=O)=O Show InChI InChI=1S/C14H15N3O5S2/c1-10-2-6-13(7-3-10)24(21,22)17-14(18)16-11-4-8-12(9-5-11)23(15,19)20/h2-9H,1H3,(H2,15,19,20)(H2,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant carbonic anhydrase 2 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170087

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show InChI InChI=1S/C13H21N5O2/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,13,15,19H,3-6,14H2,1H3,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170084

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show InChI InChI=1S/C14H23N5O/c1-2-3-9-5-19(7-11(9)20)6-10-4-16-13-12(10)17-8-18-14(13)15/h4,8-9,11,14,16,20H,2-3,5-7,15H2,1H3,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170093

(2-(4-Amino-5H-pyrrolo[3,2-d]pyrimidin-7-yl)-3,4-di...)Show InChI InChI=1S/C12H17N5O2S/c1-20-3-6-10(18)11(19)8(17-6)5-2-14-9-7(5)15-4-16-12(9)13/h2,4,6,8,10-11,14,17-19H,3H2,1H3,(H2,13,15,16)/p+1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50080733
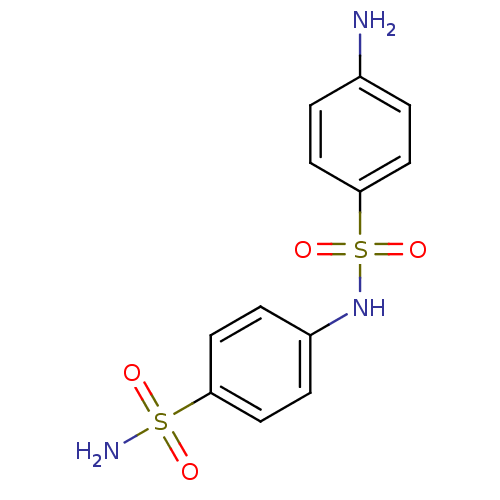
(4-(4-aminophenylsulfonamido)-1-benzenesulfonamide ...)Show InChI InChI=1S/C12H13N3O4S2/c13-9-1-5-12(6-2-9)21(18,19)15-10-3-7-11(8-4-10)20(14,16)17/h1-8,15H,13H2,(H2,14,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 9 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50349850

(CHEMBL1738787)Show InChI InChI=1S/C11H13ClN6O3S/c12-9-16-10(14-5-6-19)18-11(17-9)15-7-1-3-8(4-2-7)22(13,20)21/h1-4,19H,5-6H2,(H2,13,20,21)(H2,14,15,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant carbonic anhydrase 2 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM10857
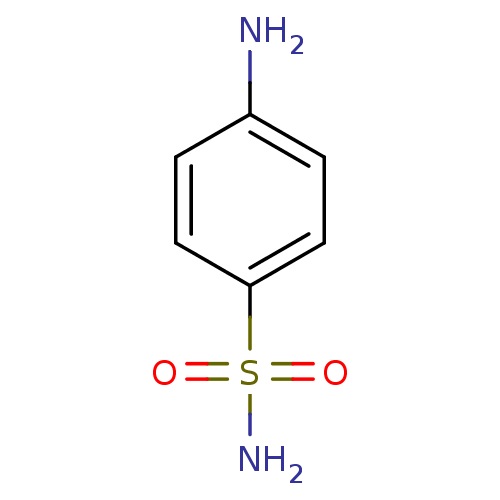
(4-aminobenzene-1-sulfonamide | CHEMBL21 | Sulfanil...)Show InChI InChI=1S/C6H8N2O2S/c7-5-1-3-6(4-2-5)11(8,9)10/h1-4H,7H2,(H2,8,9,10) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 12 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM50428408
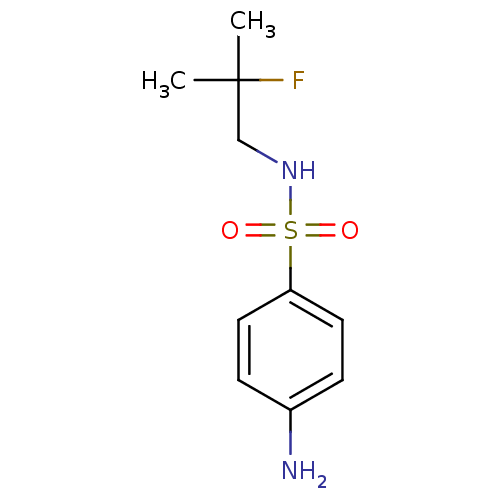
(CHEMBL2333970)Show InChI InChI=1S/C10H15FN2O2S/c1-10(2,11)7-13-16(14,15)9-5-3-8(12)4-6-9/h3-6,13H,7,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 12 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170096

(1-(4-Amino-4,5-dihydro-3H-pyrrolo[3,2-d]pyrimidin-...)Show SMILES NC1NC=Nc2c(CN3CC(O)C(COCc4ccccc4)C3)c[nH]c12 |c:3| Show InChI InChI=1S/C19H25N5O2/c20-19-18-17(22-12-23-19)14(6-21-18)7-24-8-15(16(25)9-24)11-26-10-13-4-2-1-3-5-13/h1-6,12,15-16,19,21,25H,7-11,20H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 42 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as equilibrium dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50428412
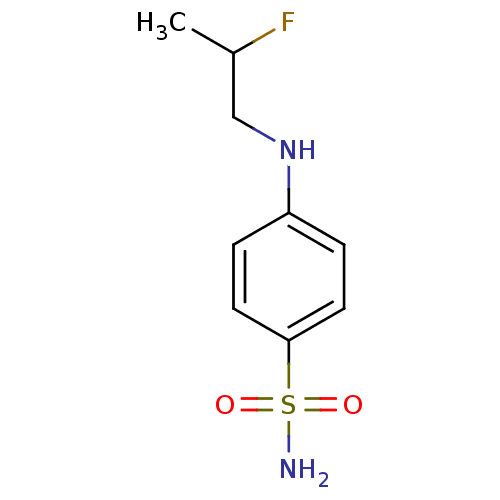
(4-(2-Fluoropropylamino)Benzenesulfonamide | CHEMBL...)Show InChI InChI=1S/C9H13FN2O2S/c1-7(10)6-12-8-2-4-9(5-3-8)15(11,13)14/h2-5,7,12H,6H2,1H3,(H2,11,13,14) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 44 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 9 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50080733

(4-(4-aminophenylsulfonamido)-1-benzenesulfonamide ...)Show InChI InChI=1S/C12H13N3O4S2/c13-9-1-5-12(6-2-9)21(18,19)15-10-3-7-11(8-4-10)20(14,16)17/h1-8,15H,13H2,(H2,14,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 46 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant carbonic anhydrase 2 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50428405
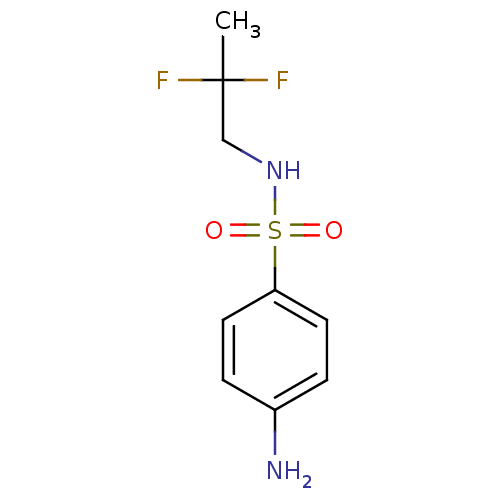
(CHEMBL2333973)Show InChI InChI=1S/C9H12F2N2O2S/c1-9(10,11)6-13-16(14,15)8-4-2-7(12)3-5-8/h2-5,13H,6,12H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 47 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 9 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50428398
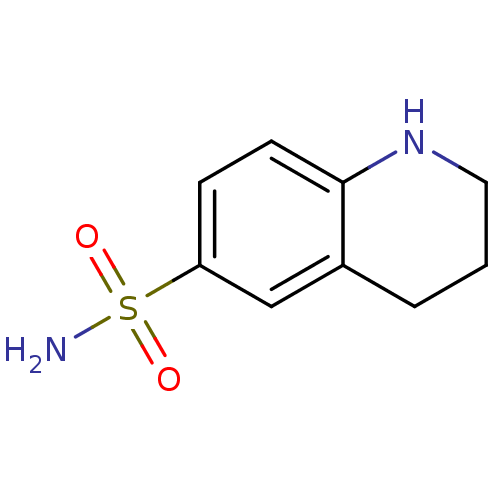
(CHEMBL2333965)Show InChI InChI=1S/C9H12N2O2S/c10-14(12,13)8-3-4-9-7(6-8)2-1-5-11-9/h3-4,6,11H,1-2,5H2,(H2,10,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 51 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 9 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50170090
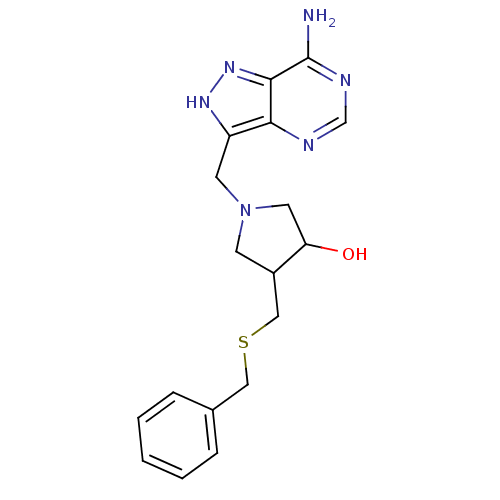
(1-(7-Amino-1H-pyrazolo[4,3-d]pyrimidin-3-ylmethyl)...)Show SMILES Nc1ncnc2c(CN3CC(O)C(CSCc4ccccc4)C3)[nH]nc12 Show InChI InChI=1S/C18H22N6OS/c19-18-17-16(20-11-21-18)14(22-23-17)7-24-6-13(15(25)8-24)10-26-9-12-4-2-1-3-5-12/h1-5,11,13,15,25H,6-10H2,(H,22,23)(H2,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP as initial dissociation constant |
J Med Chem 48: 4679-89 (2005)
Article DOI: 10.1021/jm050269z
BindingDB Entry DOI: 10.7270/Q2FJ2HKR |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50428412

(4-(2-Fluoropropylamino)Benzenesulfonamide | CHEMBL...)Show InChI InChI=1S/C9H13FN2O2S/c1-7(10)6-12-8-2-4-9(5-3-8)15(11,13)14/h2-5,7,12H,6H2,1H3,(H2,11,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 58 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant carbonic anhydrase 2 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50428401
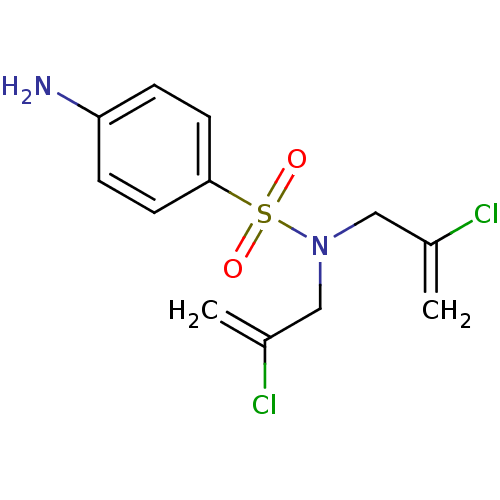
(CHEMBL2333977)Show InChI InChI=1S/C12H14Cl2N2O2S/c1-9(13)7-16(8-10(2)14)19(17,18)12-5-3-11(15)4-6-12/h3-6H,1-2,7-8,15H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 73 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 9 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM50428412

(4-(2-Fluoropropylamino)Benzenesulfonamide | CHEMBL...)Show InChI InChI=1S/C9H13FN2O2S/c1-7(10)6-12-8-2-4-9(5-3-8)15(11,13)14/h2-5,7,12H,6H2,1H3,(H2,11,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 74 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human catalytic domain of carbonic anhydrase 12 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50428408

(CHEMBL2333970)Show InChI InChI=1S/C10H15FN2O2S/c1-10(2,11)7-13-16(14,15)9-5-3-8(12)4-6-9/h3-6,13H,7,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Superacide et Chimie des syst£mes£ team-Universit£ de Poitiers
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant carbonic anhydrase 2 preincubated for 15 mins by CO2 hydration stopped-flow assay |
Bioorg Med Chem 21: 1555-63 (2013)
Article DOI: 10.1016/j.bmc.2012.05.037
BindingDB Entry DOI: 10.7270/Q25H7HM8 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data