 Found 82 hits Enz. Inhib. hit(s) with all data for entry = 50047871
Found 82 hits Enz. Inhib. hit(s) with all data for entry = 50047871 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194718
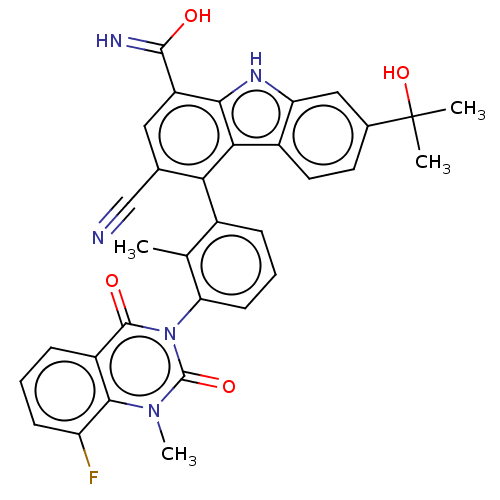
(CHEMBL3899411)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(cc(C(O)=N)c2[nH]c3cc(ccc3c12)C(C)(C)O)C#N |(-2.03,-1.34,;-1.6,.13,;-.1,.5,;.33,1.98,;-.74,3.09,;-2.23,2.73,;-2.66,1.25,;-4.16,.88,;-4.59,-.6,;-3.52,-1.71,;-6.08,-.97,;-6.51,-2.44,;-7.15,.15,;-8.65,-.22,;-9.08,-1.7,;-9.71,.89,;-9.28,2.37,;-7.78,2.74,;-6.72,1.63,;-5.22,1.99,;-4.79,3.47,;.96,-.61,;.53,-2.09,;1.6,-3.2,;3.09,-2.84,;4.16,-3.95,;5.66,-3.58,;3.73,-5.43,;3.52,-1.36,;5.06,-1.52,;6.34,-.67,;7.62,.19,;7.51,1.73,;6.13,2.41,;4.85,1.55,;4.95,.01,;2.46,-.24,;8.79,2.58,;7.94,3.86,;9.65,1.3,;10.07,3.44,;-.96,-2.46,;-2.46,-2.82,)| Show InChI InChI=1S/C33H26FN5O4/c1-16-19(7-6-10-25(16)39-31(41)21-8-5-9-23(34)29(21)38(4)32(39)42)26-17(15-35)13-22(30(36)40)28-27(26)20-12-11-18(33(2,3)43)14-24(20)37-28/h5-14,37,43H,1-4H3,(H2,36,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194722
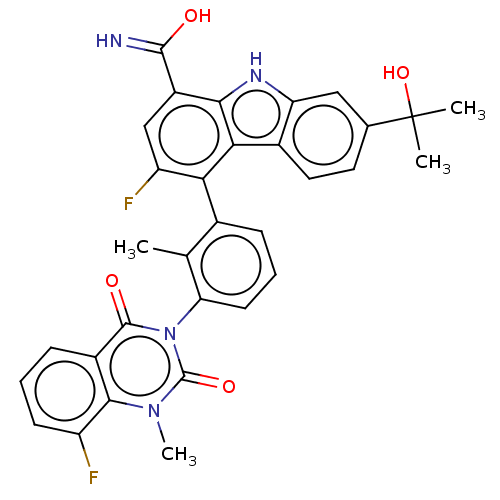
(CHEMBL3896019)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(F)cc(C(O)=N)c2[nH]c3cc(ccc3c12)C(C)(C)O |(-2.05,-1.45,;-1.63,.03,;-.14,.41,;.29,1.89,;-.78,2.99,;-2.28,2.62,;-2.7,1.14,;-4.2,.77,;-4.62,-.71,;-3.55,-1.82,;-6.11,-1.09,;-6.54,-2.57,;-7.18,.02,;-8.68,-.35,;-9.1,-1.84,;-9.75,.75,;-9.32,2.23,;-7.83,2.61,;-6.76,1.5,;-5.27,1.87,;-4.84,3.35,;.93,-.7,;.51,-2.18,;-.98,-2.55,;1.58,-3.29,;3.08,-2.91,;4.15,-4.02,;5.64,-3.65,;3.72,-5.5,;3.5,-1.43,;5.03,-1.59,;6.31,-.73,;7.58,.13,;7.47,1.67,;6.09,2.34,;4.81,1.48,;4.92,-.06,;2.43,-.33,;8.75,2.53,;7.89,3.81,;9.61,1.25,;10.03,3.39,)| Show InChI InChI=1S/C32H26F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-14,36,42H,1-4H3,(H2,35,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.450 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194717
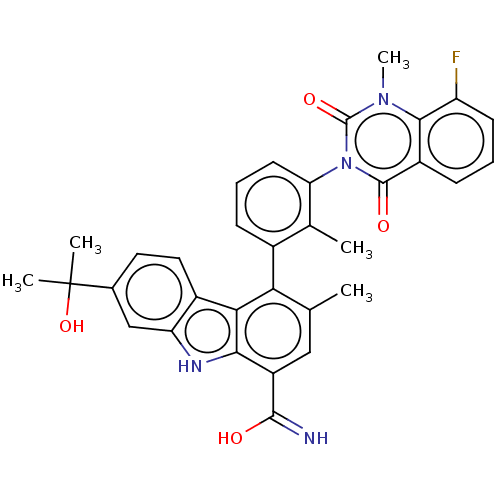
(CHEMBL3931086)Show SMILES Cc1cc(C(O)=N)c2[nH]c3cc(ccc3c2c1-c1cccc(c1C)-n1c(=O)n(C)c2c(F)cccc2c1=O)C(C)(C)O |(-.98,-2.55,;.51,-2.18,;1.58,-3.29,;3.08,-2.91,;4.15,-4.02,;5.64,-3.65,;3.72,-5.5,;3.5,-1.43,;5.03,-1.59,;6.31,-.73,;7.58,.13,;7.47,1.67,;6.09,2.34,;4.81,1.48,;4.92,-.06,;2.43,-.33,;.93,-.7,;-.14,.41,;.29,1.89,;-.78,2.99,;-2.28,2.62,;-2.7,1.14,;-1.63,.03,;-2.05,-1.45,;-4.2,.77,;-4.62,-.71,;-3.55,-1.82,;-6.11,-1.09,;-6.54,-2.57,;-7.18,.02,;-8.68,-.35,;-9.1,-1.84,;-9.75,.75,;-9.32,2.23,;-7.83,2.61,;-6.76,1.5,;-5.27,1.87,;-4.84,3.35,;8.75,2.53,;7.89,3.81,;9.61,1.25,;10.03,3.39,)| Show InChI InChI=1S/C33H29FN4O4/c1-16-14-22(30(35)39)28-27(20-13-12-18(33(3,4)42)15-24(20)36-28)26(16)19-8-7-11-25(17(19)2)38-31(40)21-9-6-10-23(34)29(21)37(5)32(38)41/h6-15,36,42H,1-5H3,(H2,35,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.650 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230081
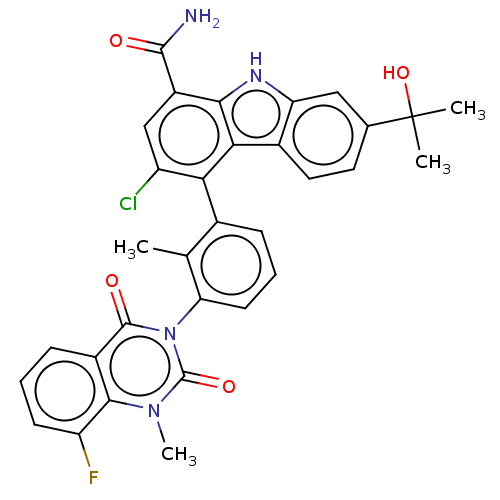
(US9334290, 1 | US9334290, 2)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(2.75,9.19,;1.42,8.42,;.08,9.19,;-1.25,8.42,;-1.25,6.88,;.08,6.11,;1.42,6.88,;2.75,6.11,;2.75,4.57,;1.42,3.8,;4.09,3.8,;4.09,2.26,;5.42,4.57,;6.75,3.8,;6.75,2.26,;8.09,4.57,;8.09,6.11,;6.75,6.88,;5.42,6.11,;4.09,6.88,;4.09,8.42,;.08,10.73,;1.42,11.5,;2.96,11.5,;1.42,13.04,;.08,13.81,;.08,15.35,;1.42,16.12,;-1.25,16.12,;-1.25,13.04,;-2.71,13.51,;-3.62,12.27,;-5.15,12.1,;-5.78,10.7,;-4.87,9.45,;-3.34,9.61,;-2.71,11.02,;-1.25,11.5,;-7.32,10.7,;-8.09,9.36,;-6.55,9.36,;-8.09,12.03,)| Show InChI InChI=1S/C32H26ClFN4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-22(34)28(19)37(4)31(38)41)25-21(33)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-14,36,42H,1-4H3,(H2,35,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230081

(US9334290, 1 | US9334290, 2)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(2.75,9.19,;1.42,8.42,;.08,9.19,;-1.25,8.42,;-1.25,6.88,;.08,6.11,;1.42,6.88,;2.75,6.11,;2.75,4.57,;1.42,3.8,;4.09,3.8,;4.09,2.26,;5.42,4.57,;6.75,3.8,;6.75,2.26,;8.09,4.57,;8.09,6.11,;6.75,6.88,;5.42,6.11,;4.09,6.88,;4.09,8.42,;.08,10.73,;1.42,11.5,;2.96,11.5,;1.42,13.04,;.08,13.81,;.08,15.35,;1.42,16.12,;-1.25,16.12,;-1.25,13.04,;-2.71,13.51,;-3.62,12.27,;-5.15,12.1,;-5.78,10.7,;-4.87,9.45,;-3.34,9.61,;-2.71,11.02,;-1.25,11.5,;-7.32,10.7,;-8.09,9.36,;-6.55,9.36,;-8.09,12.03,)| Show InChI InChI=1S/C32H26ClFN4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-22(34)28(19)37(4)31(38)41)25-21(33)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-14,36,42H,1-4H3,(H2,35,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230101
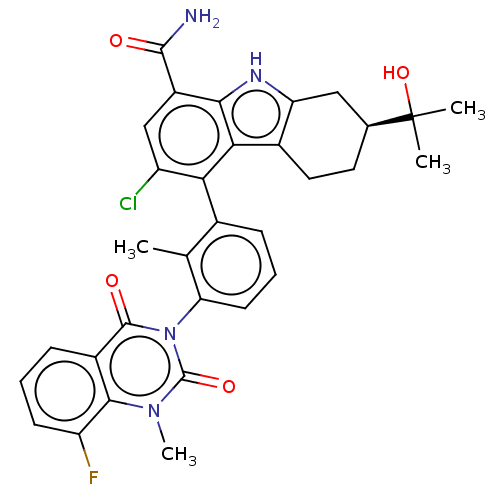
(US10106559, Example 58 | US10435415, Example 26 | ...)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3C[C@H](CCc3c12)C(C)(C)O |r,wD:33.43,(2.75,,;1.42,-.77,;.08,,;-1.25,-.77,;-1.25,-2.31,;.08,-3.08,;1.42,-2.31,;2.75,-3.08,;2.75,-4.62,;1.42,-5.39,;4.09,-5.39,;4.09,-6.93,;5.42,-4.62,;6.75,-5.39,;6.75,-6.93,;8.09,-4.62,;8.09,-3.08,;6.75,-2.31,;5.42,-3.08,;4.09,-2.31,;4.09,-.77,;.08,1.54,;1.42,2.31,;2.75,1.54,;1.42,3.85,;.08,4.62,;.08,6.16,;1.42,6.93,;-1.25,6.93,;-1.25,3.85,;-2.71,4.33,;-3.62,3.08,;-5.15,2.92,;-5.78,1.51,;-4.87,.27,;-3.34,.43,;-2.71,1.83,;-1.25,2.31,;-7.32,1.51,;-8.09,.18,;-6.55,.18,;-8.09,2.85,)| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230091
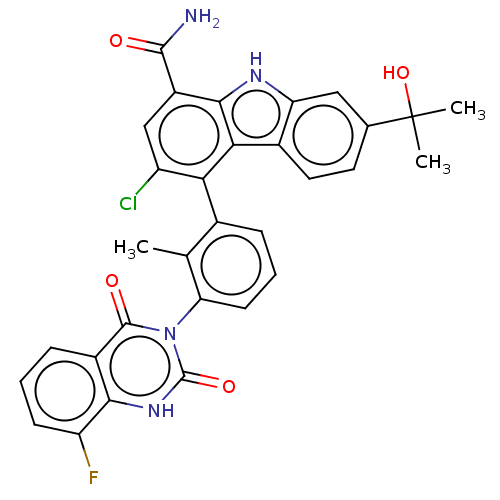
(US10106559, Example 14 | US10435415, Example 14 | ...)Show SMILES Cc1c(cccc1-n1c(=O)[nH]c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(2.75,,;1.42,-.77,;.08,,;-1.25,-.77,;-1.25,-2.31,;.08,-3.08,;1.42,-2.31,;2.75,-3.08,;2.75,-4.62,;1.42,-5.39,;4.09,-5.39,;5.42,-4.62,;6.75,-5.39,;6.75,-6.93,;8.09,-4.62,;8.09,-3.08,;6.75,-2.31,;5.42,-3.08,;4.09,-2.31,;4.09,-.77,;.08,1.54,;1.42,2.31,;2.75,1.54,;1.42,3.85,;.08,4.62,;.08,6.16,;1.42,6.93,;-1.25,6.93,;-1.25,3.85,;-2.71,4.33,;-3.62,3.08,;-5.15,2.92,;-5.78,1.51,;-4.87,.27,;-3.34,.43,;-2.71,1.83,;-1.25,2.31,;-7.32,1.51,;-8.09,.18,;-6.55,.18,;-8.09,2.85,)| Show InChI InChI=1S/C31H24ClFN4O4/c1-14-16(6-5-9-23(14)37-29(39)18-7-4-8-21(33)26(18)36-30(37)40)24-20(32)13-19(28(34)38)27-25(24)17-11-10-15(31(2,3)41)12-22(17)35-27/h4-13,35,41H,1-3H3,(H2,34,38)(H,36,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230091

(US10106559, Example 14 | US10435415, Example 14 | ...)Show SMILES Cc1c(cccc1-n1c(=O)[nH]c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(2.75,,;1.42,-.77,;.08,,;-1.25,-.77,;-1.25,-2.31,;.08,-3.08,;1.42,-2.31,;2.75,-3.08,;2.75,-4.62,;1.42,-5.39,;4.09,-5.39,;5.42,-4.62,;6.75,-5.39,;6.75,-6.93,;8.09,-4.62,;8.09,-3.08,;6.75,-2.31,;5.42,-3.08,;4.09,-2.31,;4.09,-.77,;.08,1.54,;1.42,2.31,;2.75,1.54,;1.42,3.85,;.08,4.62,;.08,6.16,;1.42,6.93,;-1.25,6.93,;-1.25,3.85,;-2.71,4.33,;-3.62,3.08,;-5.15,2.92,;-5.78,1.51,;-4.87,.27,;-3.34,.43,;-2.71,1.83,;-1.25,2.31,;-7.32,1.51,;-8.09,.18,;-6.55,.18,;-8.09,2.85,)| Show InChI InChI=1S/C31H24ClFN4O4/c1-14-16(6-5-9-23(14)37-29(39)18-7-4-8-21(33)26(18)36-30(37)40)24-20(32)13-19(28(34)38)27-25(24)17-11-10-15(31(2,3)41)12-22(17)35-27/h4-13,35,41H,1-3H3,(H2,34,38)(H,36,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230103
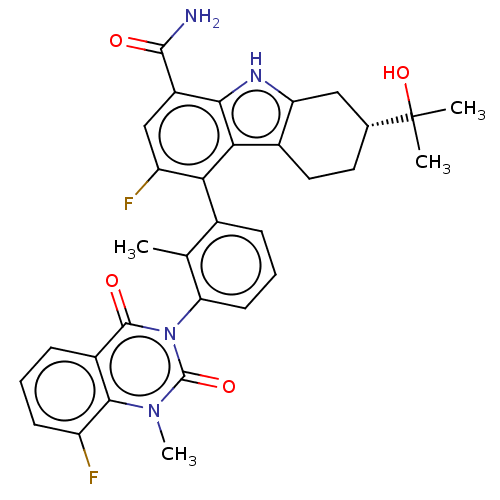
(US10106559, Example 27 | US10435415, Example 27 | ...)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(F)cc(C(N)=O)c2[nH]c3C[C@@H](CCc3c12)C(C)(C)O |r,wU:33.43,(2.75,,;1.42,-.77,;.08,,;-1.25,-.77,;-1.25,-2.31,;.08,-3.08,;1.42,-2.31,;2.75,-3.08,;2.75,-4.62,;1.42,-5.39,;4.09,-5.39,;4.09,-6.93,;5.42,-4.62,;6.75,-5.39,;6.75,-6.93,;8.09,-4.62,;8.09,-3.08,;6.75,-2.31,;5.42,-3.08,;4.09,-2.31,;4.09,-.77,;.08,1.54,;1.42,2.31,;2.75,1.54,;1.42,3.85,;.08,4.62,;.08,6.16,;1.42,6.93,;-1.25,6.93,;-1.25,3.85,;-2.71,4.33,;-3.62,3.08,;-5.15,2.92,;-5.78,1.51,;-4.87,.27,;-3.34,.43,;-2.71,1.83,;-1.25,2.31,;-7.32,1.51,;-8.09,.18,;-6.55,.18,;-8.09,2.85,)| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194723
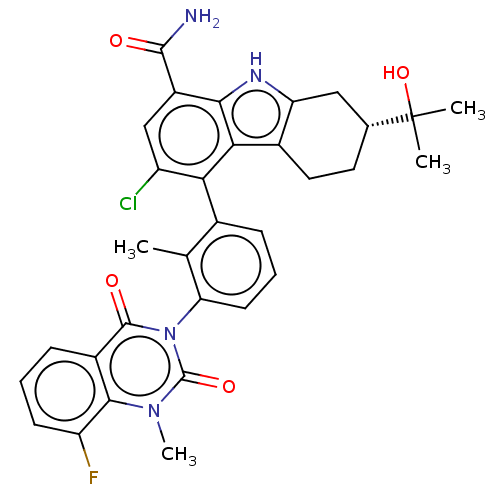
(CHEMBL3944049)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3C[C@@H](CCc3c12)C(C)(C)O |r,wU:33.43,(73.77,-22.36,;72.44,-23.13,;71.11,-22.37,;69.77,-23.14,;69.77,-24.68,;71.11,-25.45,;72.45,-24.67,;73.78,-25.43,;73.78,-26.97,;72.44,-27.74,;75.1,-27.74,;75.11,-29.28,;76.44,-26.97,;77.77,-27.73,;77.77,-29.27,;79.1,-26.96,;79.09,-25.42,;77.76,-24.66,;76.44,-25.43,;75.1,-24.66,;75.09,-23.12,;71.11,-20.83,;72.45,-20.06,;73.78,-20.83,;72.44,-18.51,;71.1,-17.75,;71.1,-16.21,;72.43,-15.44,;69.77,-15.44,;69.78,-18.52,;68.31,-18.04,;67.4,-19.28,;65.87,-19.44,;65.23,-20.84,;66.14,-22.1,;67.67,-21.94,;68.3,-20.54,;69.77,-20.06,;63.73,-20.44,;62.63,-19.34,;64.13,-18.94,;62.64,-21.53,)| Show InChI InChI=1S/C32H30ClFN4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-22(34)28(19)37(4)31(38)41)25-21(33)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194715
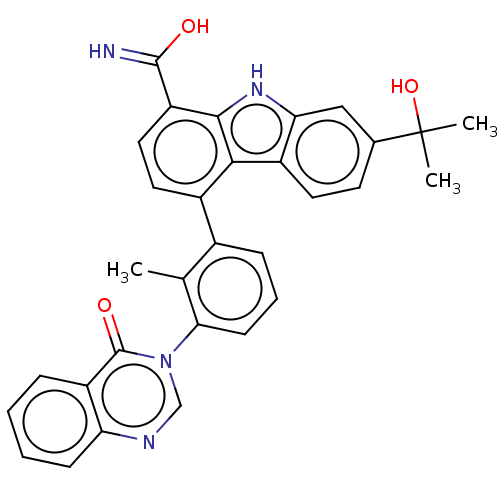
(CHEMBL3918580)Show SMILES Cc1c(cccc1-n1cnc2ccccc2c1=O)-c1ccc(C(O)=N)c2[nH]c3cc(ccc3c12)C(C)(C)O Show InChI InChI=1S/C31H26N4O3/c1-17-19(8-6-10-26(17)35-16-33-24-9-5-4-7-22(24)30(35)37)20-13-14-23(29(32)36)28-27(20)21-12-11-18(31(2,3)38)15-25(21)34-28/h4-16,34,38H,1-3H3,(H2,32,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194724
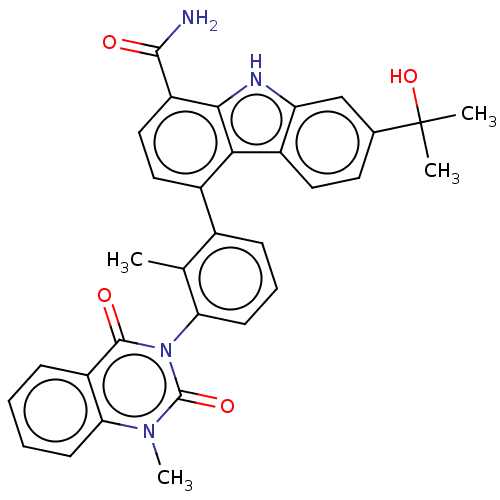
(CHEMBL3941224)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2ccccc2c1=O)-c1ccc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(15.19,-30.14,;13.85,-30.89,;12.53,-30.1,;11.19,-30.85,;11.16,-32.39,;12.48,-33.18,;13.83,-32.42,;15.15,-33.2,;15.13,-34.74,;13.78,-35.5,;16.45,-35.53,;16.43,-37.08,;17.79,-34.77,;19.11,-35.56,;20.45,-34.81,;20.47,-33.27,;19.15,-32.49,;17.82,-33.24,;16.49,-32.44,;16.51,-30.91,;12.56,-28.57,;13.91,-27.82,;13.92,-26.27,;12.6,-25.48,;12.62,-23.95,;13.96,-23.19,;11.29,-23.16,;11.26,-26.24,;9.79,-25.73,;8.86,-26.97,;7.33,-27.11,;6.68,-28.5,;7.55,-29.76,;9.09,-29.64,;9.75,-28.24,;11.23,-27.78,;5.14,-28.62,;3.74,-27.97,;5.01,-27.08,;4.49,-30.01,)| Show InChI InChI=1S/C32H28N4O4/c1-17-19(9-7-11-25(17)36-30(38)22-8-5-6-10-26(22)35(4)31(36)39)20-14-15-23(29(33)37)28-27(20)21-13-12-18(32(2,3)40)16-24(21)34-28/h5-16,34,40H,1-4H3,(H2,33,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194719
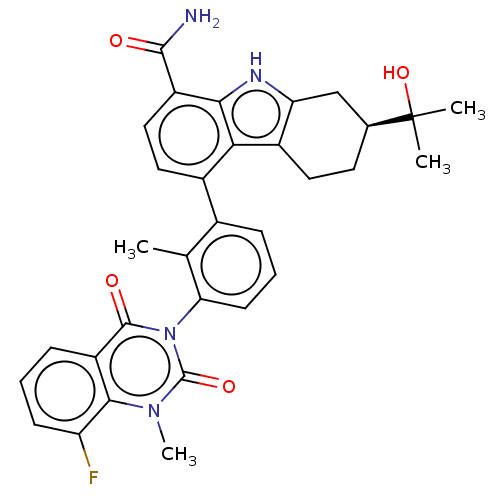
(CHEMBL3976719)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1ccc(C(N)=O)c2[nH]c3C[C@H](CCc3c12)C(C)(C)O |r,wD:32.42,(74.94,-7.74,;73.61,-8.52,;72.28,-7.75,;70.95,-8.52,;70.95,-10.06,;72.28,-10.83,;73.62,-10.05,;74.95,-10.82,;74.95,-12.36,;73.62,-13.12,;76.28,-13.12,;76.28,-14.66,;77.61,-12.35,;78.94,-13.12,;78.94,-14.66,;80.27,-12.35,;80.27,-10.8,;78.93,-10.04,;77.61,-10.81,;76.27,-10.04,;76.27,-8.5,;72.28,-6.22,;73.62,-5.45,;73.61,-3.9,;72.28,-3.13,;72.27,-1.59,;73.61,-.82,;70.94,-.83,;70.95,-3.9,;69.48,-3.42,;68.58,-4.67,;67.04,-4.82,;66.4,-6.23,;67.31,-7.48,;68.85,-7.33,;69.48,-5.92,;70.95,-5.45,;64.9,-5.82,;63.81,-4.73,;65.3,-4.33,;63.81,-6.91,)| Show InChI InChI=1S/C32H31FN4O4/c1-16-18(7-6-10-25(16)37-30(39)22-8-5-9-23(33)28(22)36(4)31(37)40)19-13-14-21(29(34)38)27-26(19)20-12-11-17(32(2,3)41)15-24(20)35-27/h5-10,13-14,17,35,41H,11-12,15H2,1-4H3,(H2,34,38)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194721
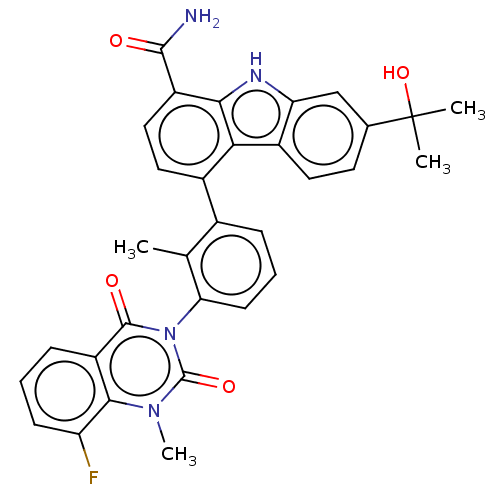
(CHEMBL3908310 | US9714234, 4)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1ccc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(15.71,-28.16,;14.37,-28.91,;13.05,-28.13,;11.71,-28.87,;11.68,-30.41,;13.01,-31.2,;14.35,-30.44,;15.68,-31.22,;15.66,-32.76,;14.31,-33.51,;16.97,-33.55,;16.95,-35.09,;18.31,-32.8,;19.63,-33.58,;19.61,-35.12,;20.98,-32.83,;20.99,-31.29,;19.67,-30.51,;18.34,-31.26,;17.01,-30.47,;17.03,-28.93,;13.08,-26.59,;14.43,-25.84,;14.45,-24.29,;13.12,-23.51,;13.14,-21.97,;14.48,-21.21,;11.82,-21.18,;11.78,-24.26,;10.31,-23.75,;9.38,-24.99,;7.85,-25.13,;7.2,-26.52,;8.07,-27.79,;9.61,-27.66,;10.27,-26.26,;11.75,-25.8,;5.66,-26.64,;4.26,-25.99,;5.53,-25.1,;5.01,-28.04,)| Show InChI InChI=1S/C32H27FN4O4/c1-16-18(7-6-10-25(16)37-30(39)22-8-5-9-23(33)28(22)36(4)31(37)40)19-13-14-21(29(34)38)27-26(19)20-12-11-17(32(2,3)41)15-24(20)35-27/h5-15,35,41H,1-4H3,(H2,34,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human PBM cells assessed as suppression of FCRgammaR/immune complex-stimulated TNFalpha production |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human peripheral B cells assessed as suppression of BCR/anti-IgM/IgG-stimulated cell proliferation |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human peripheral B cells assessed as suppression of BCR/anti-IgM/IgG-stimulated CD86 surface expression |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194721

(CHEMBL3908310 | US9714234, 4)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1ccc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(15.71,-28.16,;14.37,-28.91,;13.05,-28.13,;11.71,-28.87,;11.68,-30.41,;13.01,-31.2,;14.35,-30.44,;15.68,-31.22,;15.66,-32.76,;14.31,-33.51,;16.97,-33.55,;16.95,-35.09,;18.31,-32.8,;19.63,-33.58,;19.61,-35.12,;20.98,-32.83,;20.99,-31.29,;19.67,-30.51,;18.34,-31.26,;17.01,-30.47,;17.03,-28.93,;13.08,-26.59,;14.43,-25.84,;14.45,-24.29,;13.12,-23.51,;13.14,-21.97,;14.48,-21.21,;11.82,-21.18,;11.78,-24.26,;10.31,-23.75,;9.38,-24.99,;7.85,-25.13,;7.2,-26.52,;8.07,-27.79,;9.61,-27.66,;10.27,-26.26,;11.75,-25.8,;5.66,-26.64,;4.26,-25.99,;5.53,-25.1,;5.01,-28.04,)| Show InChI InChI=1S/C32H27FN4O4/c1-16-18(7-6-10-25(16)37-30(39)22-8-5-9-23(33)28(22)36(4)31(37)40)19-13-14-21(29(34)38)27-26(19)20-12-11-17(32(2,3)41)15-24(20)35-27/h5-15,35,41H,1-4H3,(H2,34,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194721

(CHEMBL3908310 | US9714234, 4)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1ccc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(15.71,-28.16,;14.37,-28.91,;13.05,-28.13,;11.71,-28.87,;11.68,-30.41,;13.01,-31.2,;14.35,-30.44,;15.68,-31.22,;15.66,-32.76,;14.31,-33.51,;16.97,-33.55,;16.95,-35.09,;18.31,-32.8,;19.63,-33.58,;19.61,-35.12,;20.98,-32.83,;20.99,-31.29,;19.67,-30.51,;18.34,-31.26,;17.01,-30.47,;17.03,-28.93,;13.08,-26.59,;14.43,-25.84,;14.45,-24.29,;13.12,-23.51,;13.14,-21.97,;14.48,-21.21,;11.82,-21.18,;11.78,-24.26,;10.31,-23.75,;9.38,-24.99,;7.85,-25.13,;7.2,-26.52,;8.07,-27.79,;9.61,-27.66,;10.27,-26.26,;11.75,-25.8,;5.66,-26.64,;4.26,-25.99,;5.53,-25.1,;5.01,-28.04,)| Show InChI InChI=1S/C32H27FN4O4/c1-16-18(7-6-10-25(16)37-30(39)22-8-5-9-23(33)28(22)36(4)31(37)40)19-13-14-21(29(34)38)27-26(19)20-12-11-17(32(2,3)41)15-24(20)35-27/h5-15,35,41H,1-4H3,(H2,34,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194726
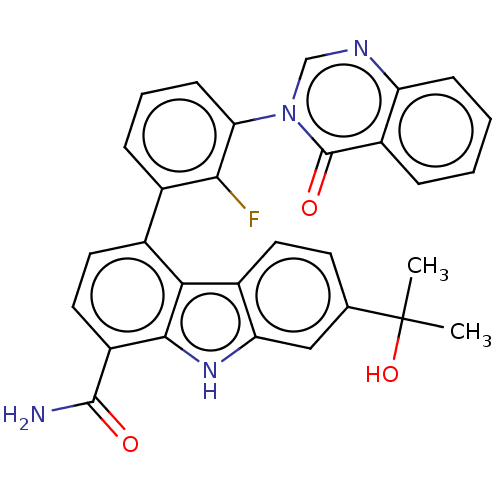
(CHEMBL3977725)Show SMILES CC(C)(O)c1ccc2c(c1)[nH]c1c(ccc(-c3cccc(c3F)-n3cnc4ccccc4c3=O)c21)C(N)=O Show InChI InChI=1S/C30H23FN4O3/c1-30(2,38)16-10-11-19-23(14-16)34-27-21(28(32)36)13-12-17(25(19)27)18-7-5-9-24(26(18)31)35-15-33-22-8-4-3-6-20(22)29(35)37/h3-15,34,38H,1-2H3,(H2,32,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 5.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194718

(CHEMBL3899411)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(cc(C(O)=N)c2[nH]c3cc(ccc3c12)C(C)(C)O)C#N |(-2.03,-1.34,;-1.6,.13,;-.1,.5,;.33,1.98,;-.74,3.09,;-2.23,2.73,;-2.66,1.25,;-4.16,.88,;-4.59,-.6,;-3.52,-1.71,;-6.08,-.97,;-6.51,-2.44,;-7.15,.15,;-8.65,-.22,;-9.08,-1.7,;-9.71,.89,;-9.28,2.37,;-7.78,2.74,;-6.72,1.63,;-5.22,1.99,;-4.79,3.47,;.96,-.61,;.53,-2.09,;1.6,-3.2,;3.09,-2.84,;4.16,-3.95,;5.66,-3.58,;3.73,-5.43,;3.52,-1.36,;5.06,-1.52,;6.34,-.67,;7.62,.19,;7.51,1.73,;6.13,2.41,;4.85,1.55,;4.95,.01,;2.46,-.24,;8.79,2.58,;7.94,3.86,;9.65,1.3,;10.07,3.44,;-.96,-2.46,;-2.46,-2.82,)| Show InChI InChI=1S/C33H26FN5O4/c1-16-19(7-6-10-25(16)39-31(41)21-8-5-9-23(34)29(21)38(4)32(39)42)26-17(15-35)13-22(30(36)40)28-27(26)20-12-11-18(33(2,3)43)14-24(20)37-28/h5-14,37,43H,1-4H3,(H2,36,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194715

(CHEMBL3918580)Show SMILES Cc1c(cccc1-n1cnc2ccccc2c1=O)-c1ccc(C(O)=N)c2[nH]c3cc(ccc3c12)C(C)(C)O Show InChI InChI=1S/C31H26N4O3/c1-17-19(8-6-10-26(17)35-16-33-24-9-5-4-7-22(24)30(35)37)20-13-14-23(29(32)36)28-27(20)21-12-11-18(31(2,3)38)15-25(21)34-28/h4-16,34,38H,1-3H3,(H2,32,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human peripheral B cells assessed as suppression of BCR/anti-IgM/IgG-stimulated cell proliferation |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Tec
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of TEC (unknown origin) |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230081

(US9334290, 1 | US9334290, 2)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(2.75,9.19,;1.42,8.42,;.08,9.19,;-1.25,8.42,;-1.25,6.88,;.08,6.11,;1.42,6.88,;2.75,6.11,;2.75,4.57,;1.42,3.8,;4.09,3.8,;4.09,2.26,;5.42,4.57,;6.75,3.8,;6.75,2.26,;8.09,4.57,;8.09,6.11,;6.75,6.88,;5.42,6.11,;4.09,6.88,;4.09,8.42,;.08,10.73,;1.42,11.5,;2.96,11.5,;1.42,13.04,;.08,13.81,;.08,15.35,;1.42,16.12,;-1.25,16.12,;-1.25,13.04,;-2.71,13.51,;-3.62,12.27,;-5.15,12.1,;-5.78,10.7,;-4.87,9.45,;-3.34,9.61,;-2.71,11.02,;-1.25,11.5,;-7.32,10.7,;-8.09,9.36,;-6.55,9.36,;-8.09,12.03,)| Show InChI InChI=1S/C32H26ClFN4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-22(34)28(19)37(4)31(38)41)25-21(33)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-14,36,42H,1-4H3,(H2,35,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194725
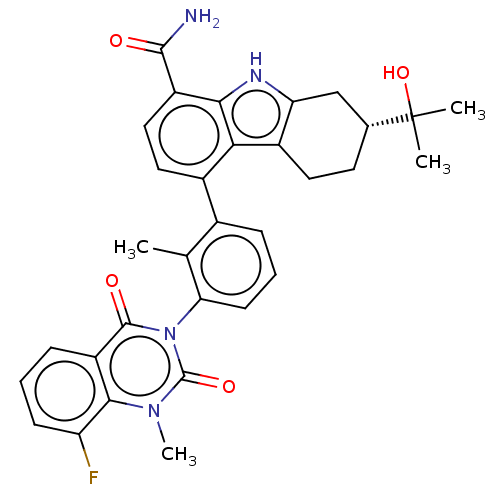
(CHEMBL3891391)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1ccc(C(N)=O)c2[nH]c3C[C@@H](CCc3c12)C(C)(C)O |r,wU:32.42,(57.41,-37.92,;56.08,-38.69,;54.75,-37.93,;53.42,-38.69,;53.42,-40.23,;54.75,-41,;56.09,-40.23,;57.42,-40.99,;57.42,-42.53,;56.08,-43.3,;58.75,-43.3,;58.75,-44.83,;60.08,-42.52,;61.41,-43.29,;61.41,-44.83,;62.74,-42.52,;62.73,-40.98,;61.4,-40.22,;60.08,-40.99,;58.74,-40.21,;58.74,-38.67,;54.75,-36.39,;56.09,-35.62,;56.08,-34.07,;54.75,-33.31,;54.74,-31.77,;56.07,-30.99,;53.41,-31,;53.42,-34.07,;51.95,-33.59,;51.04,-34.84,;49.51,-34.99,;48.87,-36.4,;49.78,-37.66,;51.31,-37.5,;51.94,-36.1,;53.42,-35.62,;47.37,-36,;46.28,-34.9,;47.77,-34.5,;46.28,-37.08,)| Show InChI InChI=1S/C32H31FN4O4/c1-16-18(7-6-10-25(16)37-30(39)22-8-5-9-23(33)28(22)36(4)31(37)40)19-13-14-21(29(34)38)27-26(19)20-12-11-17(32(2,3)41)15-24(20)35-27/h5-10,13-14,17,35,41H,11-12,15H2,1-4H3,(H2,34,38)/t17-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194722

(CHEMBL3896019)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(F)cc(C(O)=N)c2[nH]c3cc(ccc3c12)C(C)(C)O |(-2.05,-1.45,;-1.63,.03,;-.14,.41,;.29,1.89,;-.78,2.99,;-2.28,2.62,;-2.7,1.14,;-4.2,.77,;-4.62,-.71,;-3.55,-1.82,;-6.11,-1.09,;-6.54,-2.57,;-7.18,.02,;-8.68,-.35,;-9.1,-1.84,;-9.75,.75,;-9.32,2.23,;-7.83,2.61,;-6.76,1.5,;-5.27,1.87,;-4.84,3.35,;.93,-.7,;.51,-2.18,;-.98,-2.55,;1.58,-3.29,;3.08,-2.91,;4.15,-4.02,;5.64,-3.65,;3.72,-5.5,;3.5,-1.43,;5.03,-1.59,;6.31,-.73,;7.58,.13,;7.47,1.67,;6.09,2.34,;4.81,1.48,;4.92,-.06,;2.43,-.33,;8.75,2.53,;7.89,3.81,;9.61,1.25,;10.03,3.39,)| Show InChI InChI=1S/C32H26F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-14,36,42H,1-4H3,(H2,35,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194715

(CHEMBL3918580)Show SMILES Cc1c(cccc1-n1cnc2ccccc2c1=O)-c1ccc(C(O)=N)c2[nH]c3cc(ccc3c12)C(C)(C)O Show InChI InChI=1S/C31H26N4O3/c1-17-19(8-6-10-26(17)35-16-33-24-9-5-4-7-22(24)30(35)37)20-13-14-23(29(32)36)28-27(20)21-12-11-18(31(2,3)38)15-25(21)34-28/h4-16,34,38H,1-3H3,(H2,32,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human PBM cells assessed as suppression of FCRgammaR/immune complex-stimulated TNFalpha production |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ITK/TSK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of ITK (unknown origin) |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194717

(CHEMBL3931086)Show SMILES Cc1cc(C(O)=N)c2[nH]c3cc(ccc3c2c1-c1cccc(c1C)-n1c(=O)n(C)c2c(F)cccc2c1=O)C(C)(C)O |(-.98,-2.55,;.51,-2.18,;1.58,-3.29,;3.08,-2.91,;4.15,-4.02,;5.64,-3.65,;3.72,-5.5,;3.5,-1.43,;5.03,-1.59,;6.31,-.73,;7.58,.13,;7.47,1.67,;6.09,2.34,;4.81,1.48,;4.92,-.06,;2.43,-.33,;.93,-.7,;-.14,.41,;.29,1.89,;-.78,2.99,;-2.28,2.62,;-2.7,1.14,;-1.63,.03,;-2.05,-1.45,;-4.2,.77,;-4.62,-.71,;-3.55,-1.82,;-6.11,-1.09,;-6.54,-2.57,;-7.18,.02,;-8.68,-.35,;-9.1,-1.84,;-9.75,.75,;-9.32,2.23,;-7.83,2.61,;-6.76,1.5,;-5.27,1.87,;-4.84,3.35,;8.75,2.53,;7.89,3.81,;9.61,1.25,;10.03,3.39,)| Show InChI InChI=1S/C33H29FN4O4/c1-16-14-22(30(35)39)28-27(20-13-12-18(33(3,4)42)15-24(20)36-28)26(16)19-8-7-11-25(17(19)2)38-31(40)21-9-6-10-23(34)29(21)37(5)32(38)41/h6-15,36,42H,1-5H3,(H2,35,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230101

(US10106559, Example 58 | US10435415, Example 26 | ...)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3C[C@H](CCc3c12)C(C)(C)O |r,wD:33.43,(2.75,,;1.42,-.77,;.08,,;-1.25,-.77,;-1.25,-2.31,;.08,-3.08,;1.42,-2.31,;2.75,-3.08,;2.75,-4.62,;1.42,-5.39,;4.09,-5.39,;4.09,-6.93,;5.42,-4.62,;6.75,-5.39,;6.75,-6.93,;8.09,-4.62,;8.09,-3.08,;6.75,-2.31,;5.42,-3.08,;4.09,-2.31,;4.09,-.77,;.08,1.54,;1.42,2.31,;2.75,1.54,;1.42,3.85,;.08,4.62,;.08,6.16,;1.42,6.93,;-1.25,6.93,;-1.25,3.85,;-2.71,4.33,;-3.62,3.08,;-5.15,2.92,;-5.78,1.51,;-4.87,.27,;-3.34,.43,;-2.71,1.83,;-1.25,2.31,;-7.32,1.51,;-8.09,.18,;-6.55,.18,;-8.09,2.85,)| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194721

(CHEMBL3908310 | US9714234, 4)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1ccc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(15.71,-28.16,;14.37,-28.91,;13.05,-28.13,;11.71,-28.87,;11.68,-30.41,;13.01,-31.2,;14.35,-30.44,;15.68,-31.22,;15.66,-32.76,;14.31,-33.51,;16.97,-33.55,;16.95,-35.09,;18.31,-32.8,;19.63,-33.58,;19.61,-35.12,;20.98,-32.83,;20.99,-31.29,;19.67,-30.51,;18.34,-31.26,;17.01,-30.47,;17.03,-28.93,;13.08,-26.59,;14.43,-25.84,;14.45,-24.29,;13.12,-23.51,;13.14,-21.97,;14.48,-21.21,;11.82,-21.18,;11.78,-24.26,;10.31,-23.75,;9.38,-24.99,;7.85,-25.13,;7.2,-26.52,;8.07,-27.79,;9.61,-27.66,;10.27,-26.26,;11.75,-25.8,;5.66,-26.64,;4.26,-25.99,;5.53,-25.1,;5.01,-28.04,)| Show InChI InChI=1S/C32H27FN4O4/c1-16-18(7-6-10-25(16)37-30(39)22-8-5-9-23(33)28(22)36(4)31(37)40)19-13-14-21(29(34)38)27-26(19)20-12-11-17(32(2,3)41)15-24(20)35-27/h5-15,35,41H,1-4H3,(H2,34,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Blk
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BLK (unknown origin) |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase TXK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of TXK (unknown origin) |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194726

(CHEMBL3977725)Show SMILES CC(C)(O)c1ccc2c(c1)[nH]c1c(ccc(-c3cccc(c3F)-n3cnc4ccccc4c3=O)c21)C(N)=O Show InChI InChI=1S/C30H23FN4O3/c1-30(2,38)16-10-11-19-23(14-16)34-27-21(28(32)36)13-12-17(25(19)27)18-7-5-9-24(26(18)31)35-15-33-22-8-4-3-6-20(22)29(35)37/h3-15,34,38H,1-2H3,(H2,32,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194715

(CHEMBL3918580)Show SMILES Cc1c(cccc1-n1cnc2ccccc2c1=O)-c1ccc(C(O)=N)c2[nH]c3cc(ccc3c12)C(C)(C)O Show InChI InChI=1S/C31H26N4O3/c1-17-19(8-6-10-26(17)35-16-33-24-9-5-4-7-22(24)30(35)37)20-13-14-23(29(32)36)28-27(20)21-12-11-18(31(2,3)38)15-25(21)34-28/h4-16,34,38H,1-3H3,(H2,32,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Cytoplasmic tyrosine-protein kinase BMX
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BMX (unknown origin) |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230103

(US10106559, Example 27 | US10435415, Example 27 | ...)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(F)cc(C(N)=O)c2[nH]c3C[C@@H](CCc3c12)C(C)(C)O |r,wU:33.43,(2.75,,;1.42,-.77,;.08,,;-1.25,-.77,;-1.25,-2.31,;.08,-3.08,;1.42,-2.31,;2.75,-3.08,;2.75,-4.62,;1.42,-5.39,;4.09,-5.39,;4.09,-6.93,;5.42,-4.62,;6.75,-5.39,;6.75,-6.93,;8.09,-4.62,;8.09,-3.08,;6.75,-2.31,;5.42,-3.08,;4.09,-2.31,;4.09,-.77,;.08,1.54,;1.42,2.31,;2.75,1.54,;1.42,3.85,;.08,4.62,;.08,6.16,;1.42,6.93,;-1.25,6.93,;-1.25,3.85,;-2.71,4.33,;-3.62,3.08,;-5.15,2.92,;-5.78,1.51,;-4.87,.27,;-3.34,.43,;-2.71,1.83,;-1.25,2.31,;-7.32,1.51,;-8.09,.18,;-6.55,.18,;-8.09,2.85,)| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194715

(CHEMBL3918580)Show SMILES Cc1c(cccc1-n1cnc2ccccc2c1=O)-c1ccc(C(O)=N)c2[nH]c3cc(ccc3c12)C(C)(C)O Show InChI InChI=1S/C31H26N4O3/c1-17-19(8-6-10-26(17)35-16-33-24-9-5-4-7-22(24)30(35)37)20-13-14-23(29(32)36)28-27(20)21-12-11-18(31(2,3)38)15-25(21)34-28/h4-16,34,38H,1-3H3,(H2,32,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human peripheral B cells assessed as suppression of BCR/anti-IgM/IgG-stimulated CD86 surface expression |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194716
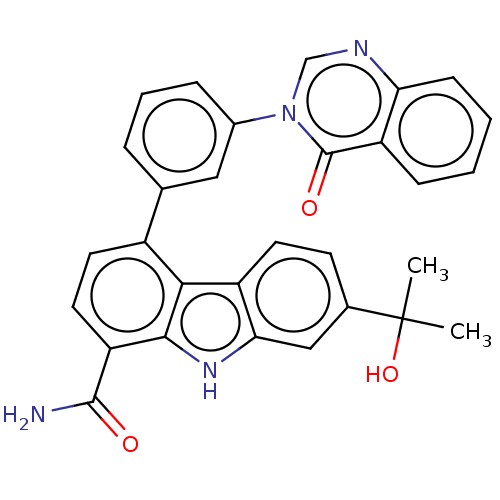
(CHEMBL3939772)Show SMILES CC(C)(O)c1ccc2c(c1)[nH]c1c(ccc(-c3cccc(c3)-n3cnc4ccccc4c3=O)c21)C(N)=O Show InChI InChI=1S/C30H24N4O3/c1-30(2,37)18-10-11-21-25(15-18)33-27-23(28(31)35)13-12-20(26(21)27)17-6-5-7-19(14-17)34-16-32-24-9-4-3-8-22(24)29(34)36/h3-16,33,37H,1-2H3,(H2,31,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of full length recombinant human His-tagged BTK expressed in baculovirus using fluoresceinated peptide incubated for 60 mins by fluorescen... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194719

(CHEMBL3976719)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1ccc(C(N)=O)c2[nH]c3C[C@H](CCc3c12)C(C)(C)O |r,wD:32.42,(74.94,-7.74,;73.61,-8.52,;72.28,-7.75,;70.95,-8.52,;70.95,-10.06,;72.28,-10.83,;73.62,-10.05,;74.95,-10.82,;74.95,-12.36,;73.62,-13.12,;76.28,-13.12,;76.28,-14.66,;77.61,-12.35,;78.94,-13.12,;78.94,-14.66,;80.27,-12.35,;80.27,-10.8,;78.93,-10.04,;77.61,-10.81,;76.27,-10.04,;76.27,-8.5,;72.28,-6.22,;73.62,-5.45,;73.61,-3.9,;72.28,-3.13,;72.27,-1.59,;73.61,-.82,;70.94,-.83,;70.95,-3.9,;69.48,-3.42,;68.58,-4.67,;67.04,-4.82,;66.4,-6.23,;67.31,-7.48,;68.85,-7.33,;69.48,-5.92,;70.95,-5.45,;64.9,-5.82,;63.81,-4.73,;65.3,-4.33,;63.81,-6.91,)| Show InChI InChI=1S/C32H31FN4O4/c1-16-18(7-6-10-25(16)37-30(39)22-8-5-9-23(33)28(22)36(4)31(37)40)19-13-14-21(29(34)38)27-26(19)20-12-11-17(32(2,3)41)15-24(20)35-27/h5-10,13-14,17,35,41H,11-12,15H2,1-4H3,(H2,34,38)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 63 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 71 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of LCK (unknown origin) |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194723

(CHEMBL3944049)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3C[C@@H](CCc3c12)C(C)(C)O |r,wU:33.43,(73.77,-22.36,;72.44,-23.13,;71.11,-22.37,;69.77,-23.14,;69.77,-24.68,;71.11,-25.45,;72.45,-24.67,;73.78,-25.43,;73.78,-26.97,;72.44,-27.74,;75.1,-27.74,;75.11,-29.28,;76.44,-26.97,;77.77,-27.73,;77.77,-29.27,;79.1,-26.96,;79.09,-25.42,;77.76,-24.66,;76.44,-25.43,;75.1,-24.66,;75.09,-23.12,;71.11,-20.83,;72.45,-20.06,;73.78,-20.83,;72.44,-18.51,;71.1,-17.75,;71.1,-16.21,;72.43,-15.44,;69.77,-15.44,;69.78,-18.52,;68.31,-18.04,;67.4,-19.28,;65.87,-19.44,;65.23,-20.84,;66.14,-22.1,;67.67,-21.94,;68.3,-20.54,;69.77,-20.06,;63.73,-20.44,;62.63,-19.34,;64.13,-18.94,;62.64,-21.53,)| Show InChI InChI=1S/C32H30ClFN4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-22(34)28(19)37(4)31(38)41)25-21(33)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 84 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human Ramos-B cells assessed as suppression of BCR/anti-IgG-stimulated Ca2+ flux after 1 hr incubation in dark by FLIPR1 assay |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 89 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human whole blood basophils assessed as suppression of FceRI/anti-IgE-stimulated CD63 surface expression |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230081

(US9334290, 1 | US9334290, 2)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(2.75,9.19,;1.42,8.42,;.08,9.19,;-1.25,8.42,;-1.25,6.88,;.08,6.11,;1.42,6.88,;2.75,6.11,;2.75,4.57,;1.42,3.8,;4.09,3.8,;4.09,2.26,;5.42,4.57,;6.75,3.8,;6.75,2.26,;8.09,4.57,;8.09,6.11,;6.75,6.88,;5.42,6.11,;4.09,6.88,;4.09,8.42,;.08,10.73,;1.42,11.5,;2.96,11.5,;1.42,13.04,;.08,13.81,;.08,15.35,;1.42,16.12,;-1.25,16.12,;-1.25,13.04,;-2.71,13.51,;-3.62,12.27,;-5.15,12.1,;-5.78,10.7,;-4.87,9.45,;-3.34,9.61,;-2.71,11.02,;-1.25,11.5,;-7.32,10.7,;-8.09,9.36,;-6.55,9.36,;-8.09,12.03,)| Show InChI InChI=1S/C32H26ClFN4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-22(34)28(19)37(4)31(38)41)25-21(33)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-14,36,42H,1-4H3,(H2,35,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human whole blood assessed as suppression of BCR/anti-IgM/IgG-stimulated CD69 surface expression after 18 hrs with agitation by ... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM230081

(US9334290, 1 | US9334290, 2)Show SMILES Cc1c(cccc1-n1c(=O)n(C)c2c(F)cccc2c1=O)-c1c(Cl)cc(C(N)=O)c2[nH]c3cc(ccc3c12)C(C)(C)O |(2.75,9.19,;1.42,8.42,;.08,9.19,;-1.25,8.42,;-1.25,6.88,;.08,6.11,;1.42,6.88,;2.75,6.11,;2.75,4.57,;1.42,3.8,;4.09,3.8,;4.09,2.26,;5.42,4.57,;6.75,3.8,;6.75,2.26,;8.09,4.57,;8.09,6.11,;6.75,6.88,;5.42,6.11,;4.09,6.88,;4.09,8.42,;.08,10.73,;1.42,11.5,;2.96,11.5,;1.42,13.04,;.08,13.81,;.08,15.35,;1.42,16.12,;-1.25,16.12,;-1.25,13.04,;-2.71,13.51,;-3.62,12.27,;-5.15,12.1,;-5.78,10.7,;-4.87,9.45,;-3.34,9.61,;-2.71,11.02,;-1.25,11.5,;-7.32,10.7,;-8.09,9.36,;-6.55,9.36,;-8.09,12.03,)| Show InChI InChI=1S/C32H26ClFN4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-22(34)28(19)37(4)31(38)41)25-21(33)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-14,36,42H,1-4H3,(H2,35,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human whole blood assessed as suppression of BCR/anti-IgM/IgG-stimulated CD69 surface expression after 18 hrs with agitation by ... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50194720

(CHEMBL3900554)Show SMILES [H][C@@]1(CCc2c(C1)[nH]c1c(cc(F)c(-c3cccc(c3C)-n3c(=O)n(C)c4c(F)cccc4c3=O)c21)C(O)=N)C(C)(C)O |r,wU:1.0,(7.64,3.3,;7.24,1.81,;5.84,2.47,;4.58,1.59,;4.71,.05,;6.1,-.6,;7.37,.28,;4.84,-1.48,;3.3,-1.34,;2.9,-2.83,;1.41,-3.22,;.33,-2.13,;-1.16,-2.53,;.73,-.64,;-.36,.45,;.05,1.93,;-1.04,3.03,;-2.53,2.63,;-2.93,1.15,;-1.85,.05,;-2.25,-1.43,;-4.42,.75,;-4.82,-.73,;-3.74,-1.83,;-6.31,-1.13,;-6.71,-2.62,;-7.4,-.04,;-8.89,-.43,;-9.29,-1.92,;-9.97,.66,;-9.57,2.15,;-8.08,2.54,;-6.99,1.45,;-5.51,1.84,;-5.1,3.33,;2.22,-.25,;3.99,-3.92,;5.48,-3.53,;3.58,-5.41,;8.77,1.67,;8.91,3.21,;8.63,.14,;10.3,1.53,)| Show InChI InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human whole blood assessed as suppression of BCR/anti-IgM/IgG-stimulated CD69 surface expression after 18 hrs with agitation by ... |
J Med Chem 59: 9173-9200 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01088
BindingDB Entry DOI: 10.7270/Q23T9K5H |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data



















































