 Found 540 hits with Last Name = 'styhler' and Initial = 'a'
Found 540 hits with Last Name = 'styhler' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128419
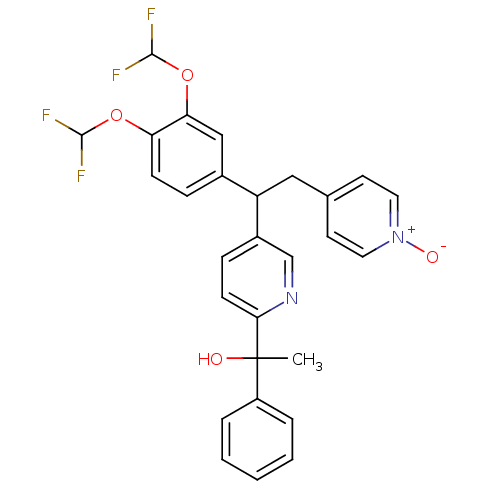
(1-{5-[1-(3,4-Bis-difluoromethoxy-phenyl)-2-(1-oxy-...)Show SMILES CC(O)(c1ccccc1)c1ccc(cn1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC(F)F)c1 Show InChI InChI=1S/C28H24F4N2O4/c1-28(35,21-5-3-2-4-6-21)25-10-8-20(17-33-25)22(15-18-11-13-34(36)14-12-18)19-7-9-23(37-26(29)30)24(16-19)38-27(31)32/h2-14,16-17,22,26-27,35H,15H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.18E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50409702
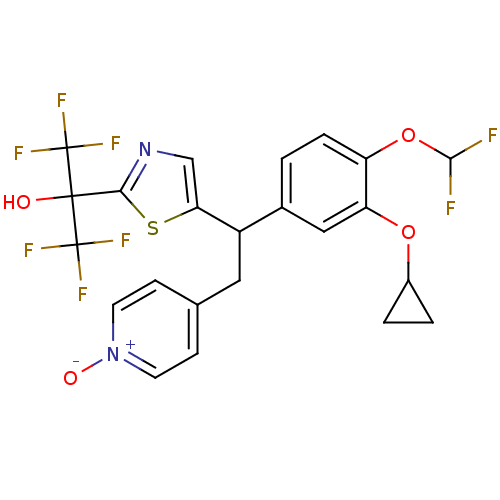
(CHEMBL2112296)Show SMILES OC(c1ncc(s1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC2CC2)c1)(C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C23H18F8N2O4S/c24-20(25)37-16-4-1-13(10-17(16)36-14-2-3-14)15(9-12-5-7-33(35)8-6-12)18-11-32-19(38-18)21(34,22(26,27)28)23(29,30)31/h1,4-8,10-11,14-15,20,34H,2-3,9H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50409699
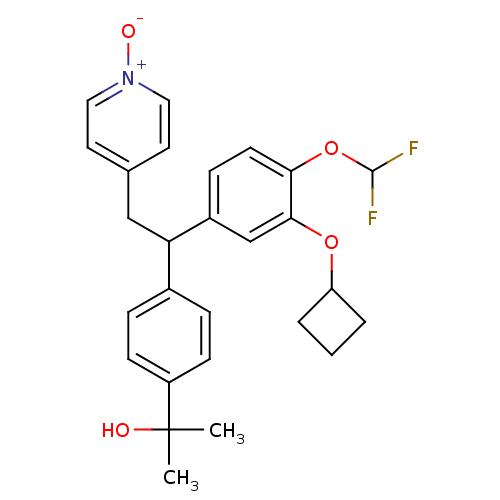
(CHEMBL2112294)Show SMILES CC(C)(O)c1ccc(cc1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC2CCC2)c1 Show InChI InChI=1S/C27H29F2NO4/c1-27(2,31)21-9-6-19(7-10-21)23(16-18-12-14-30(32)15-13-18)20-8-11-24(34-26(28)29)25(17-20)33-22-4-3-5-22/h6-15,17,22-23,26,31H,3-5,16H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.24E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128424

(2-{5-[1-(3,4-Bis-difluoromethoxy-phenyl)-2-(1-oxy-...)Show SMILES CC(C)(O)c1ccc(cn1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC(F)F)c1 Show InChI InChI=1S/C23H22F4N2O4/c1-23(2,30)20-6-4-16(13-28-20)17(11-14-7-9-29(31)10-8-14)15-3-5-18(32-21(24)25)19(12-15)33-22(26)27/h3-10,12-13,17,21-22,30H,11H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.27E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128424

(2-{5-[1-(3,4-Bis-difluoromethoxy-phenyl)-2-(1-oxy-...)Show SMILES CC(C)(O)c1ccc(cn1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC(F)F)c1 Show InChI InChI=1S/C23H22F4N2O4/c1-23(2,30)20-6-4-16(13-28-20)17(11-14-7-9-29(31)10-8-14)15-3-5-18(32-21(24)25)19(12-15)33-22(26)27/h3-10,12-13,17,21-22,30H,11H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.27E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128686

(2-{5-[1-(3-Cyclobutoxy-4-difluoromethoxy-phenyl)-2...)Show SMILES CC(C)(O)c1ccc(cn1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC2CCC2)c1 Show InChI InChI=1S/C26H28F2N2O4/c1-26(2,31)24-9-7-19(16-29-24)21(14-17-10-12-30(32)13-11-17)18-6-8-22(34-25(27)28)23(15-18)33-20-4-3-5-20/h6-13,15-16,20-21,25,31H,3-5,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128689
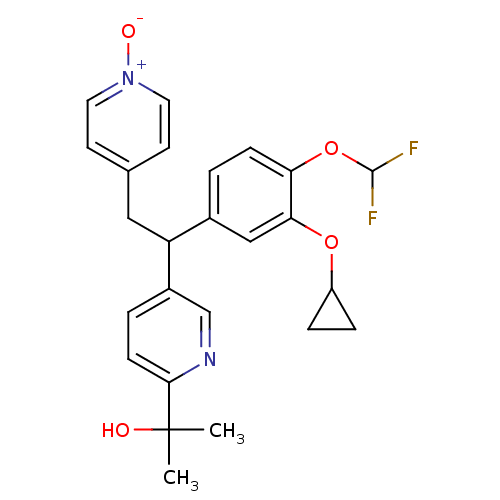
(2-{5-[1-(3-Cyclopropoxy-4-difluoromethoxy-phenyl)-...)Show SMILES CC(C)(O)c1ccc(cn1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC2CC2)c1 Show InChI InChI=1S/C25H26F2N2O4/c1-25(2,30)23-8-4-18(15-28-23)20(13-16-9-11-29(31)12-10-16)17-3-7-21(33-24(26)27)22(14-17)32-19-5-6-19/h3-4,7-12,14-15,19-20,24,30H,5-6,13H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.64E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128689

(2-{5-[1-(3-Cyclopropoxy-4-difluoromethoxy-phenyl)-...)Show SMILES CC(C)(O)c1ccc(cn1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC2CC2)c1 Show InChI InChI=1S/C25H26F2N2O4/c1-25(2,30)23-8-4-18(15-28-23)20(13-16-9-11-29(31)12-10-16)17-3-7-21(33-24(26)27)22(14-17)32-19-5-6-19/h3-4,7-12,14-15,19-20,24,30H,5-6,13H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.64E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50409700
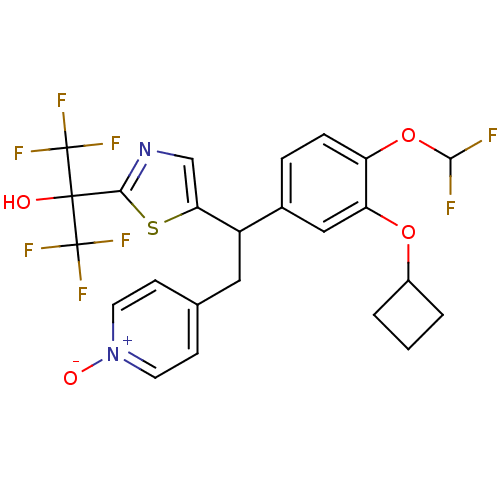
(CHEMBL2112295)Show SMILES OC(c1ncc(s1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC2CCC2)c1)(C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C24H20F8N2O4S/c25-21(26)38-17-5-4-14(11-18(17)37-15-2-1-3-15)16(10-13-6-8-34(36)9-7-13)19-12-33-20(39-19)22(35,23(27,28)29)24(30,31)32/h4-9,11-12,15-16,21,35H,1-3,10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.81E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50409701

(CHEMBL383762)Show SMILES OC(c1ncc(s1)[C@H](Cc1ccc[n+]([O-])c1)c1ccc(OC(F)F)c(OC2CC2)c1)(C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C23H18F8N2O4S/c24-20(25)37-16-6-3-13(9-17(16)36-14-4-5-14)15(8-12-2-1-7-33(35)11-12)18-10-32-19(38-18)21(34,22(26,27)28)23(29,30)31/h1-3,6-7,9-11,14-15,20,34H,4-5,8H2/t15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128683
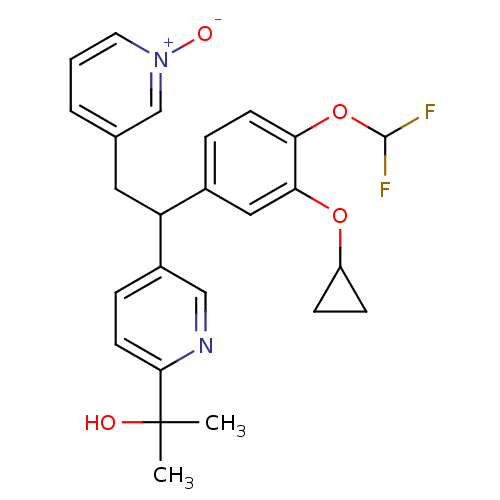
(2-{5-[1-(3-Cyclopropoxy-4-difluoromethoxy-phenyl)-...)Show SMILES CC(C)(O)c1ccc(cn1)C(Cc1ccc[n+]([O-])c1)c1ccc(OC(F)F)c(OC2CC2)c1 Show InChI InChI=1S/C25H26F2N2O4/c1-25(2,30)23-10-6-18(14-28-23)20(12-16-4-3-11-29(31)15-16)17-5-9-21(33-24(26)27)22(13-17)32-19-7-8-19/h3-6,9-11,13-15,19-20,24,30H,7-8,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.22E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128683

(2-{5-[1-(3-Cyclopropoxy-4-difluoromethoxy-phenyl)-...)Show SMILES CC(C)(O)c1ccc(cn1)C(Cc1ccc[n+]([O-])c1)c1ccc(OC(F)F)c(OC2CC2)c1 Show InChI InChI=1S/C25H26F2N2O4/c1-25(2,30)23-10-6-18(14-28-23)20(12-16-4-3-11-29(31)15-16)17-5-9-21(33-24(26)27)22(13-17)32-19-7-8-19/h3-6,9-11,13-15,19-20,24,30H,7-8,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.47E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128692
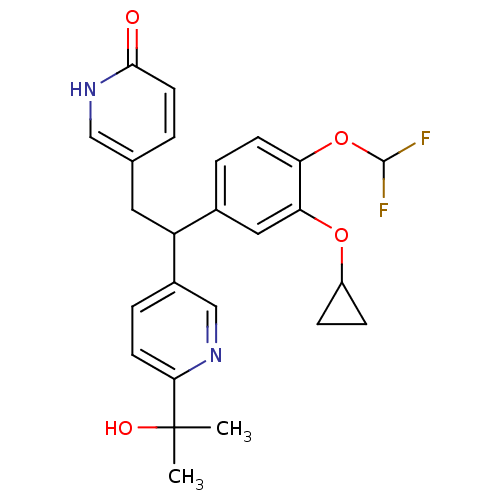
(5-{2-(3-Cyclopropoxy-4-difluoromethoxy-phenyl)-2-[...)Show SMILES CC(C)(O)c1ccc(cn1)C(Cc1ccc(=O)[nH]c1)c1ccc(OC(F)F)c(OC2CC2)c1 Show InChI InChI=1S/C25H26F2N2O4/c1-25(2,31)22-9-5-17(14-28-22)19(11-15-3-10-23(30)29-13-15)16-4-8-20(33-24(26)27)21(12-16)32-18-6-7-18/h3-5,8-10,12-14,18-19,24,31H,6-7,11H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.59E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50128685

(2-{5-[1-(3-Cyclobutoxy-4-difluoromethoxy-phenyl)-2...)Show SMILES CC(C)(O)c1ncc(s1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC2CCC2)c1 Show InChI InChI=1S/C24H26F2N2O4S/c1-24(2,29)22-27-14-21(33-22)18(12-15-8-10-28(30)11-9-15)16-6-7-19(32-23(25)26)20(13-16)31-17-4-3-5-17/h6-11,13-14,17-18,23,29H,3-5,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5.84E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50174020
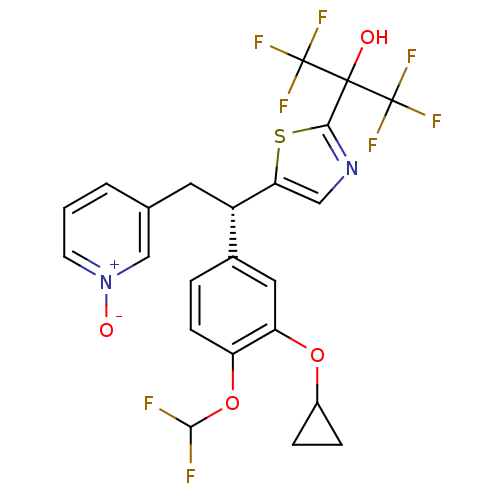
((S)-(+)-3-{2-[(3-Cyclopropyloxy-4-difluromethoxy)-...)Show SMILES OC(c1ncc(s1)[C@@H](Cc1ccc[n+]([O-])c1)c1ccc(OC(F)F)c(OC2CC2)c1)(C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C23H18F8N2O4S/c24-20(25)37-16-6-3-13(9-17(16)36-14-4-5-14)15(8-12-2-1-7-33(35)11-12)18-10-32-19(38-18)21(34,22(26,27)28)23(29,30)31/h1-3,6-7,9-11,14-15,20,34H,4-5,8H2/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.14E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
In vitro displacement of [35S]-MK- 499 from HEK 293 cells stably transfected with hERG voltage-gated potassium channel subunit Kv11.1 |
J Med Chem 46: 2413-26 (2003)
Article DOI: 10.1021/jm0204542
BindingDB Entry DOI: 10.7270/Q24X5756 |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50183805
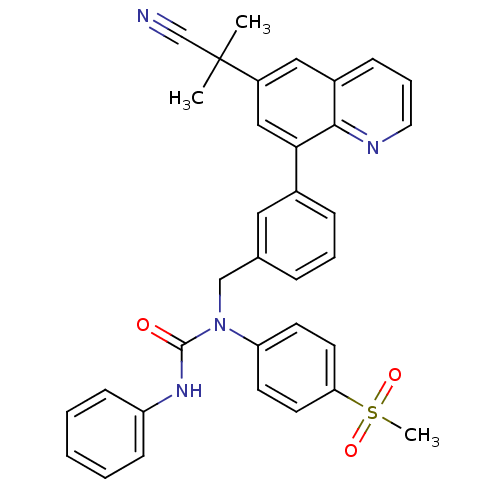
(1-((3-(6-(2-cyanopropan-2-yl)quinolin-8-yl)phenyl)...)Show SMILES CC(C)(C#N)c1cc(-c2cccc(CN(C(=O)Nc3ccccc3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C34H30N4O3S/c1-34(2,23-35)27-20-26-11-8-18-36-32(26)31(21-27)25-10-7-9-24(19-25)22-38(33(39)37-28-12-5-4-6-13-28)29-14-16-30(17-15-29)42(3,40)41/h4-21H,22H2,1-3H3,(H,37,39) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4B |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Homo sapiens (Human)) | BDBM50183805

(1-((3-(6-(2-cyanopropan-2-yl)quinolin-8-yl)phenyl)...)Show SMILES CC(C)(C#N)c1cc(-c2cccc(CN(C(=O)Nc3ccccc3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C34H30N4O3S/c1-34(2,23-35)27-20-26-11-8-18-36-32(26)31(21-27)25-10-7-9-24(19-25)22-38(33(39)37-28-12-5-4-6-13-28)29-14-16-30(17-15-29)42(3,40)41/h4-21H,22H2,1-3H3,(H,37,39) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4C |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50274028

(CHEMBL499477 | N-(3,5-dichloropyridin-4-yl)-1-(3-(...)Show SMILES Cc1noc(n1)C(=C\c1cccc(c1)-n1cc(C(=O)Nc2c(Cl)cncc2Cl)c(=O)c2cccnc12)\c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C32H22Cl2N6O5S/c1-18-37-32(45-39-18)24(20-8-10-22(11-9-20)46(2,43)44)14-19-5-3-6-21(13-19)40-17-25(29(41)23-7-4-12-36-30(23)40)31(42)38-28-26(33)15-35-16-27(28)34/h3-17H,1-2H3,(H,35,38,42)/b24-14+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Intrinsic inhibition of GST-fused human PDE4A expressed in SF9 cells |
Bioorg Med Chem Lett 18: 5554-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.09.009
BindingDB Entry DOI: 10.7270/Q24X57M1 |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50174030
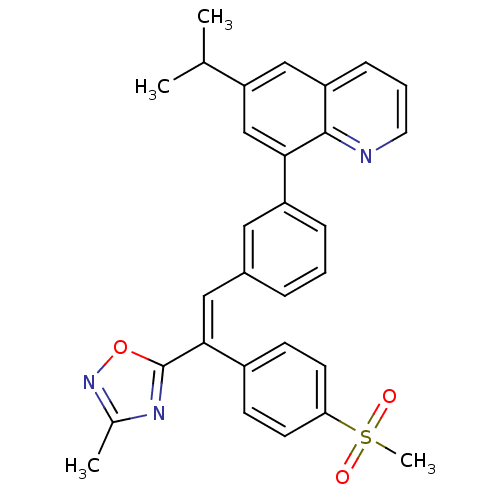
((E)-6-isopropyl-8-(3-(2-(3-methyl-1,2,4-oxadiazol-...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(\c3nc(C)no3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C30H27N3O3S/c1-19(2)25-17-24-9-6-14-31-29(24)27(18-25)23-8-5-7-21(15-23)16-28(30-32-20(3)33-36-30)22-10-12-26(13-11-22)37(4,34)35/h5-19H,1-4H3/b28-16+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4B |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50183805

(1-((3-(6-(2-cyanopropan-2-yl)quinolin-8-yl)phenyl)...)Show SMILES CC(C)(C#N)c1cc(-c2cccc(CN(C(=O)Nc3ccccc3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C34H30N4O3S/c1-34(2,23-35)27-20-26-11-8-18-36-32(26)31(21-27)25-10-7-9-24(19-25)22-38(33(39)37-28-12-5-4-6-13-28)29-14-16-30(17-15-29)42(3,40)41/h4-21H,22H2,1-3H3,(H,37,39) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4D |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50183794
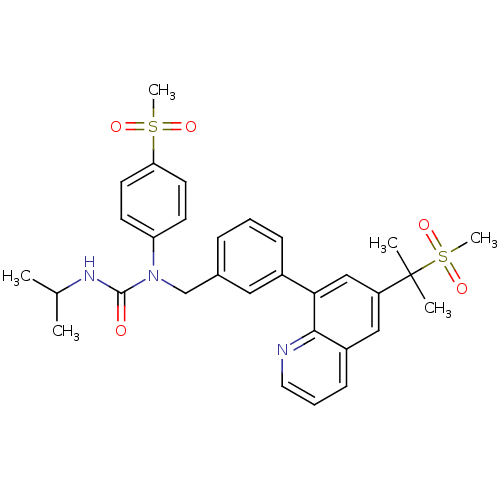
(3-isopropyl-1-(4-(methylsulfonyl)phenyl)-1-((3-(6-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)S(C)(=O)=O)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H35N3O5S2/c1-21(2)33-30(35)34(26-12-14-27(15-13-26)40(5,36)37)20-22-9-7-10-23(17-22)28-19-25(31(3,4)41(6,38)39)18-24-11-8-16-32-29(24)28/h7-19,21H,20H2,1-6H3,(H,33,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4B |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50183803

(CHEMBL206968 | N-((3-(6-(2-cyanopropan-2-yl)quinol...)Show SMILES Cc1cc(no1)C(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)C#N)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C32H28N4O4S/c1-21-15-29(35-40-21)31(37)36(26-10-12-27(13-11-26)41(4,38)39)19-22-7-5-8-23(16-22)28-18-25(32(2,3)20-33)17-24-9-6-14-34-30(24)28/h5-18H,19H2,1-4H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4D |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50183795
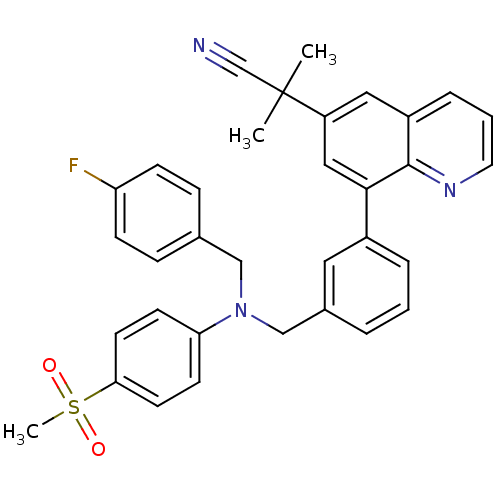
(2-(8-(3-(((4-fluorobenzyl)(4-(methylsulfonyl)pheny...)Show SMILES CC(C)(C#N)c1cc(-c2cccc(CN(Cc3ccc(F)cc3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C34H30FN3O2S/c1-34(2,23-36)28-19-27-8-5-17-37-33(27)32(20-28)26-7-4-6-25(18-26)22-38(21-24-9-11-29(35)12-10-24)30-13-15-31(16-14-30)41(3,39)40/h4-20H,21-22H2,1-3H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4B |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50183792

(1-((3-(6-(2-cyanopropan-2-yl)quinolin-8-yl)phenyl)...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)C#N)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H32N4O3S/c1-21(2)34-30(36)35(26-11-13-27(14-12-26)39(5,37)38)19-22-8-6-9-23(16-22)28-18-25(31(3,4)20-32)17-24-10-7-15-33-29(24)28/h6-18,21H,19H2,1-5H3,(H,34,36) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4A |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50183794

(3-isopropyl-1-(4-(methylsulfonyl)phenyl)-1-((3-(6-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)S(C)(=O)=O)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H35N3O5S2/c1-21(2)33-30(35)34(26-12-14-27(15-13-26)40(5,36)37)20-22-9-7-10-23(17-22)28-19-25(31(3,4)41(6,38)39)18-24-11-8-16-32-29(24)28/h7-19,21H,20H2,1-6H3,(H,33,35) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4D |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50183792

(1-((3-(6-(2-cyanopropan-2-yl)quinolin-8-yl)phenyl)...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)C#N)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H32N4O3S/c1-21(2)34-30(36)35(26-11-13-27(14-12-26)39(5,37)38)19-22-8-6-9-23(16-22)28-18-25(31(3,4)20-32)17-24-10-7-15-33-29(24)28/h6-18,21H,19H2,1-5H3,(H,34,36) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4D |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50183792

(1-((3-(6-(2-cyanopropan-2-yl)quinolin-8-yl)phenyl)...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)C#N)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H32N4O3S/c1-21(2)34-30(36)35(26-11-13-27(14-12-26)39(5,37)38)19-22-8-6-9-23(16-22)28-18-25(31(3,4)20-32)17-24-10-7-15-33-29(24)28/h6-18,21H,19H2,1-5H3,(H,34,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4B |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50183794

(3-isopropyl-1-(4-(methylsulfonyl)phenyl)-1-((3-(6-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)S(C)(=O)=O)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H35N3O5S2/c1-21(2)33-30(35)34(26-12-14-27(15-13-26)40(5,36)37)20-22-9-7-10-23(17-22)28-19-25(31(3,4)41(6,38)39)18-24-11-8-16-32-29(24)28/h7-19,21H,20H2,1-6H3,(H,33,35) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4A |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50183805

(1-((3-(6-(2-cyanopropan-2-yl)quinolin-8-yl)phenyl)...)Show SMILES CC(C)(C#N)c1cc(-c2cccc(CN(C(=O)Nc3ccccc3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C34H30N4O3S/c1-34(2,23-35)27-20-26-11-8-18-36-32(26)31(21-27)25-10-7-9-24(19-25)22-38(33(39)37-28-12-5-4-6-13-28)29-14-16-30(17-15-29)42(3,40)41/h4-21H,22H2,1-3H3,(H,37,39) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4A |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50183808

(CHEMBL383225 | N-((3-(6-(2-cyanopropan-2-yl)quinol...)Show SMILES CC(C)(C#N)c1cc(-c2cccc(CN(C(=O)c3ccccc3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C34H29N3O3S/c1-34(2,23-35)28-20-27-13-8-18-36-32(27)31(21-28)26-12-7-9-24(19-26)22-37(33(38)25-10-5-4-6-11-25)29-14-16-30(17-15-29)41(3,39)40/h4-21H,22H2,1-3H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4B |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50183803

(CHEMBL206968 | N-((3-(6-(2-cyanopropan-2-yl)quinol...)Show SMILES Cc1cc(no1)C(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)C#N)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C32H28N4O4S/c1-21-15-29(35-40-21)31(37)36(26-10-12-27(13-11-26)41(4,38)39)19-22-7-5-8-23(16-22)28-18-25(32(2,3)20-33)17-24-9-6-14-34-30(24)28/h5-18H,19H2,1-4H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4B |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50174030

((E)-6-isopropyl-8-(3-(2-(3-methyl-1,2,4-oxadiazol-...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(\c3nc(C)no3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C30H27N3O3S/c1-19(2)25-17-24-9-6-14-31-29(24)27(18-25)23-8-5-7-21(15-23)16-28(30-32-20(3)33-36-30)22-10-12-26(13-11-22)37(4,34)35/h5-19H,1-4H3/b28-16+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4A |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50274031
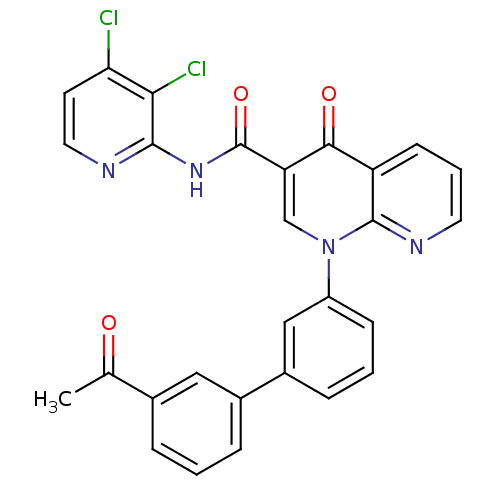
(1-(3'-Acetyl-biphenyl-3-yl)-4-oxo-1,4-dihydro-[1,8...)Show SMILES CC(=O)c1cccc(c1)-c1cccc(c1)-n1cc(C(=O)Nc2nccc(Cl)c2Cl)c(=O)c2cccnc12 Show InChI InChI=1S/C28H18Cl2N4O3/c1-16(35)17-5-2-6-18(13-17)19-7-3-8-20(14-19)34-15-22(25(36)21-9-4-11-32-27(21)34)28(37)33-26-24(30)23(29)10-12-31-26/h2-15H,1H3,(H,31,33,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Intrinsic inhibition of GST-fused human PDE4A expressed in SF9 cells |
Bioorg Med Chem Lett 18: 5554-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.09.009
BindingDB Entry DOI: 10.7270/Q24X57M1 |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50174025
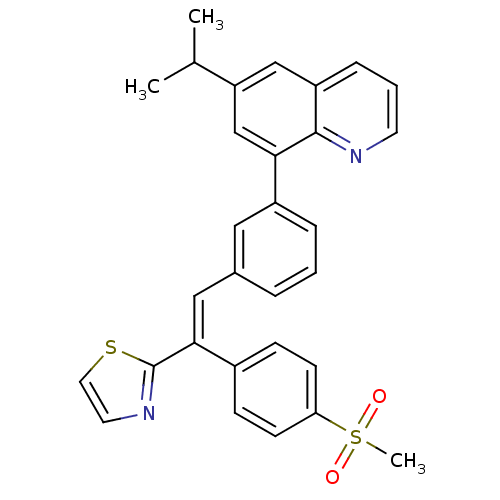
(6-isopropyl-8-(3-(2-(4-(methylsulfonyl)phenyl)-2-(...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(\c3nccs3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C30H26N2O2S2/c1-20(2)25-18-24-8-5-13-31-29(24)27(19-25)23-7-4-6-21(16-23)17-28(30-32-14-15-35-30)22-9-11-26(12-10-22)36(3,33)34/h4-20H,1-3H3/b28-17+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4D |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50183808

(CHEMBL383225 | N-((3-(6-(2-cyanopropan-2-yl)quinol...)Show SMILES CC(C)(C#N)c1cc(-c2cccc(CN(C(=O)c3ccccc3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C34H29N3O3S/c1-34(2,23-35)28-20-27-13-8-18-36-32(27)31(21-28)26-12-7-9-24(19-26)22-37(33(38)25-10-5-4-6-11-25)29-14-16-30(17-15-29)41(3,39)40/h4-21H,22H2,1-3H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4A |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50174030

((E)-6-isopropyl-8-(3-(2-(3-methyl-1,2,4-oxadiazol-...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(\c3nc(C)no3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C30H27N3O3S/c1-19(2)25-17-24-9-6-14-31-29(24)27(18-25)23-8-5-7-21(15-23)16-28(30-32-20(3)33-36-30)22-10-12-26(13-11-22)37(4,34)35/h5-19H,1-4H3/b28-16+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4D |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50174028
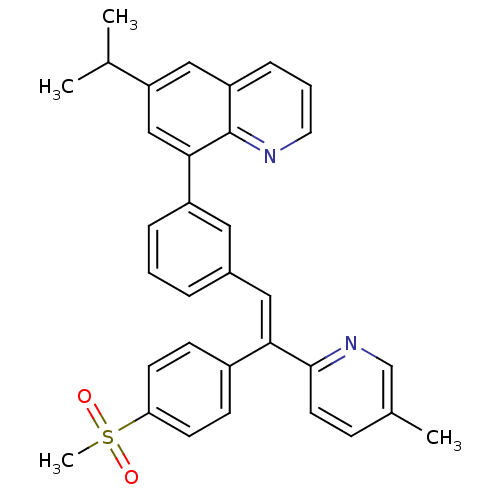
((E)-6-isopropyl-8-(3-(2-(5-methylpyridin-2-yl)-2-(...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(/c3ccc(cc3)S(C)(=O)=O)c3ccc(C)cn3)c2)c2ncccc2c1 Show InChI InChI=1S/C33H30N2O2S/c1-22(2)28-19-27-9-6-16-34-33(27)31(20-28)26-8-5-7-24(17-26)18-30(32-15-10-23(3)21-35-32)25-11-13-29(14-12-25)38(4,36)37/h5-22H,1-4H3/b30-18+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4B |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50174028

((E)-6-isopropyl-8-(3-(2-(5-methylpyridin-2-yl)-2-(...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(/c3ccc(cc3)S(C)(=O)=O)c3ccc(C)cn3)c2)c2ncccc2c1 Show InChI InChI=1S/C33H30N2O2S/c1-22(2)28-19-27-9-6-16-34-33(27)31(20-28)26-8-5-7-24(17-26)18-30(32-15-10-23(3)21-35-32)25-11-13-29(14-12-25)38(4,36)37/h5-22H,1-4H3/b30-18+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4A |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50174028

((E)-6-isopropyl-8-(3-(2-(5-methylpyridin-2-yl)-2-(...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(/c3ccc(cc3)S(C)(=O)=O)c3ccc(C)cn3)c2)c2ncccc2c1 Show InChI InChI=1S/C33H30N2O2S/c1-22(2)28-19-27-9-6-16-34-33(27)31(20-28)26-8-5-7-24(17-26)18-30(32-15-10-23(3)21-35-32)25-11-13-29(14-12-25)38(4,36)37/h5-22H,1-4H3/b30-18+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4D |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50174025

(6-isopropyl-8-(3-(2-(4-(methylsulfonyl)phenyl)-2-(...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(\c3nccs3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C30H26N2O2S2/c1-20(2)25-18-24-8-5-13-31-29(24)27(19-25)23-7-4-6-21(16-23)17-28(30-32-14-15-35-30)22-9-11-26(12-10-22)36(3,33)34/h4-20H,1-3H3/b28-17+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4B |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50183791
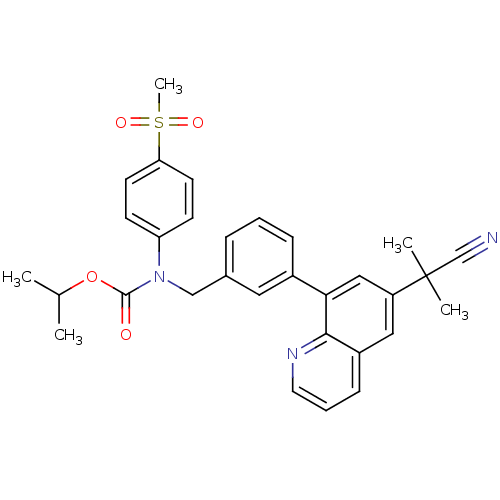
(CHEMBL209295 | isopropyl (3-(6-(2-cyanopropan-2-yl...)Show SMILES CC(C)OC(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)C#N)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H31N3O4S/c1-21(2)38-30(35)34(26-11-13-27(14-12-26)39(5,36)37)19-22-8-6-9-23(16-22)28-18-25(31(3,4)20-32)17-24-10-7-15-33-29(24)28/h6-18,21H,19H2,1-5H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4B |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50183791

(CHEMBL209295 | isopropyl (3-(6-(2-cyanopropan-2-yl...)Show SMILES CC(C)OC(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)C#N)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H31N3O4S/c1-21(2)38-30(35)34(26-11-13-27(14-12-26)39(5,36)37)19-22-8-6-9-23(16-22)28-18-25(31(3,4)20-32)17-24-10-7-15-33-29(24)28/h6-18,21H,19H2,1-5H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4A |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50125066
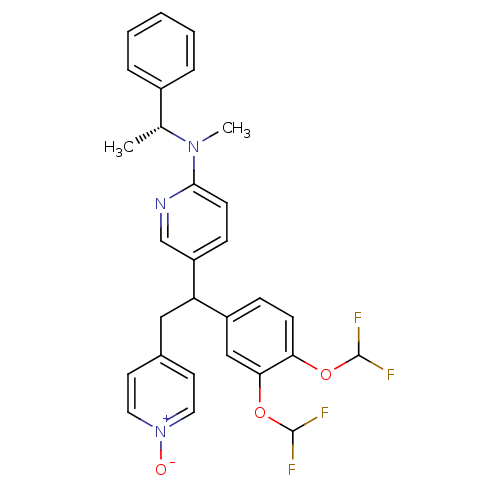
(CHEMBL165889 | {5-[1-(3,4-Bis-difluoromethoxy-phen...)Show SMILES C[C@@H](N(C)c1ccc(cn1)C(Cc1cc[n+]([O-])cc1)c1ccc(OC(F)F)c(OC(F)F)c1)c1ccccc1 Show InChI InChI=1S/C29H27F4N3O3/c1-19(21-6-4-3-5-7-21)35(2)27-11-9-23(18-34-27)24(16-20-12-14-36(37)15-13-20)22-8-10-25(38-28(30)31)26(17-22)39-29(32)33/h3-15,17-19,24,28-29H,16H2,1-2H3/t19-,24?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against human PDE4A expressed isoform using construct representing the common region of spliced variants as GST-fusion protein in... |
Bioorg Med Chem Lett 13: 741-4 (2003)
BindingDB Entry DOI: 10.7270/Q2PG1R3G |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50183791

(CHEMBL209295 | isopropyl (3-(6-(2-cyanopropan-2-yl...)Show SMILES CC(C)OC(=O)N(Cc1cccc(c1)-c1cc(cc2cccnc12)C(C)(C)C#N)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H31N3O4S/c1-21(2)38-30(35)34(26-11-13-27(14-12-26)39(5,36)37)19-22-8-6-9-23(16-22)28-18-25(31(3,4)20-32)17-24-10-7-15-33-29(24)28/h6-18,21H,19H2,1-5H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4D |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50174023
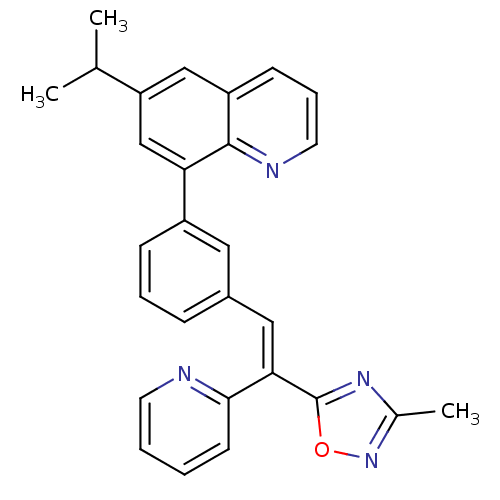
((E)-6-isopropyl-8-(3-(2-(3-methyl-1,2,4-oxadiazol-...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(\c3nc(C)no3)c3ccccn3)c2)c2ncccc2c1 Show InChI InChI=1S/C28H24N4O/c1-18(2)23-16-22-10-7-13-30-27(22)24(17-23)21-9-6-8-20(14-21)15-25(26-11-4-5-12-29-26)28-31-19(3)32-33-28/h4-18H,1-3H3/b25-15+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4B |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50183808

(CHEMBL383225 | N-((3-(6-(2-cyanopropan-2-yl)quinol...)Show SMILES CC(C)(C#N)c1cc(-c2cccc(CN(C(=O)c3ccccc3)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C34H29N3O3S/c1-34(2,23-35)28-20-27-13-8-18-36-32(27)31(21-28)26-12-7-9-24(19-26)22-37(33(38)25-10-5-4-6-11-25)29-14-16-30(17-15-29)41(3,39)40/h4-21H,22H2,1-3H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4D |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50174018
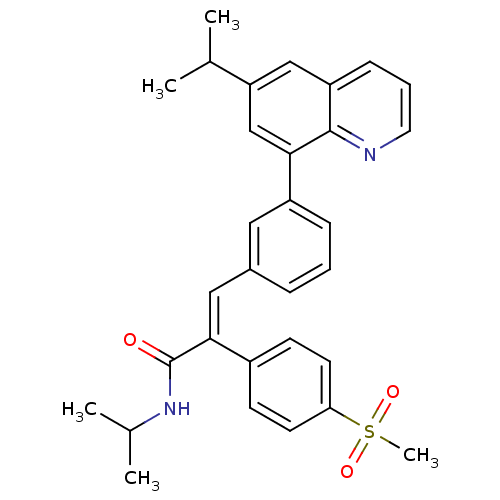
((E)-N-isopropyl-3-(3-(6-isopropylquinolin-8-yl)phe...)Show SMILES CC(C)NC(=O)C(=C\c1cccc(c1)-c1cc(cc2cccnc12)C(C)C)\c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H32N2O3S/c1-20(2)26-18-25-10-7-15-32-30(25)28(19-26)24-9-6-8-22(16-24)17-29(31(34)33-21(3)4)23-11-13-27(14-12-23)37(5,35)36/h6-21H,1-5H3,(H,33,34)/b29-17+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4A |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50183798
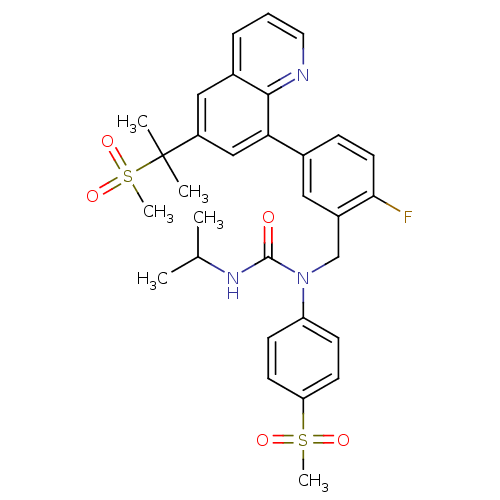
(1-((2-fluoro-5-(6-(2-(methylsulfonyl)propan-2-yl)q...)Show SMILES CC(C)NC(=O)N(Cc1cc(ccc1F)-c1cc(cc2cccnc12)C(C)(C)S(C)(=O)=O)c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H34FN3O5S2/c1-20(2)34-30(36)35(25-10-12-26(13-11-25)41(5,37)38)19-23-16-21(9-14-28(23)32)27-18-24(31(3,4)42(6,39)40)17-22-8-7-15-33-29(22)27/h7-18,20H,19H2,1-6H3,(H,34,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human GST-PDE4B |
Bioorg Med Chem Lett 16: 2608-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.043
BindingDB Entry DOI: 10.7270/Q2F18Z9N |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50174013
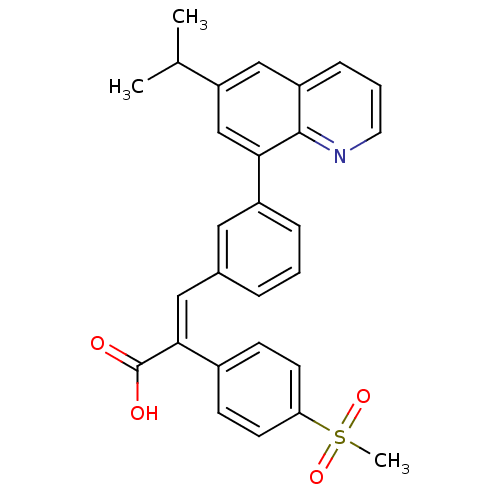
((E)-3-(3-(6-isopropylquinolin-8-yl)phenyl)-2-(4-(m...)Show SMILES CC(C)c1cc(-c2cccc(\C=C(\C(O)=O)c3ccc(cc3)S(C)(=O)=O)c2)c2ncccc2c1 Show InChI InChI=1S/C28H25NO4S/c1-18(2)23-16-22-8-5-13-29-27(22)25(17-23)21-7-4-6-19(14-21)15-26(28(30)31)20-9-11-24(12-10-20)34(3,32)33/h4-18H,1-3H3,(H,30,31)/b26-15+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4A |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50174018

((E)-N-isopropyl-3-(3-(6-isopropylquinolin-8-yl)phe...)Show SMILES CC(C)NC(=O)C(=C\c1cccc(c1)-c1cc(cc2cccnc12)C(C)C)\c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C31H32N2O3S/c1-20(2)26-18-25-10-7-15-32-30(25)28(19-26)24-9-6-8-22(16-24)17-29(31(34)33-21(3)4)23-11-13-27(14-12-23)37(5,35)36/h6-21H,1-5H3,(H,33,34)/b29-17+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDE4B |
Bioorg Med Chem Lett 15: 5241-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.036
BindingDB Entry DOI: 10.7270/Q2B56KHG |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data