 Found 59 hits Enz. Inhib. hit(s) with all data for entry = 50017432
Found 59 hits Enz. Inhib. hit(s) with all data for entry = 50017432 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cathepsin S
(Homo sapiens (Human)) | BDBM50182113
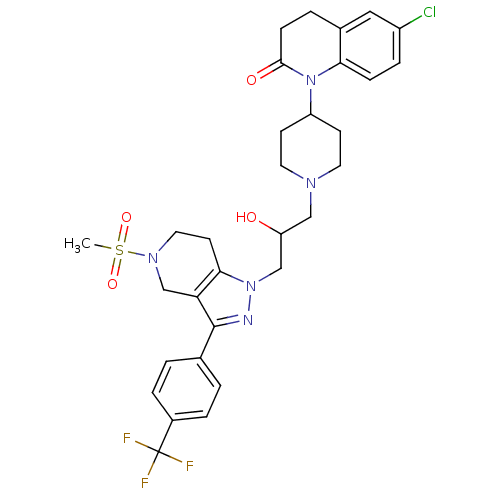
(6-chloro-1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)CCc2cc(Cl)ccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C31H35ClF3N5O4S/c1-45(43,44)38-15-12-28-26(19-38)30(20-2-5-22(6-3-20)31(33,34)35)36-39(28)18-25(41)17-37-13-10-24(11-14-37)40-27-8-7-23(32)16-21(27)4-9-29(40)42/h2-3,5-8,16,24-25,41H,4,9-15,17-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182103
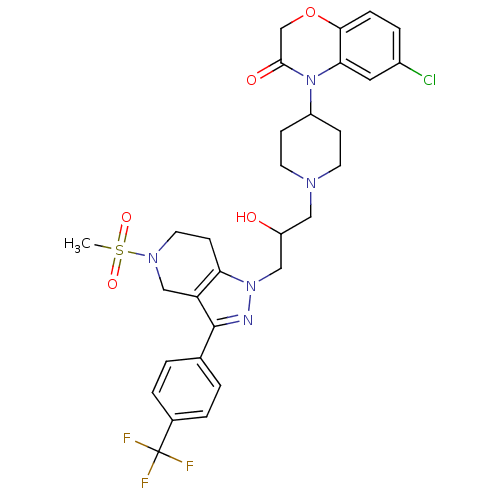
(6-chloro-4-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2ccc(Cl)cc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H33ClF3N5O5S/c1-45(42,43)37-13-10-25-24(17-37)29(19-2-4-20(5-3-19)30(32,33)34)35-38(25)16-23(40)15-36-11-8-22(9-12-36)39-26-14-21(31)6-7-27(26)44-18-28(39)41/h2-7,14,22-23,40H,8-13,15-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182117
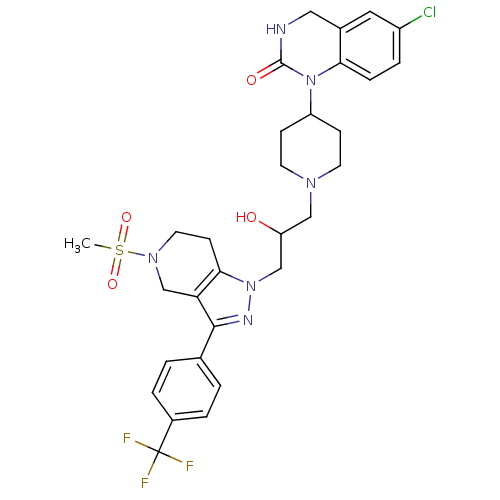
(6-chloro-1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)NCc2cc(Cl)ccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H34ClF3N6O4S/c1-45(43,44)38-13-10-27-25(18-38)28(19-2-4-21(5-3-19)30(32,33)34)36-39(27)17-24(41)16-37-11-8-23(9-12-37)40-26-7-6-22(31)14-20(26)15-35-29(40)42/h2-7,14,23-24,41H,8-13,15-18H2,1H3,(H,35,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182108

(1-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)CCc2cc(Cl)ccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C30H35BrClN5O4S/c1-42(40,41)35-15-12-28-26(19-35)30(20-2-5-22(31)6-3-20)33-36(28)18-25(38)17-34-13-10-24(11-14-34)37-27-8-7-23(32)16-21(27)4-9-29(37)39/h2-3,5-8,16,24-25,38H,4,9-15,17-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182109
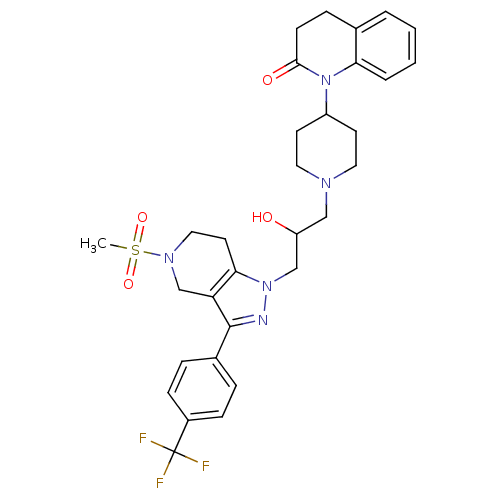
(1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)CCc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C31H36F3N5O4S/c1-44(42,43)37-17-14-28-26(20-37)30(22-6-9-23(10-7-22)31(32,33)34)35-38(28)19-25(40)18-36-15-12-24(13-16-36)39-27-5-3-2-4-21(27)8-11-29(39)41/h2-7,9-10,24-25,40H,8,11-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182110
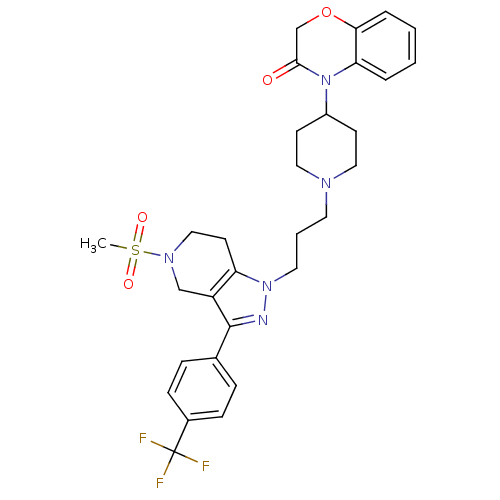
(4-(1-(3-(5-(methylsulfonyl)-3-(4-(trifluoromethyl)...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CCCN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H34F3N5O4S/c1-43(40,41)36-18-13-25-24(19-36)29(21-7-9-22(10-8-21)30(31,32)33)34-37(25)15-4-14-35-16-11-23(12-17-35)38-26-5-2-3-6-27(26)42-20-28(38)39/h2-3,5-10,23H,4,11-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182092
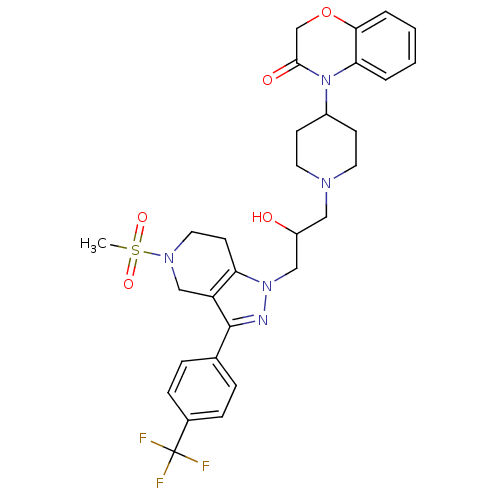
(4-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H34F3N5O5S/c1-44(41,42)36-15-12-25-24(18-36)29(20-6-8-21(9-7-20)30(31,32)33)34-37(25)17-23(39)16-35-13-10-22(11-14-35)38-26-4-2-3-5-27(26)43-19-28(38)40/h2-9,22-23,39H,10-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182101
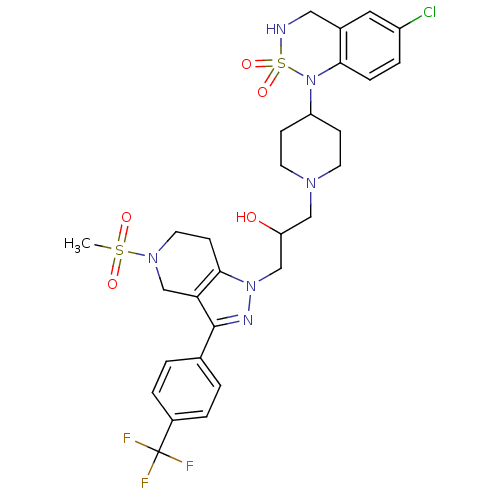
(1-[4-(6-Chloro-2,2-dioxo-3,4-dihydro-2H-2lambda*6*...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1c2ccc(Cl)cc2CNS1(=O)=O)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C29H34ClF3N6O5S2/c1-45(41,42)37-13-10-27-25(18-37)28(19-2-4-21(5-3-19)29(31,32)33)35-38(27)17-24(40)16-36-11-8-23(9-12-36)39-26-7-6-22(30)14-20(26)15-34-46(39,43)44/h2-7,14,23-24,34,40H,8-13,15-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182118
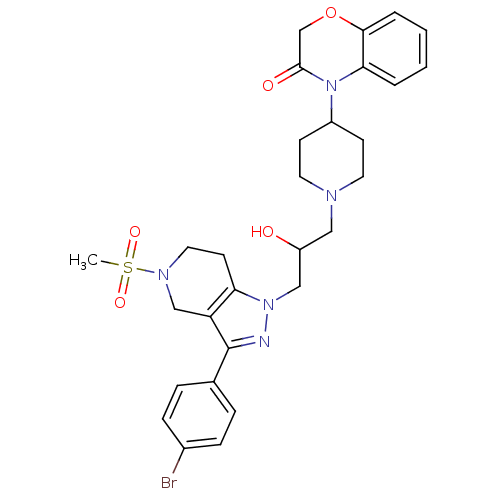
(4-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H34BrN5O5S/c1-41(38,39)33-15-12-25-24(18-33)29(20-6-8-21(30)9-7-20)31-34(25)17-23(36)16-32-13-10-22(11-14-32)35-26-4-2-3-5-27(26)40-19-28(35)37/h2-9,22-23,36H,10-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182100
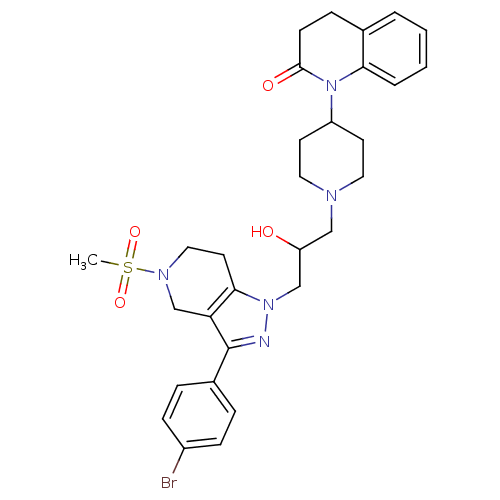
(1-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)CCc2ccccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C30H36BrN5O4S/c1-41(39,40)34-17-14-28-26(20-34)30(22-6-9-23(31)10-7-22)32-35(28)19-25(37)18-33-15-12-24(13-16-33)36-27-5-3-2-4-21(27)8-11-29(36)38/h2-7,9-10,24-25,37H,8,11-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182099
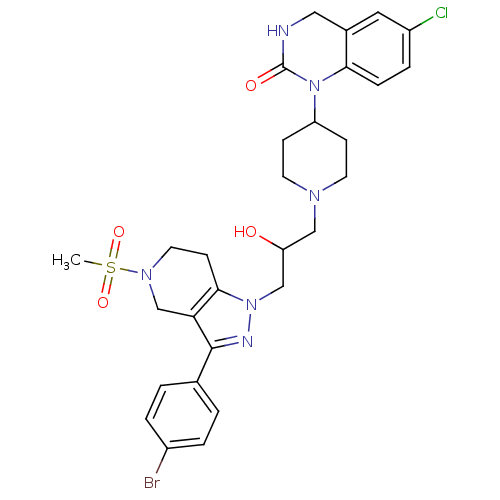
(1-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)NCc2cc(Cl)ccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H34BrClN6O4S/c1-42(40,41)35-13-10-27-25(18-35)28(19-2-4-21(30)5-3-19)33-36(27)17-24(38)16-34-11-8-23(9-12-34)37-26-7-6-22(31)14-20(26)15-32-29(37)39/h2-7,14,23-24,38H,8-13,15-18H2,1H3,(H,32,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182097
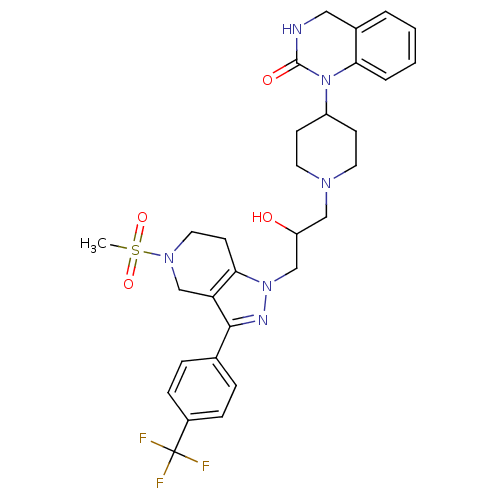
(1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)NCc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H35F3N6O4S/c1-44(42,43)37-15-12-27-25(19-37)28(20-6-8-22(9-7-20)30(31,32)33)35-38(27)18-24(40)17-36-13-10-23(11-14-36)39-26-5-3-2-4-21(26)16-34-29(39)41/h2-9,23-24,40H,10-19H2,1H3,(H,34,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182107
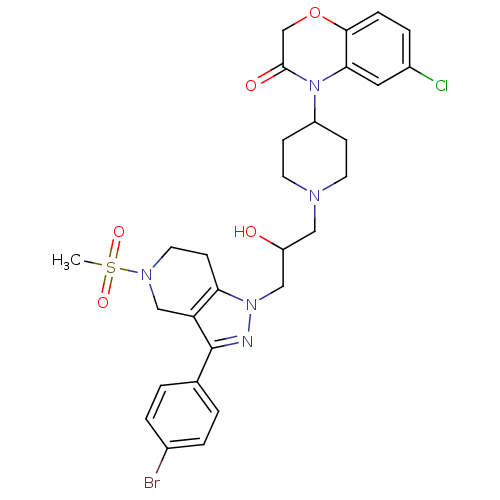
(4-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2ccc(Cl)cc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H33BrClN5O5S/c1-42(39,40)34-13-10-25-24(17-34)29(19-2-4-20(30)5-3-19)32-35(25)16-23(37)15-33-11-8-22(9-12-33)36-26-14-21(31)6-7-27(26)41-18-28(36)38/h2-7,14,22-23,37H,8-13,15-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182105
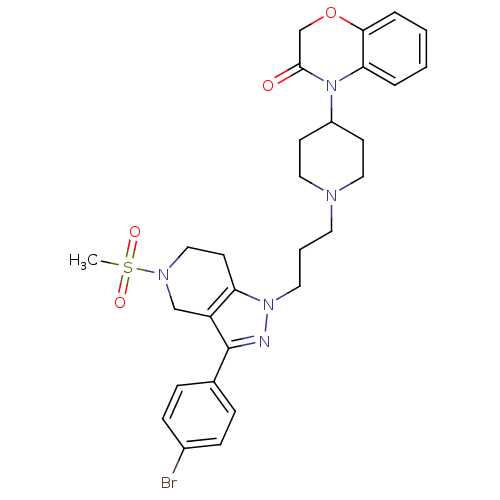
(4-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CCCN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H34BrN5O4S/c1-40(37,38)33-18-13-25-24(19-33)29(21-7-9-22(30)10-8-21)31-34(25)15-4-14-32-16-11-23(12-17-32)35-26-5-2-3-6-27(26)39-20-28(35)36/h2-3,5-10,23H,4,11-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182095
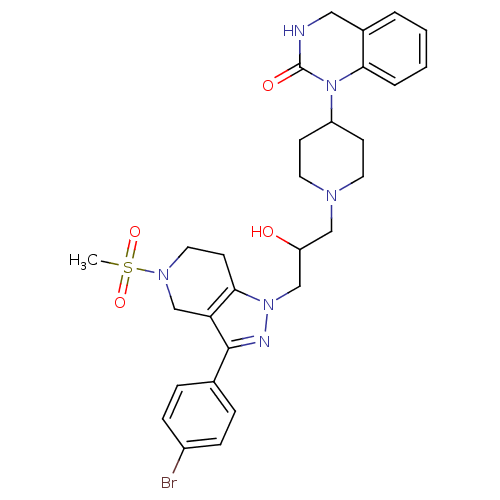
(1-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)NCc2ccccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H35BrN6O4S/c1-41(39,40)34-15-12-27-25(19-34)28(20-6-8-22(30)9-7-20)32-35(27)18-24(37)17-33-13-10-23(11-14-33)36-26-5-3-2-4-21(26)16-31-29(36)38/h2-9,23-24,37H,10-19H2,1H3,(H,31,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 65 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182104
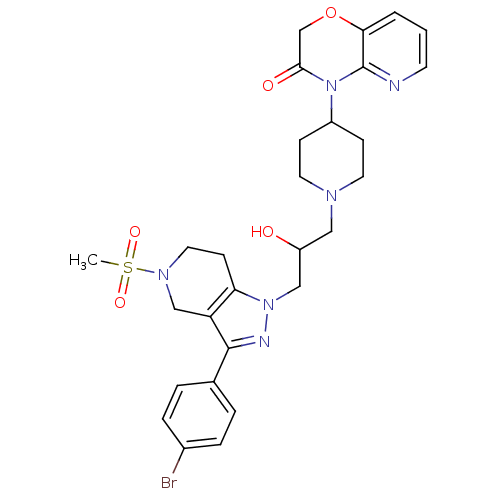
(4-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2cccnc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C28H33BrN6O5S/c1-41(38,39)33-14-10-24-23(17-33)27(19-4-6-20(29)7-5-19)31-34(24)16-22(36)15-32-12-8-21(9-13-32)35-26(37)18-40-25-3-2-11-30-28(25)35/h2-7,11,21-22,36H,8-10,12-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 68 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182089
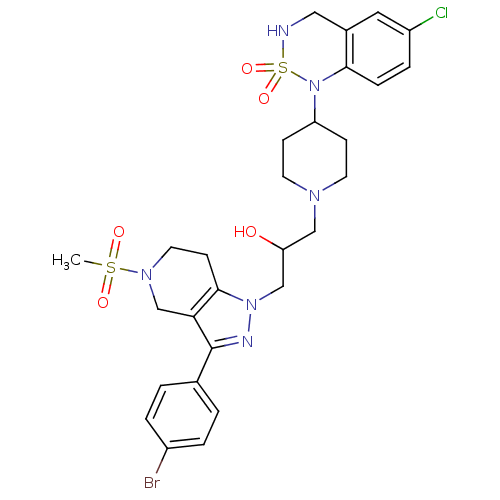
(1-[3-(4-Bromo-phenyl)-5-methanesulfonyl-4,5,6,7-te...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1c2ccc(Cl)cc2CNS1(=O)=O)-c1ccc(Br)cc1 Show InChI InChI=1S/C28H34BrClN6O5S2/c1-42(38,39)34-13-10-27-25(18-34)28(19-2-4-21(29)5-3-19)32-35(27)17-24(37)16-33-11-8-23(9-12-33)36-26-7-6-22(30)14-20(26)15-31-43(36,40)41/h2-7,14,23-24,31,37H,8-13,15-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 75 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182098
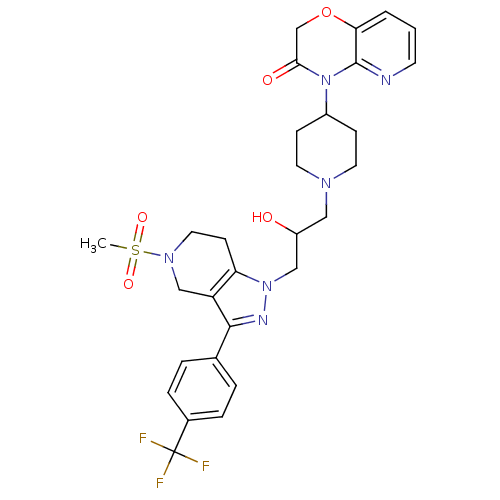
(4-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2cccnc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C29H33F3N6O5S/c1-44(41,42)36-14-10-24-23(17-36)27(19-4-6-20(7-5-19)29(30,31)32)34-37(24)16-22(39)15-35-12-8-21(9-13-35)38-26(40)18-43-25-3-2-11-33-28(25)38/h2-7,11,21-22,39H,8-10,12-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182093
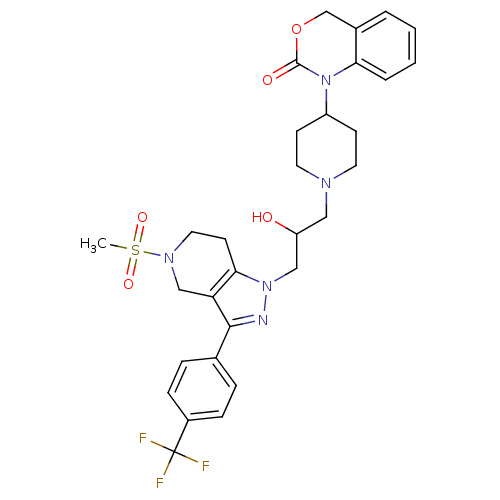
(1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)OCc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H34F3N5O5S/c1-44(41,42)36-15-12-27-25(18-36)28(20-6-8-22(9-7-20)30(31,32)33)34-37(27)17-24(39)16-35-13-10-23(11-14-35)38-26-5-3-2-4-21(26)19-43-29(38)40/h2-9,23-24,39H,10-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 88 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182102
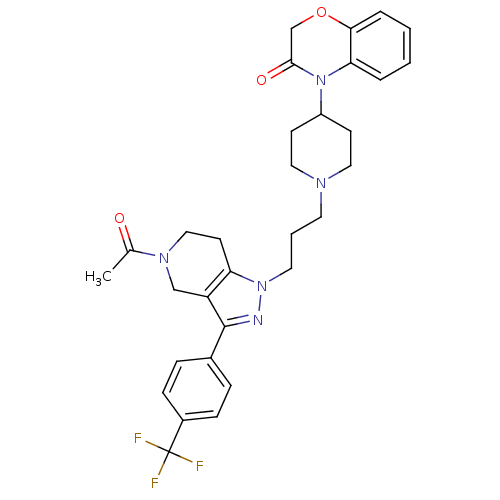
(4-(1-(3-(5-acetyl-3-(4-(trifluoromethyl)phenyl)-4,...)Show SMILES CC(=O)N1CCc2c(C1)c(nn2CCCN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C31H34F3N5O3/c1-21(40)37-18-13-26-25(19-37)30(22-7-9-23(10-8-22)31(32,33)34)35-38(26)15-4-14-36-16-11-24(12-17-36)39-27-5-2-3-6-28(27)42-20-29(39)41/h2-3,5-10,24H,4,11-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182087
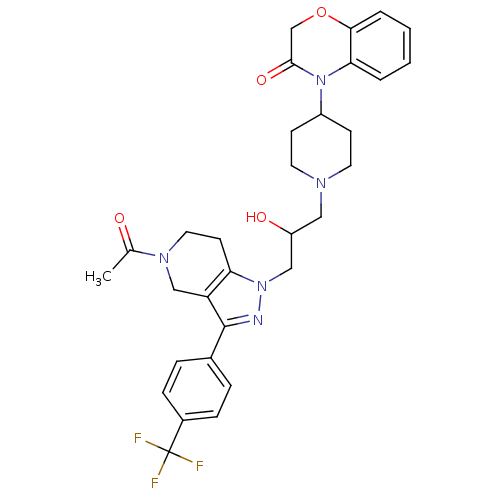
(4-(1-(3-(5-acetyl-3-(4-(trifluoromethyl)phenyl)-4,...)Show SMILES CC(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C31H34F3N5O4/c1-20(40)37-15-12-26-25(18-37)30(21-6-8-22(9-7-21)31(32,33)34)35-38(26)17-24(41)16-36-13-10-23(11-14-36)39-27-4-2-3-5-28(27)43-19-29(39)42/h2-9,23-24,41H,10-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182091
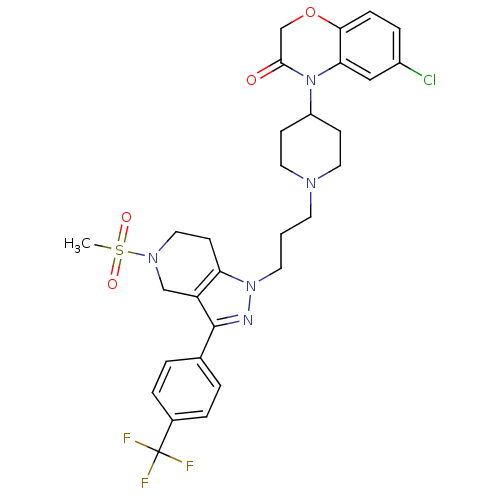
(6-chloro-4-(1-(3-(5-(methylsulfonyl)-3-(4-(trifluo...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CCCN1CCC(CC1)N1C(=O)COc2ccc(Cl)cc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H33ClF3N5O4S/c1-44(41,42)37-16-11-25-24(18-37)29(20-3-5-21(6-4-20)30(32,33)34)35-38(25)13-2-12-36-14-9-23(10-15-36)39-26-17-22(31)7-8-27(26)43-19-28(39)40/h3-8,17,23H,2,9-16,18-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182114
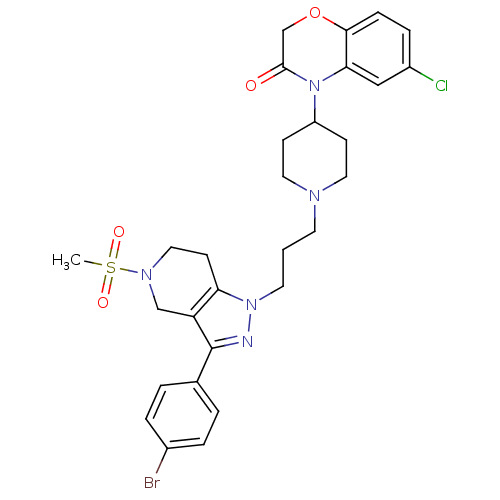
(4-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CCCN1CCC(CC1)N1C(=O)COc2ccc(Cl)cc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H33BrClN5O4S/c1-41(38,39)34-16-11-25-24(18-34)29(20-3-5-21(30)6-4-20)32-35(25)13-2-12-33-14-9-23(10-15-33)36-26-17-22(31)7-8-27(26)40-19-28(36)37/h3-8,17,23H,2,9-16,18-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182116
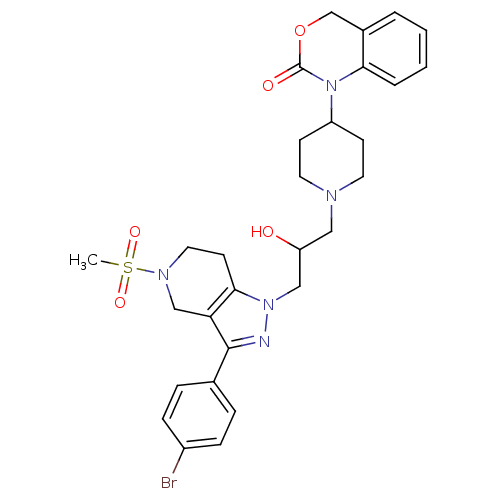
(1-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)OCc2ccccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H34BrN5O5S/c1-41(38,39)33-15-12-27-25(18-33)28(20-6-8-22(30)9-7-20)31-34(27)17-24(36)16-32-13-10-23(11-14-32)35-26-5-3-2-4-21(26)19-40-29(35)37/h2-9,23-24,36H,10-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 133 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182094
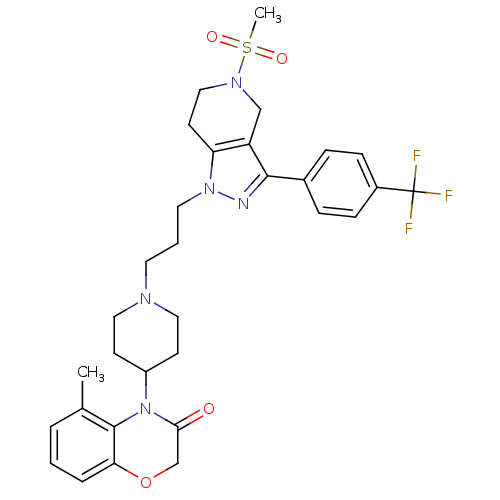
(5-methyl-4-(1-(3-(5-(methylsulfonyl)-3-(4-(trifluo...)Show SMILES Cc1cccc2OCC(=O)N(C3CCN(CCCn4nc(c5CN(CCc45)S(C)(=O)=O)-c4ccc(cc4)C(F)(F)F)CC3)c12 Show InChI InChI=1S/C31H36F3N5O4S/c1-21-5-3-6-27-30(21)39(28(40)20-43-27)24-11-16-36(17-12-24)14-4-15-38-26-13-18-37(44(2,41)42)19-25(26)29(35-38)22-7-9-23(10-8-22)31(32,33)34/h3,5-10,24H,4,11-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 145 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182115
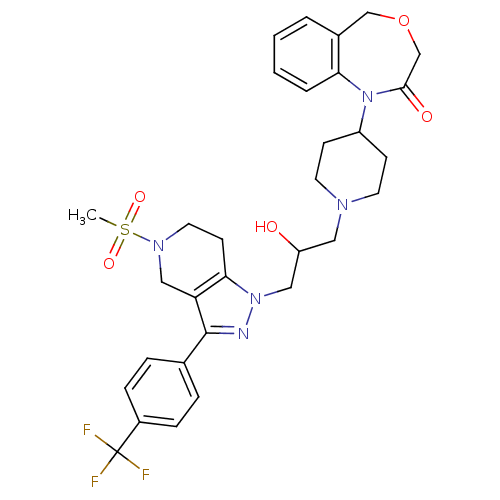
(1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1c2ccccc2COCC1=O)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C31H36F3N5O5S/c1-45(42,43)37-15-12-28-26(18-37)30(21-6-8-23(9-7-21)31(32,33)34)35-38(28)17-25(40)16-36-13-10-24(11-14-36)39-27-5-3-2-4-22(27)19-44-20-29(39)41/h2-9,24-25,40H,10-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 163 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182106
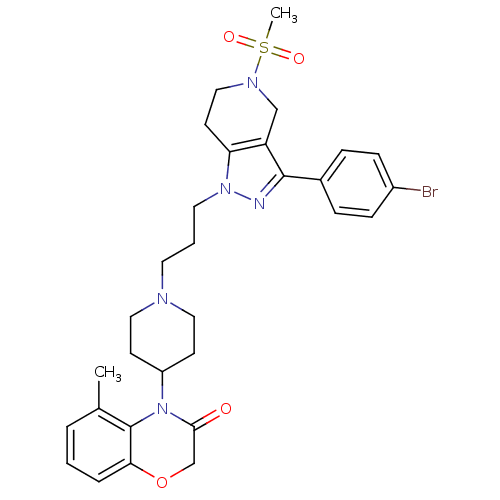
(4-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES Cc1cccc2OCC(=O)N(C3CCN(CCCn4nc(c5CN(CCc45)S(C)(=O)=O)-c4ccc(Br)cc4)CC3)c12 Show InChI InChI=1S/C30H36BrN5O4S/c1-21-5-3-6-27-30(21)36(28(37)20-40-27)24-11-16-33(17-12-24)14-4-15-35-26-13-18-34(41(2,38)39)19-25(26)29(32-35)22-7-9-23(31)10-8-22/h3,5-10,24H,4,11-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182120

(1-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,6,7-te...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1CCCc2ccccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C30H38BrN5O3S/c1-40(38,39)34-18-14-29-27(21-34)30(23-8-10-24(31)11-9-23)32-36(29)20-26(37)19-33-16-12-25(13-17-33)35-15-4-6-22-5-2-3-7-28(22)35/h2-3,5,7-11,25-26,37H,4,6,12-21H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 322 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182092

(4-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H34F3N5O5S/c1-44(41,42)36-15-12-25-24(18-36)29(20-6-8-21(9-7-20)30(31,32)33)34-37(25)17-23(39)16-35-13-10-22(11-14-35)38-26-4-2-3-5-27(26)43-19-28(38)40/h2-9,22-23,39H,10-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 410 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182108

(1-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)CCc2cc(Cl)ccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C30H35BrClN5O4S/c1-42(40,41)35-15-12-28-26(19-35)30(20-2-5-22(31)6-3-20)33-36(28)18-25(38)17-34-13-10-24(11-14-34)37-27-8-7-23(32)16-21(27)4-9-29(37)39/h2-3,5-8,16,24-25,38H,4,9-15,17-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 750 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182102

(4-(1-(3-(5-acetyl-3-(4-(trifluoromethyl)phenyl)-4,...)Show SMILES CC(=O)N1CCc2c(C1)c(nn2CCCN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C31H34F3N5O3/c1-21(40)37-18-13-26-25(19-37)30(22-7-9-23(10-8-22)31(32,33)34)35-38(26)15-4-14-36-16-11-24(12-17-36)39-27-5-2-3-6-28(27)42-20-29(39)41/h2-3,5-10,24H,4,11-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 810 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182090

(CHEMBL382419 | tert-butyl 1-(2-hydroxy-3-(4-(3-oxo...)Show SMILES CC(C)(C)OC(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C34H40F3N5O5/c1-33(2,3)47-32(45)40-17-14-27-26(20-40)31(22-8-10-23(11-9-22)34(35,36)37)38-41(27)19-25(43)18-39-15-12-24(13-16-39)42-28-6-4-5-7-29(28)46-21-30(42)44/h4-11,24-25,43H,12-21H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 826 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182112
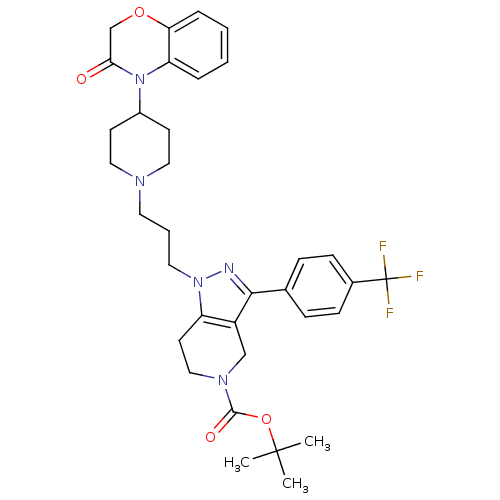
(CHEMBL206594 | tert-butyl 1-(3-(4-(3-oxo-2,3-dihyd...)Show SMILES CC(C)(C)OC(=O)N1CCc2c(C1)c(nn2CCCN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C34H40F3N5O4/c1-33(2,3)46-32(44)40-20-15-27-26(21-40)31(23-9-11-24(12-10-23)34(35,36)37)38-41(27)17-6-16-39-18-13-25(14-19-39)42-28-7-4-5-8-29(28)45-22-30(42)43/h4-5,7-12,25H,6,13-22H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182104

(4-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2cccnc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C28H33BrN6O5S/c1-41(38,39)33-14-10-24-23(17-33)27(19-4-6-20(29)7-5-19)31-34(24)16-22(36)15-32-12-8-21(9-13-32)35-26(37)18-40-25-3-2-11-30-28(25)35/h2-7,11,21-22,36H,8-10,12-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182115

(1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1c2ccccc2COCC1=O)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C31H36F3N5O5S/c1-45(42,43)37-15-12-28-26(18-37)30(21-6-8-23(9-7-21)31(32,33)34)35-38(28)17-25(40)16-36-13-10-24(11-14-36)39-27-5-3-2-4-22(27)19-44-20-29(39)41/h2-9,24-25,40H,10-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 930 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182110

(4-(1-(3-(5-(methylsulfonyl)-3-(4-(trifluoromethyl)...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CCCN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H34F3N5O4S/c1-43(40,41)36-18-13-25-24(19-36)29(21-7-9-22(10-8-21)30(31,32)33)34-37(25)15-4-14-35-16-11-23(12-17-35)38-26-5-2-3-6-27(26)42-20-28(38)39/h2-3,5-10,23H,4,11-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 950 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182113

(6-chloro-1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)CCc2cc(Cl)ccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C31H35ClF3N5O4S/c1-45(43,44)38-15-12-28-26(19-38)30(20-2-5-22(6-3-20)31(33,34)35)36-39(28)18-25(41)17-37-13-10-24(11-14-37)40-27-8-7-23(32)16-21(27)4-9-29(40)42/h2-3,5-8,16,24-25,41H,4,9-15,17-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 980 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182087

(4-(1-(3-(5-acetyl-3-(4-(trifluoromethyl)phenyl)-4,...)Show SMILES CC(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C31H34F3N5O4/c1-20(40)37-15-12-26-25(18-37)30(21-6-8-22(9-7-21)31(32,33)34)35-38(26)17-24(41)16-36-13-10-23(11-14-36)39-27-4-2-3-5-28(27)43-19-29(39)42/h2-9,23-24,41H,10-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182095

(1-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)NCc2ccccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H35BrN6O4S/c1-41(39,40)34-15-12-27-25(19-34)28(20-6-8-22(30)9-7-20)32-35(27)18-24(37)17-33-13-10-23(11-14-33)36-26-5-3-2-4-21(26)16-31-29(36)38/h2-9,23-24,37H,10-19H2,1H3,(H,31,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182101

(1-[4-(6-Chloro-2,2-dioxo-3,4-dihydro-2H-2lambda*6*...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1c2ccc(Cl)cc2CNS1(=O)=O)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C29H34ClF3N6O5S2/c1-45(41,42)37-13-10-27-25(18-37)28(19-2-4-21(5-3-19)29(31,32)33)35-38(27)17-24(40)16-36-11-8-23(9-12-36)39-26-7-6-22(30)14-20(26)15-34-46(39,43)44/h2-7,14,23-24,34,40H,8-13,15-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182098

(4-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2cccnc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C29H33F3N6O5S/c1-44(41,42)36-14-10-24-23(17-36)27(19-4-6-20(7-5-19)29(30,31)32)34-37(24)16-22(39)15-35-12-8-21(9-13-35)38-26(40)18-43-25-3-2-11-33-28(25)38/h2-7,11,21-22,39H,8-10,12-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182103

(6-chloro-4-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)COc2ccc(Cl)cc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H33ClF3N5O5S/c1-45(42,43)37-13-10-25-24(17-37)29(19-2-4-20(5-3-19)30(32,33)34)35-38(25)16-23(40)15-36-11-8-22(9-12-36)39-26-14-21(31)6-7-27(26)44-18-28(39)41/h2-7,14,22-23,40H,8-13,15-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182097

(1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)NCc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H35F3N6O4S/c1-44(42,43)37-15-12-27-25(19-37)28(20-6-8-22(9-7-20)30(31,32)33)35-38(27)18-24(40)17-36-13-10-23(11-14-36)39-26-5-3-2-4-21(26)16-34-29(39)41/h2-9,23-24,40H,10-19H2,1H3,(H,34,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182093

(1-(1-(2-hydroxy-3-(5-(methylsulfonyl)-3-(4-(triflu...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)OCc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H34F3N5O5S/c1-44(41,42)36-15-12-27-25(18-36)28(20-6-8-22(9-7-20)30(31,32)33)34-37(27)17-24(39)16-35-13-10-23(11-14-35)38-26-5-3-2-4-21(26)19-43-29(38)40/h2-9,23-24,39H,10-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182099

(1-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1C(=O)NCc2cc(Cl)ccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H34BrClN6O4S/c1-42(40,41)35-13-10-27-25(18-35)28(19-2-4-21(30)5-3-19)33-36(27)17-24(38)16-34-11-8-23(9-12-34)37-26-7-6-22(31)14-20(26)15-32-29(37)39/h2-7,14,23-24,38H,8-13,15-18H2,1H3,(H,32,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182106

(4-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES Cc1cccc2OCC(=O)N(C3CCN(CCCn4nc(c5CN(CCc45)S(C)(=O)=O)-c4ccc(Br)cc4)CC3)c12 Show InChI InChI=1S/C30H36BrN5O4S/c1-21-5-3-6-27-30(21)36(28(37)20-40-27)24-11-16-33(17-12-24)14-4-15-35-26-13-18-34(41(2,38)39)19-25(26)29(32-35)22-7-9-23(31)10-8-22/h3,5-10,24H,4,11-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
HLA class II histocompatibility antigen gamma chain
(Homo sapiens (Human)) | BDBM50182105

(4-(1-(3-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CCCN1CCC(CC1)N1C(=O)COc2ccccc12)-c1ccc(Br)cc1 Show InChI InChI=1S/C29H34BrN5O4S/c1-40(37,38)33-18-13-25-24(19-33)29(21-7-9-22(30)10-8-21)31-34(25)15-4-14-32-16-11-23(12-17-32)35-26-5-2-3-6-27(26)39-20-28(35)36/h2-3,5-10,23H,4,11-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of MHC2 invariant chain |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182088

(1-(5-(methylsulfonyl)-3-(4-(trifluoromethyl)phenyl...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCN(CC1)c1ccnc2ccccc12)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C30H33F3N6O3S/c1-43(41,42)38-13-11-28-25(20-38)29(21-6-8-22(9-7-21)30(31,32)33)35-39(28)19-23(40)18-36-14-16-37(17-15-36)27-10-12-34-26-5-3-2-4-24(26)27/h2-10,12,23,40H,11,13-20H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182111
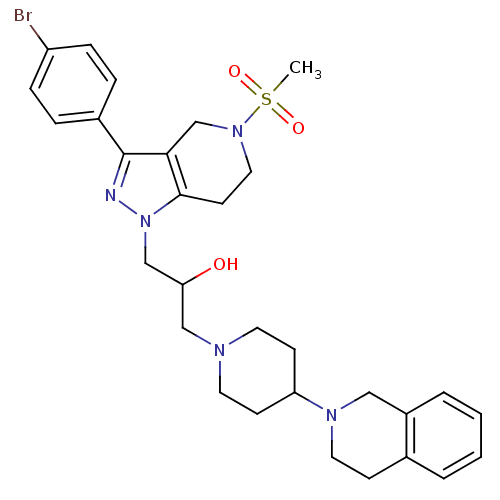
(1-(3-(4-bromophenyl)-5-(methylsulfonyl)-4,5,6,7-te...)Show SMILES CS(=O)(=O)N1CCc2c(C1)c(nn2CC(O)CN1CCC(CC1)N1CCc2ccccc2C1)-c1ccc(Br)cc1 Show InChI InChI=1S/C30H38BrN5O3S/c1-40(38,39)35-17-13-29-28(21-35)30(23-6-8-25(31)9-7-23)32-36(29)20-27(37)19-33-14-11-26(12-15-33)34-16-10-22-4-2-3-5-24(22)18-34/h2-9,26-27,37H,10-21H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.78E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50182119
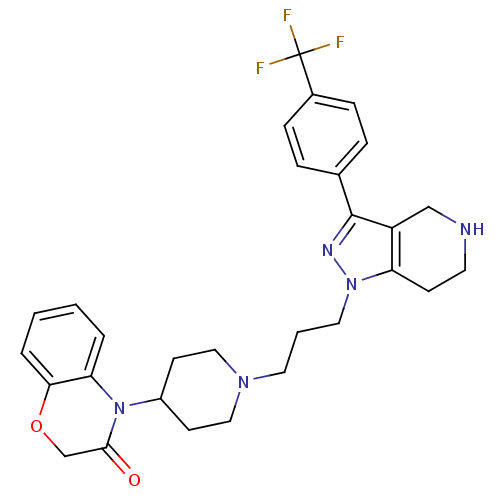
(4-(1-(3-(3-(4-(trifluoromethyl)phenyl)-4,5,6,7-tet...)Show SMILES FC(F)(F)c1ccc(cc1)-c1nn(CCCN2CCC(CC2)N2C(=O)COc3ccccc23)c2CCNCc12 Show InChI InChI=1S/C29H32F3N5O2/c30-29(31,32)21-8-6-20(7-9-21)28-23-18-33-13-10-24(23)36(34-28)15-3-14-35-16-11-22(12-17-35)37-25-4-1-2-5-26(25)39-19-27(37)38/h1-2,4-9,22,33H,3,10-19H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin S |
Bioorg Med Chem Lett 16: 2209-12 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.038
BindingDB Entry DOI: 10.7270/Q2K35T7W |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data