 Found 5306 hits of ic50 data for polymerid = 1234
Found 5306 hits of ic50 data for polymerid = 1234 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50221967

((2S,3S)-3-amino-2-(4'-fluoro-biphenyl-4-yl)-4-((S)...)Show SMILES CN(C)C(=O)[C@H]([C@H](N)C(=O)N1CC[C@H](F)C1)c1ccc(cc1)-c1ccc(F)cc1 |r| Show InChI InChI=1S/C22H25F2N3O2/c1-26(2)21(28)19(20(25)22(29)27-12-11-18(24)13-27)16-5-3-14(4-6-16)15-7-9-17(23)10-8-15/h3-10,18-20H,11-13,25H2,1-2H3/t18-,19-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a |
Matrix Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of DPP4 |
Bioorg Med Chem 17: 1783-802 (2009)
Article DOI: 10.1016/j.bmc.2009.01.061
BindingDB Entry DOI: 10.7270/Q2VD70DJ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50612314

(CHEMBL5279548) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.0270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM205995

(US9255098, Ex. 1 | US9255098, Ex. 4)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nc4cc(F)ccc4s3)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C23H24FN7O2S/c1-3-4-10-30-19-20(27-22(30)29-9-5-6-15(25)12-29)28(2)23(33)31(21(19)32)13-18-26-16-11-14(24)7-8-17(16)34-18/h7-8,11,15H,5-6,9-10,12-13,25H2,1-2H3/t15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | 25 |
Chengdu Easton Pharmaceutical Co., Ltd.
US Patent
| Assay Description
Gly-Pro-7-amido-4-methylcoumarin can be hydrolyzed by dipeptidyl peptidase IV (DPP-IV) at room temperature, to generate 7-amido-4-methyl coumarin, wh... |
US Patent US9255098 (2016)
BindingDB Entry DOI: 10.7270/Q2FF3R62 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50221977

((2S,3S)-2-amino-3-(4'-fluoro-biphenyl-4-yl)-1-((S)...)Show SMILES C[C@H]([C@H](N)C(=O)N1CC[C@H](F)C1)c1ccc(cc1)-c1ccc(F)cc1 |r| Show InChI InChI=1S/C20H22F2N2O/c1-13(19(23)20(25)24-11-10-18(22)12-24)14-2-4-15(5-3-14)16-6-8-17(21)9-7-16/h2-9,13,18-19H,10-12,23H2,1H3/t13-,18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.0640 | n/a | n/a | n/a | n/a | n/a | n/a |
Matrix Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of DPP4 |
Bioorg Med Chem 17: 1783-802 (2009)
Article DOI: 10.1016/j.bmc.2009.01.061
BindingDB Entry DOI: 10.7270/Q2VD70DJ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM205995

(US9255098, Ex. 1 | US9255098, Ex. 4)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nc4cc(F)ccc4s3)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C23H24FN7O2S/c1-3-4-10-30-19-20(27-22(30)29-9-5-6-15(25)12-29)28(2)23(33)31(21(19)32)13-18-26-16-11-14(24)7-8-17(16)34-18/h7-8,11,15H,5-6,9-10,12-13,25H2,1-2H3/t15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | n/a | 25 |
Chengdu Easton Pharmaceutical Co., Ltd.
US Patent
| Assay Description
Gly-Pro-7-amido-4-methylcoumarin can be hydrolyzed by dipeptidyl peptidase IV (DPP-IV) at room temperature, to generate 7-amido-4-methyl coumarin, wh... |
US Patent US9255098 (2016)
BindingDB Entry DOI: 10.7270/Q2FF3R62 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM408611

(US10358449, Compound I-3)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nccnc3C#N)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C21H23N9O2/c1-3-4-10-29-17-18(26-20(29)28-9-5-6-14(23)12-28)27(2)21(32)30(19(17)31)13-16-15(11-22)24-7-8-25-16/h7-8,14H,5-6,9-10,12-13,23H2,1-2H3/t14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a |
Jiangsu Tasly Diyi Pharmaceutical Co, LTD.
US Patent
| Assay Description
DPP-IV could hydrolyze Gly-Pro-Aminoluciferin at room temperature to generate Aminoluciferin, which could produce �glow type� luminescent signals in ... |
US Patent US10358449 (2019)
BindingDB Entry DOI: 10.7270/Q29S1TCK |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50611802

(CHEMBL5269668) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11593
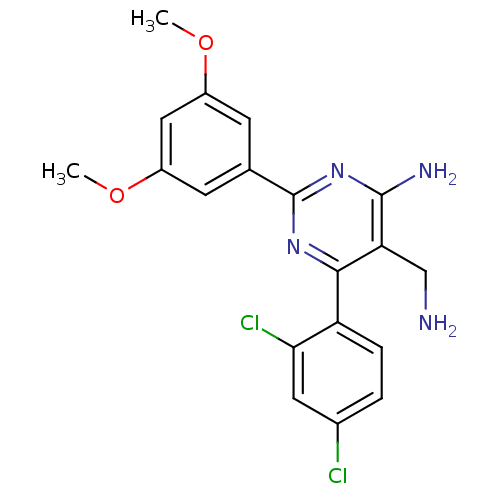
(5-(aminomethyl)-6-(2,4-dichlorophenyl)-2-(3,5-dime...)Show SMILES COc1cc(OC)cc(c1)-c1nc(N)c(CN)c(n1)-c1ccc(Cl)cc1Cl Show InChI InChI=1S/C19H18Cl2N4O2/c1-26-12-5-10(6-13(8-12)27-2)19-24-17(15(9-22)18(23)25-19)14-4-3-11(20)7-16(14)21/h3-8H,9,22H2,1-2H3,(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of dipeptidyl peptidase 4 (unknown origin) |
Citation and Details
Article DOI: 10.1007/s00044-012-0455-6
BindingDB Entry DOI: 10.7270/Q2HH6NZ4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50516930
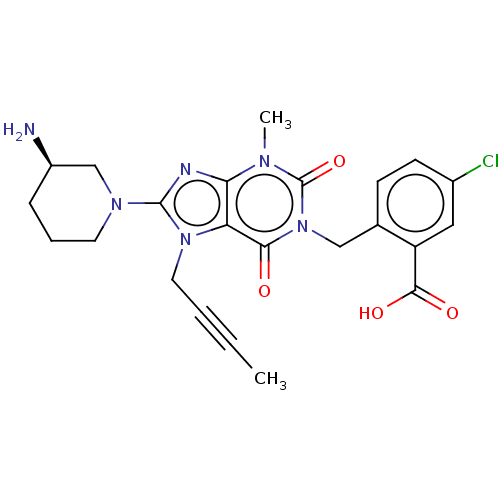
(CHEMBL4448186)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3ccc(Cl)cc3C(O)=O)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C23H25ClN6O4/c1-3-4-10-29-18-19(26-22(29)28-9-5-6-16(25)13-28)27(2)23(34)30(20(18)31)12-14-7-8-15(24)11-17(14)21(32)33/h7-8,11,16H,5-6,9-10,12-13,25H2,1-2H3,(H,32,33)/t16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Guangxi Medical University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human DPP4 expressed in baculovirus infected Sf9 insect cells using Gly-Pro-AMC as substrate preincubated for 15 mins follo... |
Eur J Med Chem 180: 509-523 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.045
BindingDB Entry DOI: 10.7270/Q2Q243MN |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50228403

((R)-8-(3-aminopiperidin-1-yl)-7-(but-2-ynyl)-3-met...)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nc(C)c4ccccc4n3)c(=O)c12)N1CCC[C@@H](N)C1 Show InChI InChI=1S/C25H28N8O2/c1-4-5-13-32-21-22(29-24(32)31-12-8-9-17(26)14-31)30(3)25(35)33(23(21)34)15-20-27-16(2)18-10-6-7-11-19(18)28-20/h6-7,10-11,17H,8-9,12-15,26H2,1-3H3/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 preincubated for 30 mins followed by Gly-Pro-AMC addition measured for 50 mins by continuous fluorescence assay |
ACS Med Chem Lett 7: 498-501 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00027
BindingDB Entry DOI: 10.7270/Q2CN75SM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11593

(5-(aminomethyl)-6-(2,4-dichlorophenyl)-2-(3,5-dime...)Show SMILES COc1cc(OC)cc(c1)-c1nc(N)c(CN)c(n1)-c1ccc(Cl)cc1Cl Show InChI InChI=1S/C19H18Cl2N4O2/c1-26-12-5-10(6-13(8-12)27-2)19-24-17(15(9-22)18(23)25-19)14-4-3-11(20)7-16(14)21/h3-8H,9,22H2,1-2H3,(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate University of the Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of DPP4 |
Bioorg Med Chem Lett 21: 6630-5 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.093
BindingDB Entry DOI: 10.7270/Q2DN45GR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50050513

((R)-1-((S)-2-amino-3-methylbutanoyl)pyrrolidin-2-y...)Show InChI InChI=1S/C9H19BN2O3/c1-6(2)8(11)9(13)12-5-3-4-7(12)10(14)15/h6-8,14-15H,3-5,11H2,1-2H3/t7-,8-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Tufts University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 507-10 (2007)
Article DOI: 10.1016/j.bmcl.2006.10.012
BindingDB Entry DOI: 10.7270/Q2T43SQH |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11593

(5-(aminomethyl)-6-(2,4-dichlorophenyl)-2-(3,5-dime...)Show SMILES COc1cc(OC)cc(c1)-c1nc(N)c(CN)c(n1)-c1ccc(Cl)cc1Cl Show InChI InChI=1S/C19H18Cl2N4O2/c1-26-12-5-10(6-13(8-12)27-2)19-24-17(15(9-22)18(23)25-19)14-4-3-11(20)7-16(14)21/h3-8H,9,22H2,1-2H3,(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
| Assay Description
DPP-IV inhibitors were measured for their ability to inhibit DPP-IV mediated cleavage of Ala-Pro-7-amido-4-trifluoromethylcoumarin in a fluorogenic a... |
Bioorg Med Chem Lett 14: 1491-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.01.019
BindingDB Entry DOI: 10.7270/Q22B8W7M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM205996
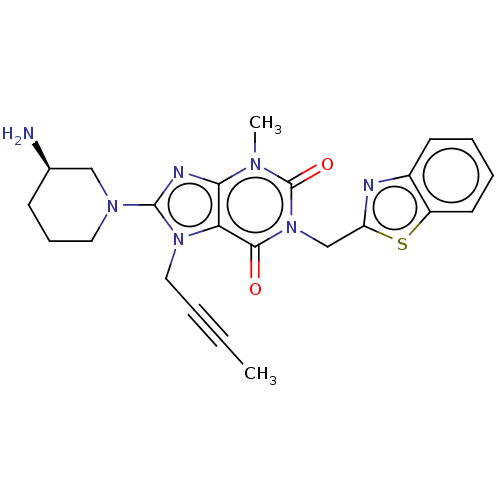
(US9255098, Ex. 2 | US9255098, Ex. 5)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nc4ccccc4s3)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C23H25N7O2S/c1-3-4-12-29-19-20(26-22(29)28-11-7-8-15(24)13-28)27(2)23(32)30(21(19)31)14-18-25-16-9-5-6-10-17(16)33-18/h5-6,9-10,15H,7-8,11-14,24H2,1-2H3/t15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | 25 |
Chengdu Easton Pharmaceutical Co., Ltd.
US Patent
| Assay Description
Gly-Pro-7-amido-4-methylcoumarin can be hydrolyzed by dipeptidyl peptidase IV (DPP-IV) at room temperature, to generate 7-amido-4-methyl coumarin, wh... |
US Patent US9255098 (2016)
BindingDB Entry DOI: 10.7270/Q2FF3R62 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50151798
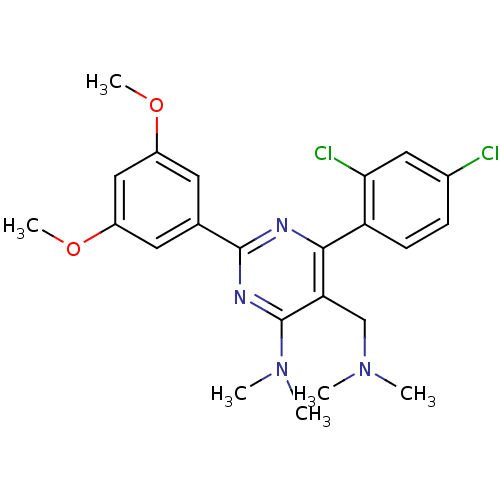
(CHEMBL186877 | [6-(2,4-Dichloro-phenyl)-2-(3,5-dim...)Show SMILES COc1cc(OC)cc(c1)-c1nc(N(C)C)c(CN(C)C)c(n1)-c1ccc(Cl)cc1Cl Show InChI InChI=1S/C23H26Cl2N4O2/c1-28(2)13-19-21(18-8-7-15(24)11-20(18)25)26-22(27-23(19)29(3)4)14-9-16(30-5)12-17(10-14)31-6/h7-12H,13H2,1-6H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Dipeptidyl peptidase IV |
Bioorg Med Chem Lett 14: 4759-62 (2004)
Article DOI: 10.1016/j.bmcl.2004.06.099
BindingDB Entry DOI: 10.7270/Q2XK8F1Q |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50228403

((R)-8-(3-aminopiperidin-1-yl)-7-(but-2-ynyl)-3-met...)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nc(C)c4ccccc4n3)c(=O)c12)N1CCC[C@@H](N)C1 Show InChI InChI=1S/C25H28N8O2/c1-4-5-13-32-21-22(29-24(32)31-12-8-9-17(26)14-31)30(3)25(35)33(23(21)34)15-20-27-16(2)18-10-6-7-11-19(18)28-20/h6-7,10-11,17H,8-9,12-15,26H2,1-3H3/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human C-terminal step-tagged DPP4 expressed using baculovirus system |
Bioorg Med Chem Lett 22: 1464-8 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.054
BindingDB Entry DOI: 10.7270/Q2SF2WN8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50170939
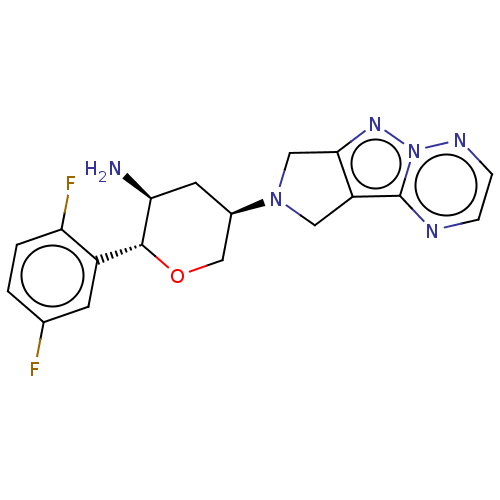
(CHEMBL3806052)Show SMILES N[C@H]1C[C@H](CO[C@@H]1c1cc(F)ccc1F)N1Cc2nn3nccnc3c2C1 |r| Show InChI InChI=1S/C18H18F2N6O/c19-10-1-2-14(20)12(5-10)17-15(21)6-11(9-27-17)25-7-13-16(8-25)24-26-18(13)22-3-4-23-26/h1-5,11,15,17H,6-9,21H2/t11-,15+,17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 26: 2622-6 (2016)
Article DOI: 10.1016/j.bmcl.2016.04.020
BindingDB Entry DOI: 10.7270/Q2Z321JP |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50607268
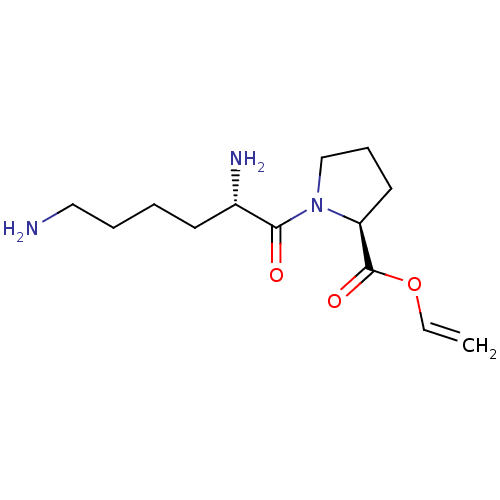
(CHEMBL5220657) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.129018
BindingDB Entry DOI: 10.7270/Q2251P86 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50166318

((S)-1-[(2S,4S)-4-(3-Chloro-4-cyano-phenylamino)-py...)Show SMILES Clc1cc(N[C@@H]2CN[C@@H](C2)C(=O)N2CCC[C@H]2C#N)ccc1C#N Show InChI InChI=1S/C17H18ClN5O/c18-15-6-12(4-3-11(15)8-19)22-13-7-16(21-10-13)17(24)23-5-1-2-14(23)9-20/h3-4,6,13-14,16,21-22H,1-2,5,7,10H2/t13-,14-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibitory concentration against human dipeptidyl-peptidase IV |
Bioorg Med Chem Lett 15: 2441-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.077
BindingDB Entry DOI: 10.7270/Q27P8XWJ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50166319

(4-[(3S,5S)-5-((S)-2-Cyano-pyrrolidine-1-carbonyl)-...)Show SMILES O=C([C@@H]1C[C@@H](CN1)Nc1ccc(C#N)c(c1)C#N)N1CCC[C@H]1C#N Show InChI InChI=1S/C18H18N6O/c19-8-12-3-4-14(6-13(12)9-20)23-15-7-17(22-11-15)18(25)24-5-1-2-16(24)10-21/h3-4,6,15-17,22-23H,1-2,5,7,11H2/t15-,16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibitory concentration against human dipeptidyl-peptidase IV |
Bioorg Med Chem Lett 15: 2441-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.077
BindingDB Entry DOI: 10.7270/Q27P8XWJ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50166319

(4-[(3S,5S)-5-((S)-2-Cyano-pyrrolidine-1-carbonyl)-...)Show SMILES O=C([C@@H]1C[C@@H](CN1)Nc1ccc(C#N)c(c1)C#N)N1CCC[C@H]1C#N Show InChI InChI=1S/C18H18N6O/c19-8-12-3-4-14(6-13(12)9-20)23-15-7-17(22-11-15)18(25)24-5-1-2-16(24)10-21/h3-4,6,15-17,22-23H,1-2,5,7,11H2/t15-,16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Matrix Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem 17: 1783-802 (2009)
Article DOI: 10.1016/j.bmc.2009.01.061
BindingDB Entry DOI: 10.7270/Q2VD70DJ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50170919
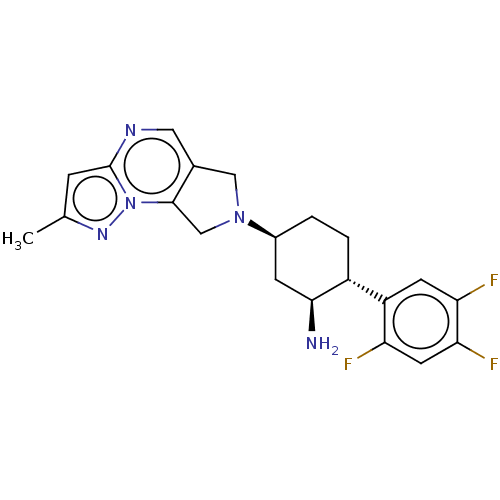
(CHEMBL3805966)Show SMILES Cc1cc2ncc3CN(Cc3n2n1)[C@H]1CC[C@@H]([C@@H](N)C1)c1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C21H22F3N5/c1-11-4-21-26-8-12-9-28(10-20(12)29(21)27-11)13-2-3-14(19(25)5-13)15-6-17(23)18(24)7-16(15)22/h4,6-8,13-14,19H,2-3,5,9-10,25H2,1H3/t13-,14+,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 26: 2622-6 (2016)
Article DOI: 10.1016/j.bmcl.2016.04.020
BindingDB Entry DOI: 10.7270/Q2Z321JP |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50607267
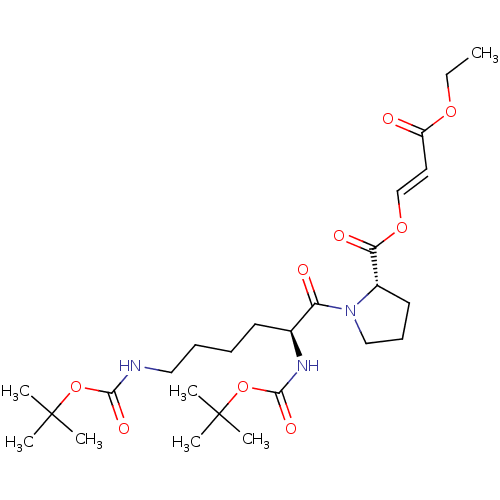
(CHEMBL5220613)Show SMILES CCOC(=O)\C=C\OC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCNC(=O)OC(C)(C)C)NC(=O)OC(C)(C)C |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.129018
BindingDB Entry DOI: 10.7270/Q2251P86 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM205996

(US9255098, Ex. 2 | US9255098, Ex. 5)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nc4ccccc4s3)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C23H25N7O2S/c1-3-4-12-29-19-20(26-22(29)28-11-7-8-15(24)13-28)27(2)23(32)30(21(19)31)14-18-25-16-9-5-6-10-17(16)33-18/h5-6,9-10,15H,7-8,11-14,24H2,1-2H3/t15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.150 | n/a | n/a | n/a | n/a | n/a | 25 |
Chengdu Easton Pharmaceutical Co., Ltd.
US Patent
| Assay Description
Gly-Pro-7-amido-4-methylcoumarin can be hydrolyzed by dipeptidyl peptidase IV (DPP-IV) at room temperature, to generate 7-amido-4-methyl coumarin, wh... |
US Patent US9255098 (2016)
BindingDB Entry DOI: 10.7270/Q2FF3R62 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50166318

((S)-1-[(2S,4S)-4-(3-Chloro-4-cyano-phenylamino)-py...)Show SMILES Clc1cc(N[C@@H]2CN[C@@H](C2)C(=O)N2CCC[C@H]2C#N)ccc1C#N Show InChI InChI=1S/C17H18ClN5O/c18-15-6-12(4-3-11(15)8-19)22-13-7-16(21-10-13)17(24)23-5-1-2-14(23)9-20/h3-4,6,13-14,16,21-22H,1-2,5,7,10H2/t13-,14-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a |
Matrix Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem 17: 1783-802 (2009)
Article DOI: 10.1016/j.bmc.2009.01.061
BindingDB Entry DOI: 10.7270/Q2VD70DJ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50607263
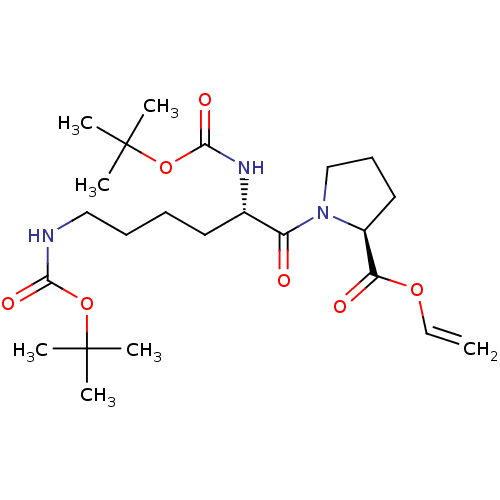
(CHEMBL5220594)Show SMILES CC(C)(C)OC(=O)NCCCC[C@H](NC(=O)OC(C)(C)C)C(=O)N1CCC[C@H]1C(=O)OC=C |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.129018
BindingDB Entry DOI: 10.7270/Q2251P86 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM136294

(US8853212, DDP-4 Inhibitor 10)Show SMILES N[C@H]1C[C@H](CSC1c1cc(F)ccc1F)N1Cc2n[nH]c(C(N)=O)c2C1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 0.170 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Merck Sharp & Dohme Corp
US Patent
| Assay Description
A continuous fluorometric assay is employed with the substrate Gly-Pro-AMC, which is cleaved by DPP-4 to release the fluorescent AMC leaving group. T... |
US Patent US8853212 (2014)
BindingDB Entry DOI: 10.7270/Q2C53JK4 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50516916

(CHEMBL4465482)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3cc(Cl)ccc3C(O)=O)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C23H25ClN6O4/c1-3-4-10-29-18-19(26-22(29)28-9-5-6-16(25)13-28)27(2)23(34)30(20(18)31)12-14-11-15(24)7-8-17(14)21(32)33/h7-8,11,16H,5-6,9-10,12-13,25H2,1-2H3,(H,32,33)/t16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Guangxi Medical University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human DPP4 expressed in baculovirus infected Sf9 insect cells using Gly-Pro-AMC as substrate preincubated for 15 mins follo... |
Eur J Med Chem 180: 509-523 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.045
BindingDB Entry DOI: 10.7270/Q2Q243MN |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50166308

((S)-1-[(2S,4S)-4-(4-Nitro-phenylamino)-pyrrolidine...)Show SMILES [O-][N+](=O)c1ccc(N[C@@H]2CN[C@@H](C2)C(=O)N2CCC[C@H]2C#N)cc1 Show InChI InChI=1S/C16H19N5O3/c17-9-14-2-1-7-20(14)16(22)15-8-12(10-18-15)19-11-3-5-13(6-4-11)21(23)24/h3-6,12,14-15,18-19H,1-2,7-8,10H2/t12-,14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibitory concentration against human dipeptidyl-peptidase IV |
Bioorg Med Chem Lett 15: 2441-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.077
BindingDB Entry DOI: 10.7270/Q27P8XWJ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50371255

(CHEMBL1203953)Show SMILES N[C@@H](CC(=O)N1CCn2c(nnc2C(F)(F)F)[C@H]1Cc1ccc(F)cc1)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C23H20F7N5O/c24-14-3-1-12(2-4-14)7-19-21-32-33-22(23(28,29)30)35(21)6-5-34(19)20(36)10-15(31)8-13-9-17(26)18(27)11-16(13)25/h1-4,9,11,15,19H,5-8,10,31H2/t15-,19-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of DPP4 |
J Med Chem 51: 589-602 (2008)
Article DOI: 10.1021/jm070330v
BindingDB Entry DOI: 10.7270/Q2B27W4M |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM479311

(US10894794, Comp. Code No. GLP-142)Show SMILES FC(F)(F)c1nnc2CN(CC(=O)Nc3cccc(c3)C#N)CCn12 Show InChI InChI=1S/C15H13F3N6O/c16-15(17,18)14-22-21-12-8-23(4-5-24(12)14)9-13(25)20-11-3-1-2-10(6-11)7-19/h1-3,6H,4-5,8-9H2,(H,20,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
King Abdulaziz University
US Patent
| Assay Description
itagliptin phosphate monohydrate and the different tested compounds were dissolved in dimethyl sulphoxide (DMSO) and diluted with tris buffer (pH 8.0... |
US Patent US10894794 (2021)
BindingDB Entry DOI: 10.7270/Q25T3PJ1 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50166320

((S)-1-[(2S,4S)-4-(4-Cyano-phenylamino)-pyrrolidine...)Show SMILES O=C([C@@H]1C[C@@H](CN1)Nc1ccc(cc1)C#N)N1CCC[C@H]1C#N Show InChI InChI=1S/C17H19N5O/c18-9-12-3-5-13(6-4-12)21-14-8-16(20-11-14)17(23)22-7-1-2-15(22)10-19/h3-6,14-16,20-21H,1-2,7-8,11H2/t14-,15-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibitory concentration against human dipeptidyl-peptidase IV |
Bioorg Med Chem Lett 15: 2441-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.077
BindingDB Entry DOI: 10.7270/Q27P8XWJ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM142088

(US11096924, DASH-inhibitors 2243 | US11559537, Com...)Show InChI InChI=1S/C7H15BN2O2S/c1-5(9)7(13)10-4-2-3-6(10)8(11)12/h5-6,11-12H,2-4,9H2,1H3/t5-,6-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
This assay is described in Bachovchin et al. Nature Chemical Biology 10, 656-663 (2014). Briefly, purified enzymes are coupled to Luminex microsphere... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q28W3J7Z |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50391561
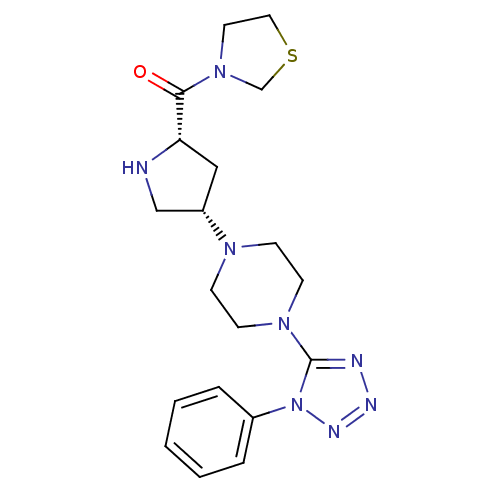
(CHEMBL2147704)Show SMILES O=C([C@@H]1C[C@@H](CN1)N1CCN(CC1)c1nnnn1-c1ccccc1)N1CCSC1 |r| Show InChI InChI=1S/C19H26N8OS/c28-18(26-10-11-29-14-26)17-12-16(13-20-17)24-6-8-25(9-7-24)19-21-22-23-27(19)15-4-2-1-3-5-15/h1-5,16-17,20H,6-14H2/t16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of DPP4 in human plasma using GLY-Pro-MCA as substrate after 60 mins by fluorescence assay |
Bioorg Med Chem 20: 5705-19 (2012)
Article DOI: 10.1016/j.bmc.2012.08.012
BindingDB Entry DOI: 10.7270/Q2JQ123C |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM205997
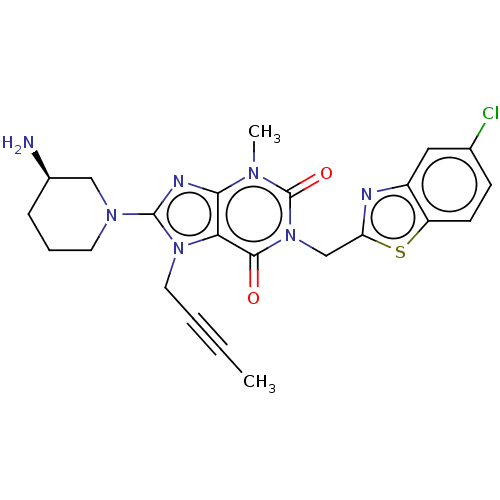
(US9255098, Ex. 3)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nc4cc(Cl)ccc4s3)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C23H24ClN7O2S/c1-3-4-10-30-19-20(27-22(30)29-9-5-6-15(25)12-29)28(2)23(33)31(21(19)32)13-18-26-16-11-14(24)7-8-17(16)34-18/h7-8,11,15H,5-6,9-10,12-13,25H2,1-2H3/t15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | 25 |
Chengdu Easton Pharmaceutical Co., Ltd.
US Patent
| Assay Description
Gly-Pro-7-amido-4-methylcoumarin can be hydrolyzed by dipeptidyl peptidase IV (DPP-IV) at room temperature, to generate 7-amido-4-methyl coumarin, wh... |
US Patent US9255098 (2016)
BindingDB Entry DOI: 10.7270/Q2FF3R62 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11589
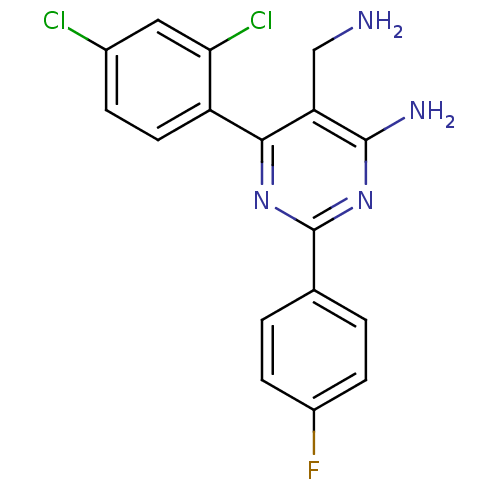
(1c-para-F | 5-(aminomethyl)-6-(2,4-dichlorophenyl)...)Show InChI InChI=1S/C17H13Cl2FN4/c18-10-3-6-12(14(19)7-10)15-13(8-21)16(22)24-17(23-15)9-1-4-11(20)5-2-9/h1-7H,8,21H2,(H2,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
| Assay Description
DPP-IV inhibitors were measured for their ability to inhibit DPP-IV mediated cleavage of Ala-Pro-7-amido-4-trifluoromethylcoumarin in a fluorogenic a... |
Bioorg Med Chem Lett 14: 1491-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.01.019
BindingDB Entry DOI: 10.7270/Q22B8W7M |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11588
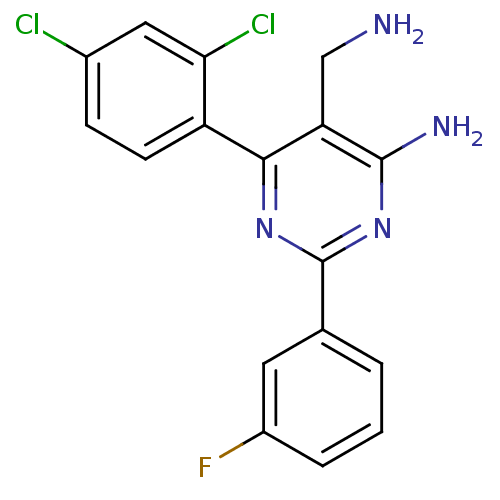
(1c-meta-F | 5-(aminomethyl)-6-(2,4-dichlorophenyl)...)Show InChI InChI=1S/C17H13Cl2FN4/c18-10-4-5-12(14(19)7-10)15-13(8-21)16(22)24-17(23-15)9-2-1-3-11(20)6-9/h1-7H,8,21H2,(H2,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
| Assay Description
DPP-IV inhibitors were measured for their ability to inhibit DPP-IV mediated cleavage of Ala-Pro-7-amido-4-trifluoromethylcoumarin in a fluorogenic a... |
Bioorg Med Chem Lett 14: 1491-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.01.019
BindingDB Entry DOI: 10.7270/Q22B8W7M |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM408607

(US10358449, Compound I-1)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3cccc(F)c3C#N)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C23H24FN7O2/c1-3-4-11-30-19-20(27-22(30)29-10-6-8-16(26)14-29)28(2)23(33)31(21(19)32)13-15-7-5-9-18(24)17(15)12-25/h5,7,9,16H,6,8,10-11,13-14,26H2,1-2H3/t16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
Jiangsu Tasly Diyi Pharmaceutical Co, LTD.
US Patent
| Assay Description
DPP-IV could hydrolyze Gly-Pro-Aminoluciferin at room temperature to generate Aminoluciferin, which could produce �glow type� luminescent signals in ... |
US Patent US10358449 (2019)
BindingDB Entry DOI: 10.7270/Q29S1TCK |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50374938
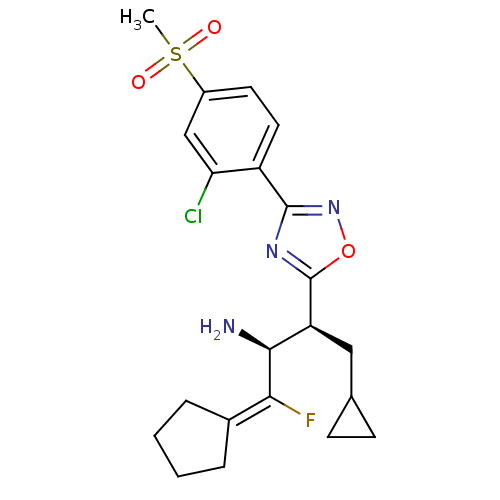
(CHEMBL402163)Show SMILES [#6]S(=O)(=O)c1ccc(-c2noc(n2)-[#6@@H](-[#6]-[#6]-2-[#6]-[#6]-2)-[#6@H](-[#7])-[#6](\F)=[#6]-2/[#6]-[#6]-[#6]-[#6]-2)c(Cl)c1 |r| Show InChI InChI=1S/C21H25ClFN3O3S/c1-30(27,28)14-8-9-15(17(22)11-14)20-25-21(29-26-20)16(10-12-6-7-12)19(24)18(23)13-4-2-3-5-13/h8-9,11-12,16,19H,2-7,10,24H2,1H3/t16-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck& Co. Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DPP4 expressed in insect cell |
Bioorg Med Chem Lett 18: 2409-13 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.050
BindingDB Entry DOI: 10.7270/Q2HD7WJ0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM479309

(US10894794, Comp. Code No. GLP-130)Show SMILES C[C@H](NC(=O)c1cccc(Cl)c1)C(=O)N1CCn2c(C1)nnc2C(F)(F)F |r| Show InChI InChI=1S/C16H15ClF3N5O2/c1-9(21-13(26)10-3-2-4-11(17)7-10)14(27)24-5-6-25-12(8-24)22-23-15(25)16(18,19)20/h2-4,7,9H,5-6,8H2,1H3,(H,21,26)/t9-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
King Abdulaziz University
US Patent
| Assay Description
itagliptin phosphate monohydrate and the different tested compounds were dissolved in dimethyl sulphoxide (DMSO) and diluted with tris buffer (pH 8.0... |
US Patent US10894794 (2021)
BindingDB Entry DOI: 10.7270/Q25T3PJ1 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50228403

((R)-8-(3-aminopiperidin-1-yl)-7-(but-2-ynyl)-3-met...)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nc(C)c4ccccc4n3)c(=O)c12)N1CCC[C@@H](N)C1 Show InChI InChI=1S/C25H28N8O2/c1-4-5-13-32-21-22(29-24(32)31-12-8-9-17(26)14-31)30(3)25(35)33(23(21)34)15-20-27-16(2)18-10-6-7-11-19(18)28-20/h6-7,10-11,17H,8-9,12-15,26H2,1-3H3/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
US Patent
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
Jiangsu Tasly Diyi Pharmaceutical Co, LTD.
US Patent
| Assay Description
DPP-IV could hydrolyze Gly-Pro-Aminoluciferin at room temperature to generate Aminoluciferin, which could produce �glow type� luminescent signals in ... |
US Patent US10358449 (2019)
BindingDB Entry DOI: 10.7270/Q29S1TCK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50516911

(CHEMBL4461587)Show SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3ccc(Br)cc3C(O)=O)c(=O)c12)N1CCC[C@@H](N)C1 |r| Show InChI InChI=1S/C23H25BrN6O4/c1-3-4-10-29-18-19(26-22(29)28-9-5-6-16(25)13-28)27(2)23(34)30(20(18)31)12-14-7-8-15(24)11-17(14)21(32)33/h7-8,11,16H,5-6,9-10,12-13,25H2,1-2H3,(H,32,33)/t16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a |
Guangxi Medical University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human DPP4 expressed in baculovirus infected Sf9 insect cells using Gly-Pro-AMC as substrate preincubated for 15 mins follo... |
Eur J Med Chem 180: 509-523 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.045
BindingDB Entry DOI: 10.7270/Q2Q243MN |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50170922

(CHEMBL3805629)Show SMILES Cc1nc2ncc3CN(Cc3n2n1)[C@H]1CC[C@@H]([C@@H](N)C1)c1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C20H21F3N6/c1-10-26-20-25-7-11-8-28(9-19(11)29(20)27-10)12-2-3-13(18(24)4-12)14-5-16(22)17(23)6-15(14)21/h5-7,12-13,18H,2-4,8-9,24H2,1H3/t12-,13+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 26: 2622-6 (2016)
Article DOI: 10.1016/j.bmcl.2016.04.020
BindingDB Entry DOI: 10.7270/Q2Z321JP |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50170923
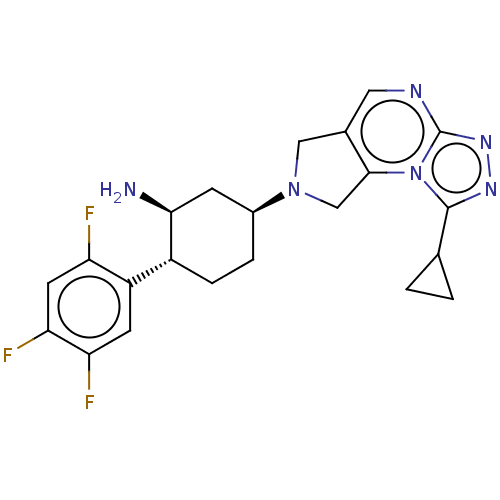
(CHEMBL3806041)Show SMILES N[C@H]1C[C@H](CC[C@@H]1c1cc(F)c(F)cc1F)N1Cc2cnc3nnc(C4CC4)n3c2C1 |r| Show InChI InChI=1S/C22H23F3N6/c23-16-7-18(25)17(24)6-15(16)14-4-3-13(5-19(14)26)30-9-12-8-27-22-29-28-21(11-1-2-11)31(22)20(12)10-30/h6-8,11,13-14,19H,1-5,9-10,26H2/t13-,14+,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 26: 2622-6 (2016)
Article DOI: 10.1016/j.bmcl.2016.04.020
BindingDB Entry DOI: 10.7270/Q2Z321JP |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50166315
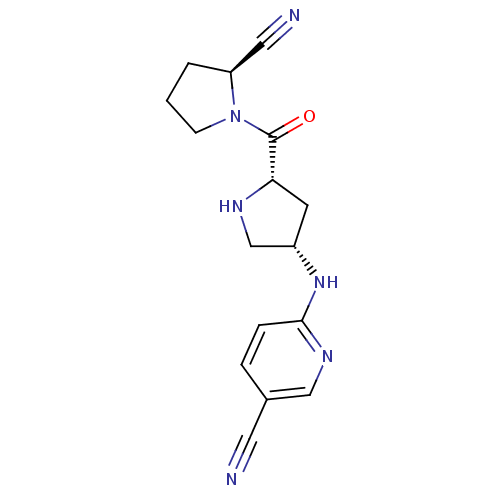
(6-((3S,5S)-5-((S)-2-cyanopyrrolidine-1-carbonyl)py...)Show SMILES O=C([C@@H]1C[C@@H](CN1)Nc1ccc(cn1)C#N)N1CCC[C@H]1C#N Show InChI InChI=1S/C16H18N6O/c17-7-11-3-4-15(20-9-11)21-12-6-14(19-10-12)16(23)22-5-1-2-13(22)8-18/h3-4,9,12-14,19H,1-2,5-6,10H2,(H,20,21)/t12-,13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibitory concentration against human dipeptidyl-peptidase IV |
Bioorg Med Chem Lett 15: 2441-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.077
BindingDB Entry DOI: 10.7270/Q27P8XWJ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50166315

(6-((3S,5S)-5-((S)-2-cyanopyrrolidine-1-carbonyl)py...)Show SMILES O=C([C@@H]1C[C@@H](CN1)Nc1ccc(cn1)C#N)N1CCC[C@H]1C#N Show InChI InChI=1S/C16H18N6O/c17-7-11-3-4-15(20-9-11)21-12-6-14(19-10-12)16(23)22-5-1-2-13(22)8-18/h3-4,9,12-14,19H,1-2,5-6,10H2,(H,20,21)/t12-,13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of DPP4 in human plasma by fluorescence assay |
Bioorg Med Chem 15: 641-55 (2006)
Article DOI: 10.1016/j.bmc.2006.10.059
BindingDB Entry DOI: 10.7270/Q21N81X1 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50607273

(CHEMBL5219939)Show SMILES CC(C)(C)OC(=O)NCC(=O)N1CCC[C@H]1C(=O)O\C=C\c1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.129018
BindingDB Entry DOI: 10.7270/Q2251P86 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50050525
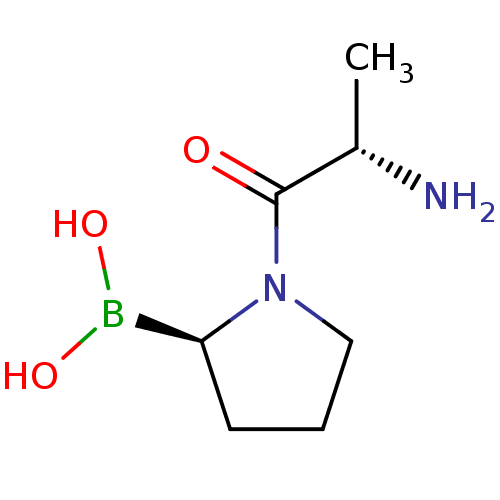
((R)-1-((S)-2-aminopropanoyl)pyrrolidin-2-ylboronic...)Show InChI InChI=1S/C7H15BN2O3/c1-5(9)7(11)10-4-2-3-6(10)8(12)13/h5-6,12-13H,2-4,9H2,1H3/t5-,6-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| US Patent
| n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | 2.0 | n/a |
Trustees of Tufts College
US Patent
| Assay Description
The inhibitor solution is prepared by dissolving 3-5 mg of inhibitor in pH 2 solution (0.01 N HCl), such that the concentration of the solution is eq... |
US Patent US8933056 (2015)
BindingDB Entry DOI: 10.7270/Q2ST7NJQ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50391559
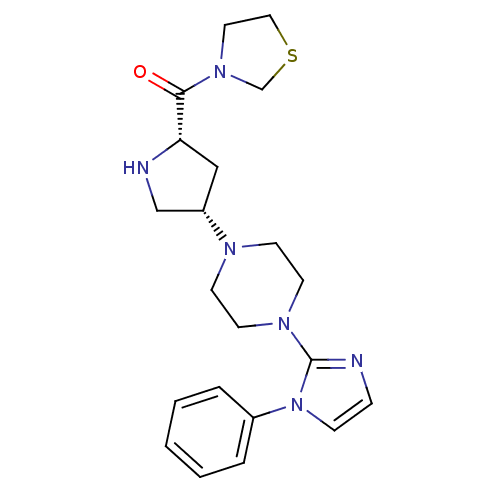
(CHEMBL2147703)Show SMILES O=C([C@@H]1C[C@@H](CN1)N1CCN(CC1)c1nccn1-c1ccccc1)N1CCSC1 |r| Show InChI InChI=1S/C21H28N6OS/c28-20(26-12-13-29-16-26)19-14-18(15-23-19)24-8-10-25(11-9-24)21-22-6-7-27(21)17-4-2-1-3-5-17/h1-7,18-19,23H,8-16H2/t18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of DPP4 in human plasma using GLY-Pro-MCA as substrate after 60 mins by fluorescence assay |
Bioorg Med Chem 20: 5705-19 (2012)
Article DOI: 10.1016/j.bmc.2012.08.012
BindingDB Entry DOI: 10.7270/Q2JQ123C |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50607269
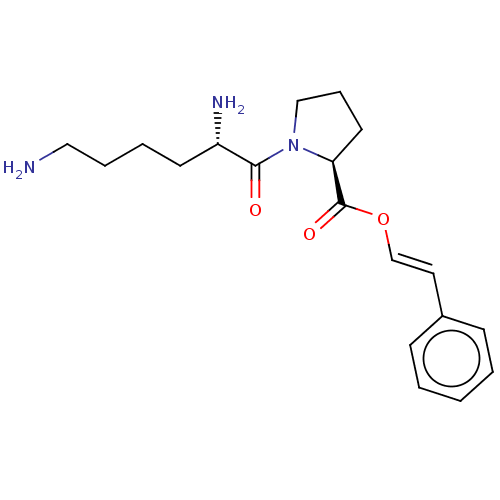
(CHEMBL5220750)Show SMILES NCCCC[C@H](N)C(=O)N1CCC[C@H]1C(=O)O\C=C\c1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.129018
BindingDB Entry DOI: 10.7270/Q2251P86 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data