 Found 55 hits Enz. Inhib. hit(s) with all data for entry = 50030875
Found 55 hits Enz. Inhib. hit(s) with all data for entry = 50030875 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303176
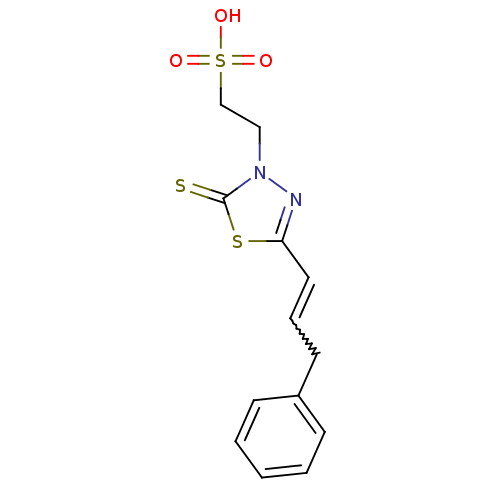
(2-((Z)-4-oxo-5-((E)-3-phenylallylidene)-2-thioxoth...)Show SMILES OS(=O)(=O)CCn1nc(C=CCc2ccccc2)sc1=S |w:10.10| Show InChI InChI=1S/C13H14N2O3S3/c16-21(17,18)10-9-15-13(19)20-12(14-15)8-4-7-11-5-2-1-3-6-11/h1-6,8H,7,9-10H2,(H,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 809 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303177
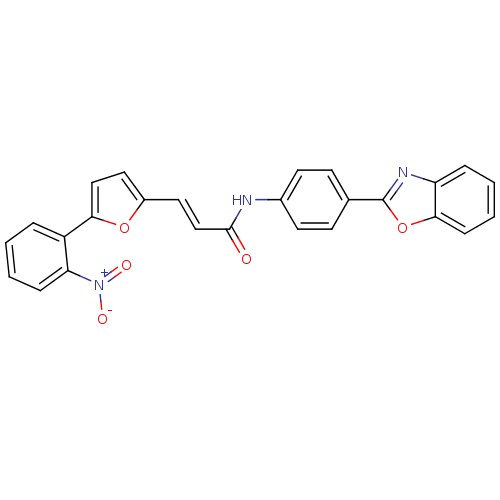
(CHEMBL565369 | N-(4-(benzo[d]oxazol-2-yl)phenyl)-3...)Show SMILES [O-][N+](=O)c1ccccc1-c1ccc(\C=C\C(=O)Nc2ccc(cc2)-c2nc3ccccc3o2)o1 Show InChI InChI=1S/C26H17N3O5/c30-25(16-14-19-13-15-23(33-19)20-5-1-3-7-22(20)29(31)32)27-18-11-9-17(10-12-18)26-28-21-6-2-4-8-24(21)34-26/h1-16H,(H,27,30)/b16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.76E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303178
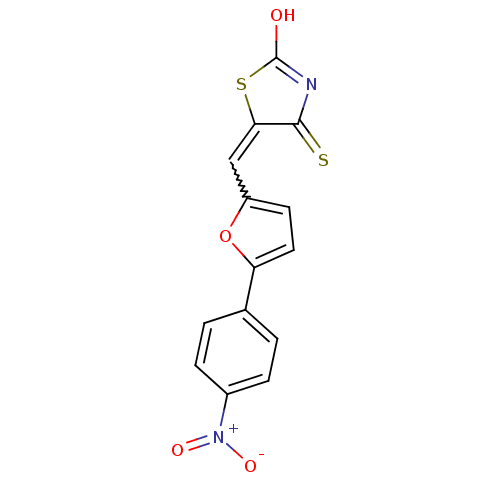
(5-((5-(4-nitrophenyl)furan-2-yl)methylene)-4-thiox...)Show SMILES OC1=NC(=S)C(S1)=Cc1ccc(o1)-c1ccc(cc1)[N+]([O-])=O |w:7.8,t:1| Show InChI InChI=1S/C14H8N2O4S2/c17-14-15-13(21)12(22-14)7-10-5-6-11(20-10)8-1-3-9(4-2-8)16(18)19/h1-7H,(H,15,17,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.86E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303179
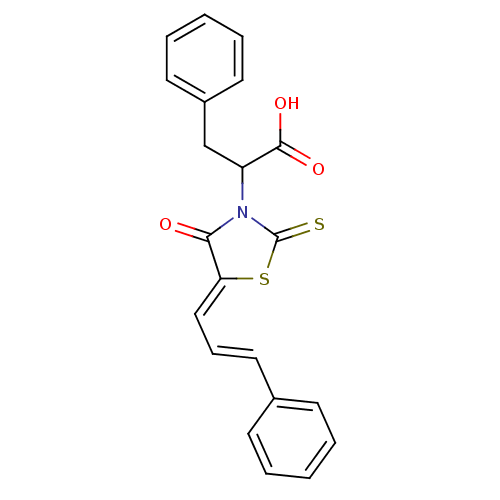
(2-(4-oxo-5-(3-phenylallylidene)-2-thioxothiazolidi...)Show SMILES OC(=O)C(Cc1ccccc1)N1C(=S)S\C(=C/C=C/c2ccccc2)C1=O Show InChI InChI=1S/C21H17NO3S2/c23-19-18(13-7-12-15-8-3-1-4-9-15)27-21(26)22(19)17(20(24)25)14-16-10-5-2-6-11-16/h1-13,17H,14H2,(H,24,25)/b12-7+,18-13- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.01E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM11994
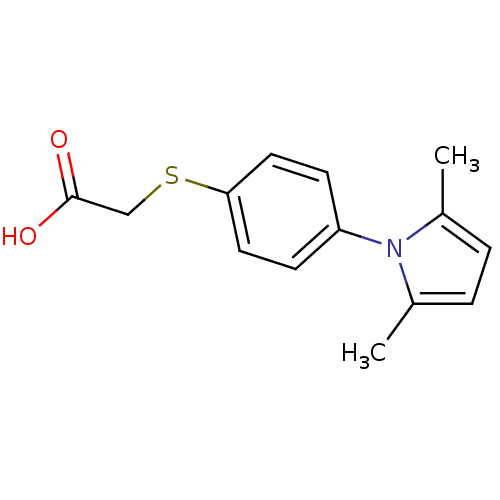
(2-{[4-(2,5-dimethyl-1H-pyrrol-1-yl)phenyl]sulfanyl...)Show InChI InChI=1S/C14H15NO2S/c1-10-3-4-11(2)15(10)12-5-7-13(8-6-12)18-9-14(16)17/h3-8H,9H2,1-2H3,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 2.09E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303180
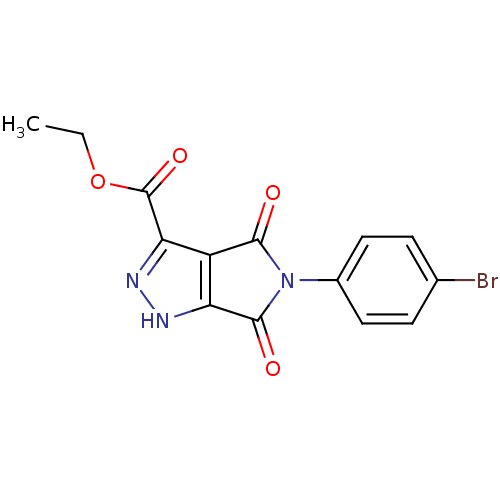
(CHEMBL577311 | ethyl 5-(4-bromophenyl)-4,6-dioxo-2...)Show SMILES CCOC(=O)c1n[nH]c2C(=O)N(C(=O)c12)c1ccc(Br)cc1 Show InChI InChI=1S/C14H10BrN3O4/c1-2-22-14(21)11-9-10(16-17-11)13(20)18(12(9)19)8-5-3-7(15)4-6-8/h3-6H,2H2,1H3,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.64E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303181

(1-(dibenzo[b,d]furan-3-yl)-3-(naphthalen-1-yl)thio...)Show InChI InChI=1S/C23H16N2OS/c27-23(25-20-10-5-7-15-6-1-2-8-17(15)20)24-16-12-13-19-18-9-3-4-11-21(18)26-22(19)14-16/h1-14H,(H2,24,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 2.94E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303182
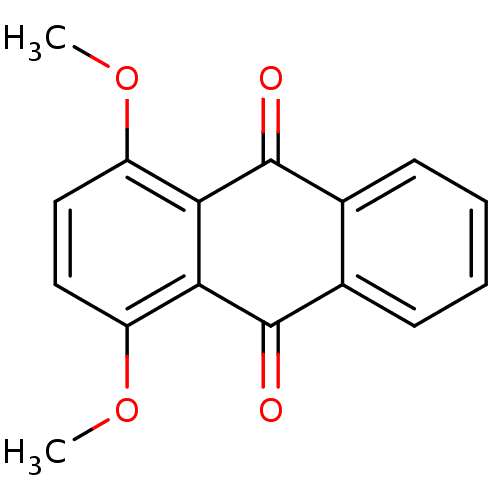
(1,4-dimethoxyanthracene-9,10-dione | CHEMBL570408 ...)Show InChI InChI=1S/C16H12O4/c1-19-11-7-8-12(20-2)14-13(11)15(17)9-5-3-4-6-10(9)16(14)18/h3-8H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.07E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM28851
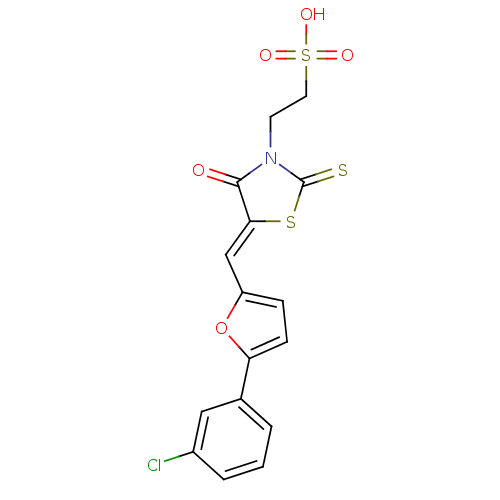
(2-[(5Z)-5-{[5-(3-chlorophenyl)furan-2-yl]methylide...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2ccc(o2)-c2cccc(Cl)c2)C1=O Show InChI InChI=1S/C16H12ClNO5S3/c17-11-3-1-2-10(8-11)13-5-4-12(23-13)9-14-15(19)18(16(24)25-14)6-7-26(20,21)22/h1-5,8-9H,6-7H2,(H,20,21,22)/b14-9- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.13E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303184
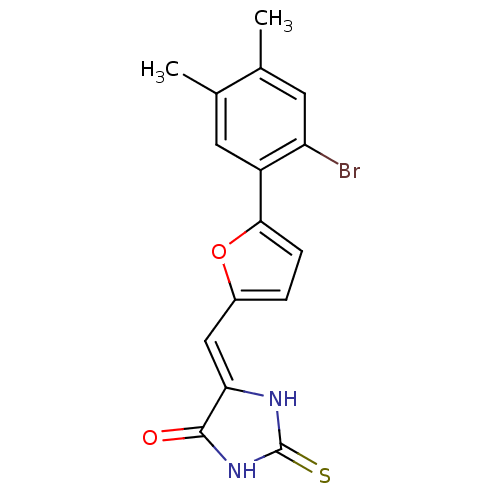
(5-((5-(2-bromo-4,5-dimethylphenyl)furan-2-yl)methy...)Show SMILES Cc1cc(Br)c(cc1C)-c1ccc(\C=C2/NC(=S)NC2=O)o1 Show InChI InChI=1S/C16H13BrN2O2S/c1-8-5-11(12(17)6-9(8)2)14-4-3-10(21-14)7-13-15(20)19-16(22)18-13/h3-7H,1-2H3,(H2,18,19,20,22)/b13-7- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.19E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303185
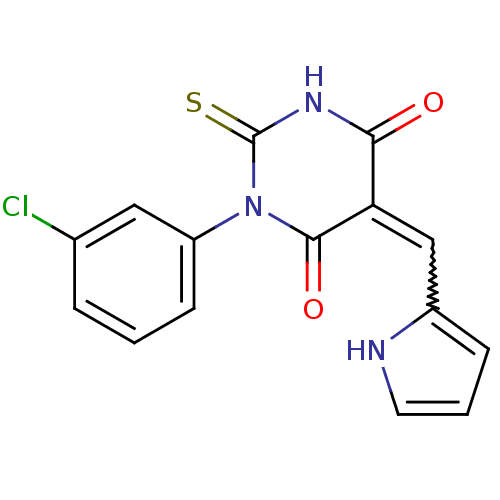
((E/Z)-5-((1H-pyrrol-2-yl)methylene)-1-(3-chlorophe...)Show SMILES Clc1cccc(c1)N1C(=S)NC(=O)C(=Cc2ccc[nH]2)C1=O |w:14.15| Show InChI InChI=1S/C15H10ClN3O2S/c16-9-3-1-5-11(7-9)19-14(21)12(13(20)18-15(19)22)8-10-4-2-6-17-10/h1-8,17H,(H,18,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 3.49E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM15186
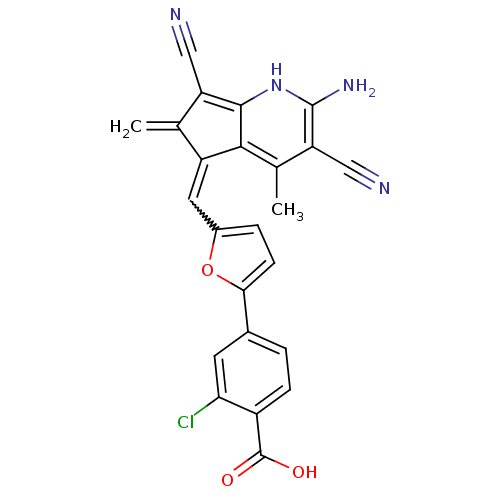
(4-(5-{[(5E)-2-amino-3,7-dicyano-4,6-dimethyl-5H-cy...)Show SMILES Cc1c(C#N)c(N)[nH]c2c(C#N)c(=C)c(=Cc3ccc(o3)-c3ccc(C(O)=O)c(Cl)c3)c12 |w:15.15| Show InChI InChI=1S/C24H15ClN4O3/c1-11-16(21-12(2)18(10-27)23(28)29-22(21)17(11)9-26)8-14-4-6-20(32-14)13-3-5-15(24(30)31)19(25)7-13/h3-8,29H,1,28H2,2H3,(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.87E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303186
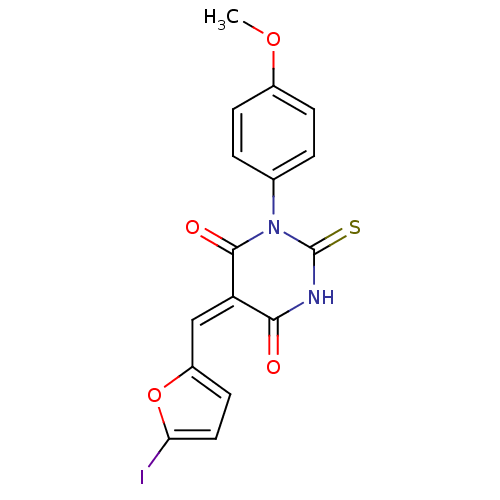
((E/Z)-5-((5-iodofuran-2-yl)methylene)-1-(4-methoxy...)Show SMILES COc1ccc(cc1)N1C(=S)NC(=O)\C(=C/c2ccc(I)o2)C1=O Show InChI InChI=1S/C16H11IN2O4S/c1-22-10-4-2-9(3-5-10)19-15(21)12(14(20)18-16(19)24)8-11-6-7-13(17)23-11/h2-8H,1H3,(H,18,20,24)/b12-8+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.44E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303187
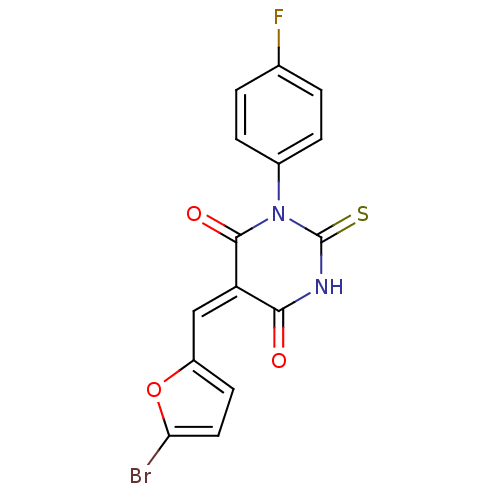
(5-((5-bromofuran-2-yl)methylene)-1-(4-fluorophenyl...)Show SMILES Fc1ccc(cc1)N1C(=S)NC(=O)\C(=C/c2ccc(Br)o2)C1=O Show InChI InChI=1S/C15H8BrFN2O3S/c16-12-6-5-10(22-12)7-11-13(20)18-15(23)19(14(11)21)9-3-1-8(17)2-4-9/h1-7H,(H,18,20,23)/b11-7+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.21E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM11985

(4-(2,5-dimethyl-1H-pyrrol-1-yl)-2-hydroxybenzoic a...)Show InChI InChI=1S/C13H13NO3/c1-8-3-4-9(2)14(8)10-5-6-11(13(16)17)12(15)7-10/h3-7,15H,1-2H3,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.28E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303188
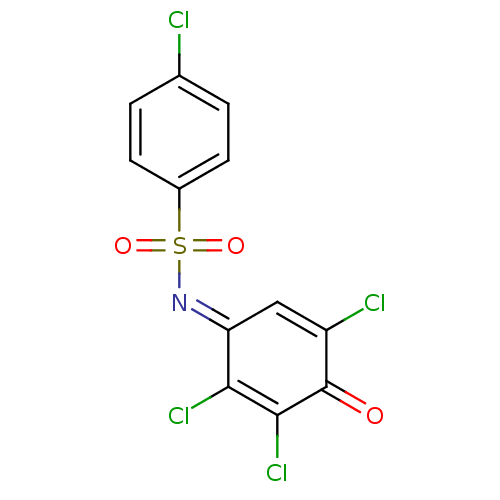
(4-chloro-N-(2,3,5-trichloro-4-oxocyclohexa-2,5-die...)Show SMILES ClC1=C\C(=N/S(=O)(=O)c2ccc(Cl)cc2)C(Cl)=C(Cl)C1=O |t:1,17| Show InChI InChI=1S/C12H5Cl4NO3S/c13-6-1-3-7(4-2-6)21(19,20)17-9-5-8(14)12(18)11(16)10(9)15/h1-5H/b17-9+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.34E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303189
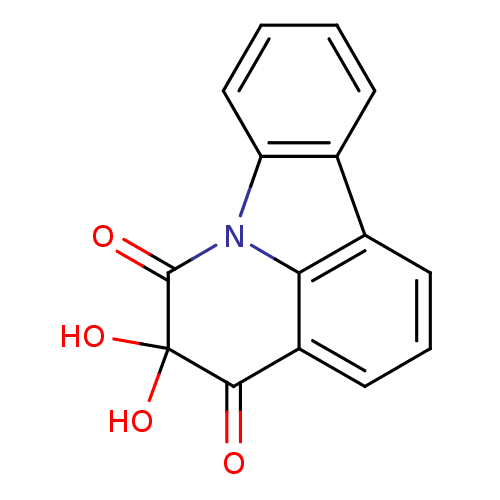
(5,5-dihydroxy-4H-pyrido[3,2,1-jk]carbazole-4,6(5H)...)Show InChI InChI=1S/C15H9NO4/c17-13-10-6-3-5-9-8-4-1-2-7-11(8)16(12(9)10)14(18)15(13,19)20/h1-7,19-20H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.38E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303190
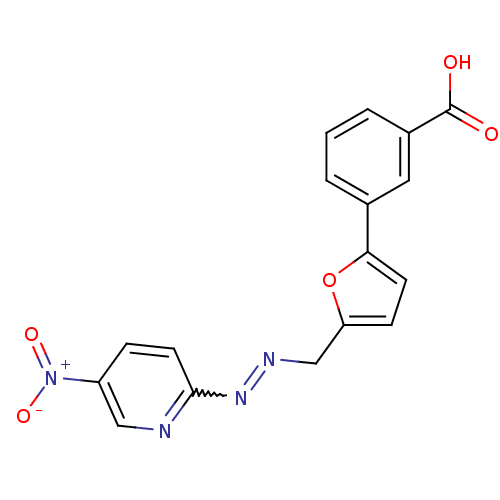
(3-(5-((2-(5-nitropyridin-2-yl)hydrazono)methyl)fur...)Show SMILES OC(=O)c1cccc(c1)-c1ccc(CN=Nc2ccc(cn2)[N+]([O-])=O)o1 |w:15.16| Show InChI InChI=1S/C17H12N4O5/c22-17(23)12-3-1-2-11(8-12)15-6-5-14(26-15)10-19-20-16-7-4-13(9-18-16)21(24)25/h1-9H,10H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.38E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303191
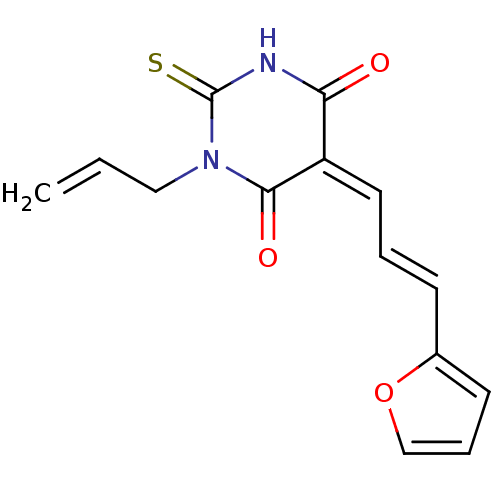
((E/Z)-1-allyl-5-(3-(furan-2-yl)allylidene)-2-thiox...)Show InChI InChI=1S/C14H12N2O3S/c1-2-8-16-13(18)11(12(17)15-14(16)20)7-3-5-10-6-4-9-19-10/h2-7,9H,1,8H2,(H,15,17,20)/b5-3+,11-7- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.58E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303192
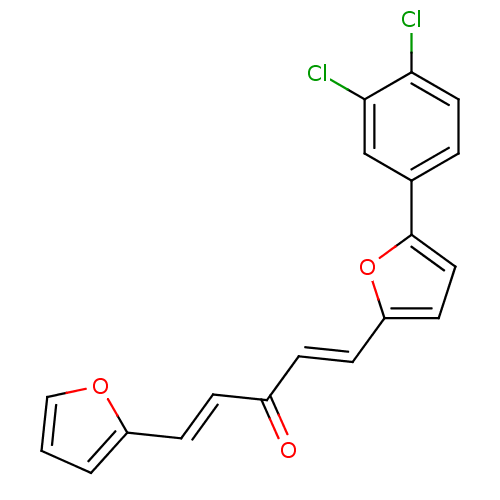
(1-(5-(3,4-dichlorophenyl)furan-2-yl)-5-(furan-2-yl...)Show SMILES Clc1ccc(cc1Cl)-c1ccc(\C=C\C(=O)\C=C\c2ccco2)o1 Show InChI InChI=1S/C19H12Cl2O3/c20-17-9-3-13(12-18(17)21)19-10-8-16(24-19)7-5-14(22)4-6-15-2-1-11-23-15/h1-12H/b6-4+,7-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.59E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303193
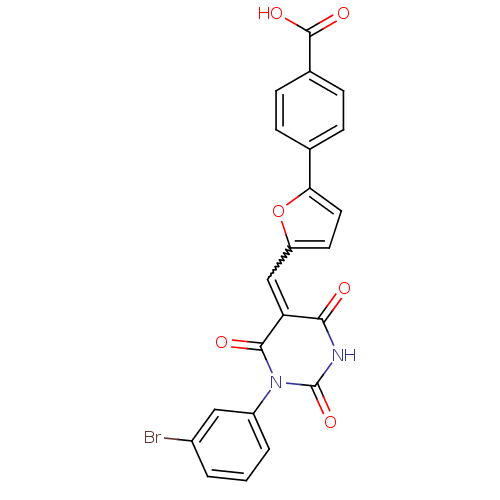
((E/Z)-4-(5-((1-(3-bromophenyl)-2,4,6-trioxotetrahy...)Show SMILES OC(=O)c1ccc(cc1)-c1ccc(C=C2C(=O)NC(=O)N(C2=O)c2cccc(Br)c2)o1 |w:13.13| Show InChI InChI=1S/C22H13BrN2O6/c23-14-2-1-3-15(10-14)25-20(27)17(19(26)24-22(25)30)11-16-8-9-18(31-16)12-4-6-13(7-5-12)21(28)29/h1-11H,(H,28,29)(H,24,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.62E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase expressed in Escherichia coli by Michaelis-Menton kinetic studies |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM28879

(2-[(5Z)-5-[(3-{4-[(4-chlorophenyl)methoxy]phenyl}-...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccc(Cl)cc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-22-10-6-19(7-11-22)18-37-24-12-8-20(9-13-24)26-21(17-32(30-26)23-4-2-1-3-5-23)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303173
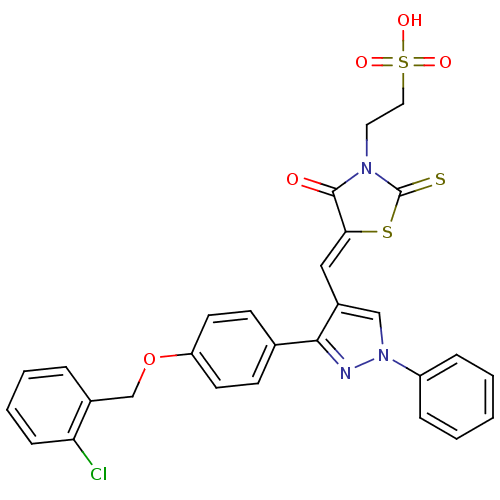
(2-(5-((3-(4-(2-chlorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3Cl)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 71 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303174

(2-(5-((3-(4-(2-fluorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3F)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22FN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 74 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50303175

(2-(5-((3-(4-(benzyloxy)phenyl)-1-phenyl-1H-pyrazol...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H23N3O5S3/c32-27-25(38-28(37)30(27)15-16-39(33,34)35)17-22-18-31(23-9-5-2-6-10-23)29-26(22)21-11-13-24(14-12-21)36-19-20-7-3-1-4-8-20/h1-14,17-18H,15-16,19H2,(H,33,34,35)/b25-17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM28884
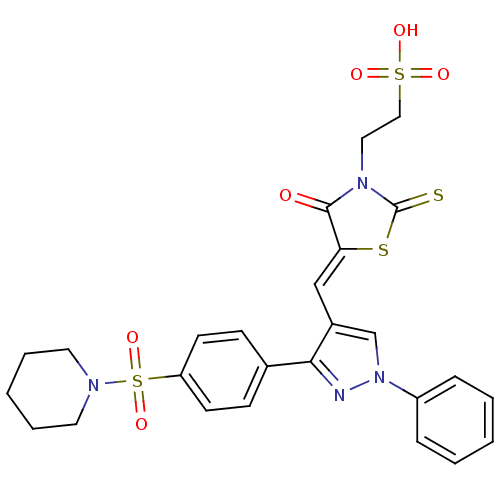
(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50303173

(2-(5-((3-(4-(2-chlorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3Cl)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant CD45 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50303174

(2-(5-((3-(4-(2-fluorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3F)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22FN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant PTP1B |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 1
(Homo sapiens (Human)) | BDBM28879

(2-[(5Z)-5-[(3-{4-[(4-chlorophenyl)methoxy]phenyl}-...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccc(Cl)cc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-22-10-6-19(7-11-22)18-37-24-12-8-20(9-13-24)26-21(17-32(30-26)23-4-2-1-3-5-23)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 460 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant MKP1 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM28879

(2-[(5Z)-5-[(3-{4-[(4-chlorophenyl)methoxy]phenyl}-...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccc(Cl)cc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-22-10-6-19(7-11-22)18-37-24-12-8-20(9-13-24)26-21(17-32(30-26)23-4-2-1-3-5-23)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 460 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant CD45 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM28879

(2-[(5Z)-5-[(3-{4-[(4-chlorophenyl)methoxy]phenyl}-...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccc(Cl)cc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-22-10-6-19(7-11-22)18-37-24-12-8-20(9-13-24)26-21(17-32(30-26)23-4-2-1-3-5-23)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 460 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant PTP1B |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 1
(Homo sapiens (Human)) | BDBM50303173

(2-(5-((3-(4-(2-chlorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3Cl)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 470 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant MKP1 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50303174

(2-(5-((3-(4-(2-fluorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3F)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22FN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant CD45 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 1
(Homo sapiens (Human)) | BDBM50303174

(2-(5-((3-(4-(2-fluorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3F)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22FN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 520 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant MKP1 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50303173

(2-(5-((3-(4-(2-chlorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3Cl)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 590 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant PTP1B |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50303175

(2-(5-((3-(4-(benzyloxy)phenyl)-1-phenyl-1H-pyrazol...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H23N3O5S3/c32-27-25(38-28(37)30(27)15-16-39(33,34)35)17-22-18-31(23-9-5-2-6-10-23)29-26(22)21-11-13-24(14-12-21)36-19-20-7-3-1-4-8-20/h1-14,17-18H,15-16,19H2,(H,33,34,35)/b25-17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 610 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant CD45 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 7
(Homo sapiens (Human)) | BDBM28879

(2-[(5Z)-5-[(3-{4-[(4-chlorophenyl)methoxy]phenyl}-...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccc(Cl)cc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-22-10-6-19(7-11-22)18-37-24-12-8-20(9-13-24)26-21(17-32(30-26)23-4-2-1-3-5-23)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 620 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant HePTP |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 1
(Homo sapiens (Human)) | BDBM50303175

(2-(5-((3-(4-(benzyloxy)phenyl)-1-phenyl-1H-pyrazol...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H23N3O5S3/c32-27-25(38-28(37)30(27)15-16-39(33,34)35)17-22-18-31(23-9-5-2-6-10-23)29-26(22)21-11-13-24(14-12-21)36-19-20-7-3-1-4-8-20/h1-14,17-18H,15-16,19H2,(H,33,34,35)/b25-17- | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 780 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant MKP1 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 7
(Homo sapiens (Human)) | BDBM50303174

(2-(5-((3-(4-(2-fluorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3F)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22FN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 870 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant HePTP |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 7
(Homo sapiens (Human)) | BDBM50303173

(2-(5-((3-(4-(2-chlorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3Cl)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant HePTP |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 7
(Homo sapiens (Human)) | BDBM50303175

(2-(5-((3-(4-(benzyloxy)phenyl)-1-phenyl-1H-pyrazol...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H23N3O5S3/c32-27-25(38-28(37)30(27)15-16-39(33,34)35)17-22-18-31(23-9-5-2-6-10-23)29-26(22)21-11-13-24(14-12-21)36-19-20-7-3-1-4-8-20/h1-14,17-18H,15-16,19H2,(H,33,34,35)/b25-17- | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant HePTP |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM28881
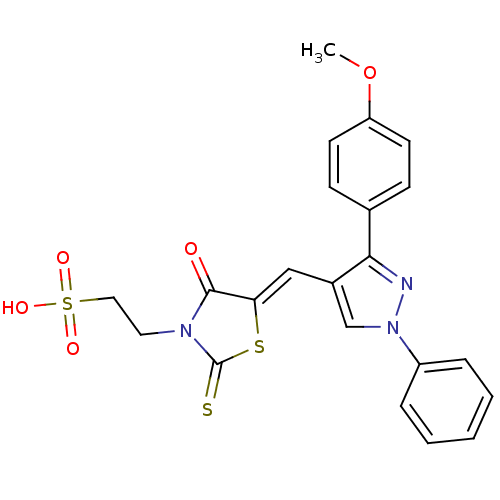
(2-[(5Z)-5-{[3-(4-methoxyphenyl)-1-phenyl-1H-pyrazo...)Show SMILES COc1ccc(cc1)-c1nn(cc1\C=C1/SC(=S)N(CCS(O)(=O)=O)C1=O)-c1ccccc1 Show InChI InChI=1S/C22H19N3O5S3/c1-30-18-9-7-15(8-10-18)20-16(14-25(23-20)17-5-3-2-4-6-17)13-19-21(26)24(22(31)32-19)11-12-33(27,28)29/h2-10,13-14H,11-12H2,1H3,(H,27,28,29)/b19-13- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM28882
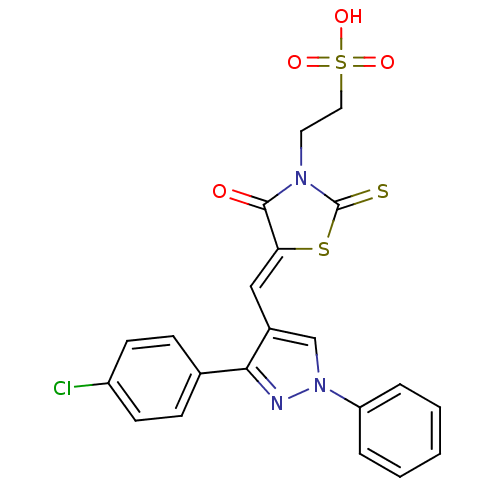
(2-[(5Z)-5-{[3-(4-chlorophenyl)-1-phenyl-1H-pyrazol...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(Cl)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C21H16ClN3O4S3/c22-16-8-6-14(7-9-16)19-15(13-25(23-19)17-4-2-1-3-5-17)12-18-20(26)24(21(30)31-18)10-11-32(27,28)29/h1-9,12-13H,10-11H2,(H,27,28,29)/b18-12- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant CD45 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50303175

(2-(5-((3-(4-(benzyloxy)phenyl)-1-phenyl-1H-pyrazol...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H23N3O5S3/c32-27-25(38-28(37)30(27)15-16-39(33,34)35)17-22-18-31(23-9-5-2-6-10-23)29-26(22)21-11-13-24(14-12-21)36-19-20-7-3-1-4-8-20/h1-14,17-18H,15-16,19H2,(H,33,34,35)/b25-17- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant PTP1B |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant PTP1B |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
M-phase inducer phosphatase 1
(Homo sapiens (Human)) | BDBM28879

(2-[(5Z)-5-[(3-{4-[(4-chlorophenyl)methoxy]phenyl}-...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccc(Cl)cc3)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22ClN3O5S3/c29-22-10-6-19(7-11-22)18-37-24-12-8-20(9-13-24)26-21(17-32(30-26)23-4-2-1-3-5-23)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Cdc25a |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 7
(Homo sapiens (Human)) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant HePTP |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM28883
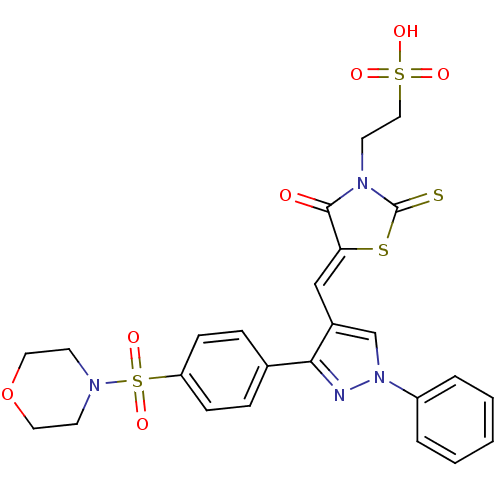
(2-[(5Z)-5-({3-[4-(morpholine-4-sulfonyl)phenyl]-1-...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCOCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C25H24N4O7S4/c30-24-22(38-25(37)28(24)12-15-39(31,32)33)16-19-17-29(20-4-2-1-3-5-20)26-23(19)18-6-8-21(9-7-18)40(34,35)27-10-13-36-14-11-27/h1-9,16-17H,10-15H2,(H,31,32,33)/b22-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
M-phase inducer phosphatase 1
(Homo sapiens (Human)) | BDBM50303174

(2-(5-((3-(4-(2-fluorobenzyloxy)phenyl)-1-phenyl-1H...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(OCc3ccccc3F)cc2)-c2ccccc2)C1=O Show InChI InChI=1S/C28H22FN3O5S3/c29-24-9-5-4-6-20(24)18-37-23-12-10-19(11-13-23)26-21(17-32(30-26)22-7-2-1-3-8-22)16-25-27(33)31(28(38)39-25)14-15-40(34,35)36/h1-13,16-17H,14-15,18H2,(H,34,35,36)/b25-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Cdc25a |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data