 Found 16 hits in this display
Found 16 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Beta-arrestin-1
(RABBIT) | BDBM50018550
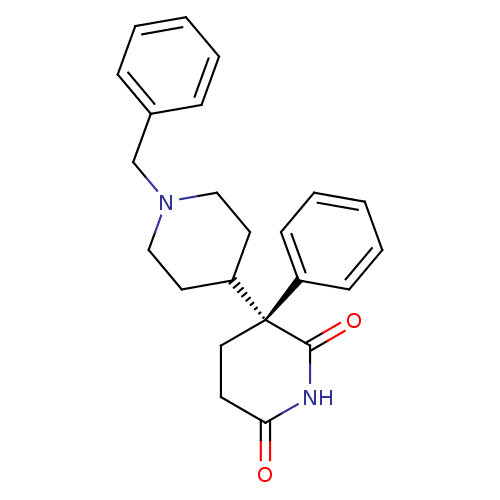
(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 240: 51-8 (1987)
BindingDB Entry DOI: 10.7270/Q2XW4H9Q |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(RAT) | BDBM50018550

(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50018550

(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 500-7 (1998)
BindingDB Entry DOI: 10.7270/Q2HX1B6S |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(RAT) | BDBM50018550

(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 500-7 (1998)
BindingDB Entry DOI: 10.7270/Q2HX1B6S |
More data for this
Ligand-Target Pair | |
Beta-arrestin-1
(RABBIT) | BDBM50018550

(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 240: 51-8 (1987)
BindingDB Entry DOI: 10.7270/Q2XW4H9Q |
More data for this
Ligand-Target Pair | |
Beta-arrestin-1
(RABBIT) | BDBM50018550

(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 571 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 240: 51-8 (1987)
BindingDB Entry DOI: 10.7270/Q2XW4H9Q |
More data for this
Ligand-Target Pair | |
Beta-arrestin-1
(RABBIT) | BDBM50018550

(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 7.28E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 240: 51-8 (1987)
BindingDB Entry DOI: 10.7270/Q2XW4H9Q |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50018550

(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 9.06E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 500-7 (1998)
BindingDB Entry DOI: 10.7270/Q2HX1B6S |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(RAT) | BDBM50018550

(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 500-7 (1998)
BindingDB Entry DOI: 10.7270/Q2HX1B6S |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M5
(Mus musculus) | BDBM50228206
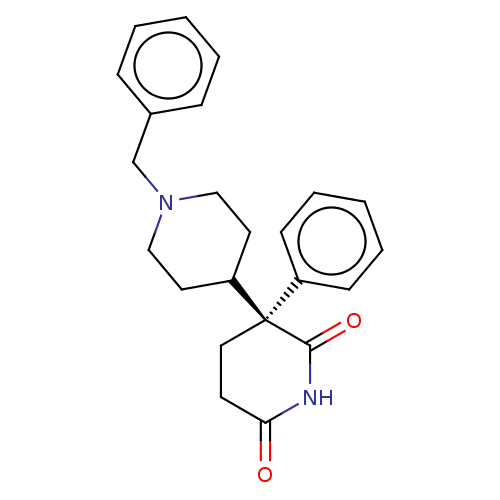
(Dexetimide | R-16470 [as hydrochloride])Show SMILES O=C1CC[C@@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Johns Hopkins Medical Institutions
Curated by ChEMBL
| Assay Description
Ability (10 ug/kg) to inhibit binding of [125I]iododexetimide to muscarinic receptor in mice |
J Med Chem 32: 1057-62 (1989)
BindingDB Entry DOI: 10.7270/Q29S1T87 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1/M2/M3/M4/M5
(RAT) | BDBM50018556
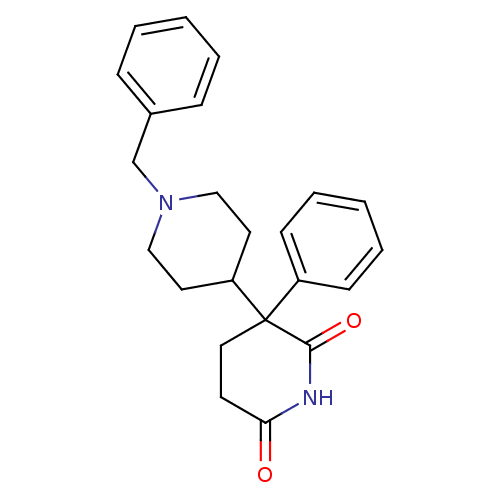
((+)-1'-benzyl-3-phenyl-3,4'-bipiperidine-2,6-dione...)Show SMILES O=C1CCC(C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Johns Hopkins Medical Institutions
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methylscopolamine from rat brain homogenate |
J Med Chem 32: 1057-62 (1989)
BindingDB Entry DOI: 10.7270/Q29S1T87 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M5
(Mus musculus) | BDBM50018556

((+)-1'-benzyl-3-phenyl-3,4'-bipiperidine-2,6-dione...)Show SMILES O=C1CCC(C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 8.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johns Hopkins Medical Institutions
Curated by ChEMBL
| Assay Description
Ability (10 ug/kg) to inhibit binding of [125I]iododexetimide to muscarinic receptor in mice |
J Med Chem 32: 1057-62 (1989)
BindingDB Entry DOI: 10.7270/Q29S1T87 |
More data for this
Ligand-Target Pair | |
C-X-C chemokine receptor type 3
(Homo sapiens (Human)) | BDBM50018556

((+)-1'-benzyl-3-phenyl-3,4'-bipiperidine-2,6-dione...)Show SMILES O=C1CCC(C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson PRD
Curated by ChEMBL
| Assay Description
Antagonist activity at human CXCR3 expressed in CHO cells assessed as inhibition of ITAC-stimulated [35S]GTPgammaS binding pretreated 30 mins before ... |
Bioorg Med Chem Lett 18: 5819-23 (2009)
Article DOI: 10.1016/j.bmcl.2008.07.115
BindingDB Entry DOI: 10.7270/Q2RV0NQB |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(Homo sapiens (Human)) | BDBM50018556

((+)-1'-benzyl-3-phenyl-3,4'-bipiperidine-2,6-dione...)Show SMILES O=C1CCC(C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson PRD
Curated by ChEMBL
| Assay Description
Binding affinity to human muscarinic M1 receptor |
Bioorg Med Chem Lett 18: 5819-23 (2009)
Article DOI: 10.1016/j.bmcl.2008.07.115
BindingDB Entry DOI: 10.7270/Q2RV0NQB |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50018556

((+)-1'-benzyl-3-phenyl-3,4'-bipiperidine-2,6-dione...)Show SMILES O=C1CCC(C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson PRD
Curated by ChEMBL
| Assay Description
Binding affinity to human muscarinic M2 receptor |
Bioorg Med Chem Lett 18: 5819-23 (2009)
Article DOI: 10.1016/j.bmcl.2008.07.115
BindingDB Entry DOI: 10.7270/Q2RV0NQB |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50018556

((+)-1'-benzyl-3-phenyl-3,4'-bipiperidine-2,6-dione...)Show SMILES O=C1CCC(C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson PRD
Curated by ChEMBL
| Assay Description
Binding affinity to human muscarinic M3 receptor |
Bioorg Med Chem Lett 18: 5819-23 (2009)
Article DOI: 10.1016/j.bmcl.2008.07.115
BindingDB Entry DOI: 10.7270/Q2RV0NQB |
More data for this
Ligand-Target Pair | |