

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50214492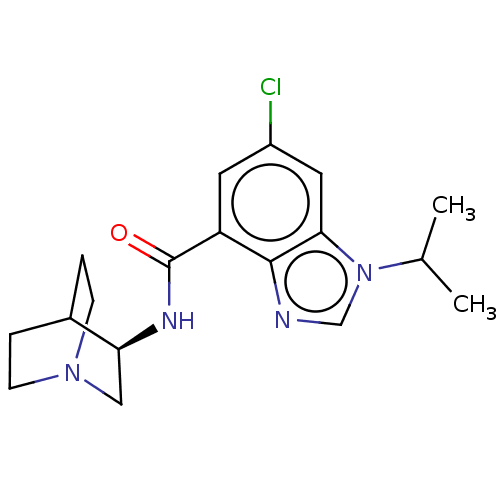 (CHEMBL145725 | RS-16566) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | 0.145 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of [3H](R)-zacopride from ondansetron-treated NG-108-15 cell membranes | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50056419 (4-Amino-N-(1-aza-bicyclo[2.2.2]oct-3-yl)-5-chloro-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | 0.182 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of [3H]-BRL 43694 from NG-108-15 cell membranes | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50214492 (CHEMBL145725 | RS-16566) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | 0.537 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of [3H]-BRL 43694 from NG-108-15 cell membranes | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50214494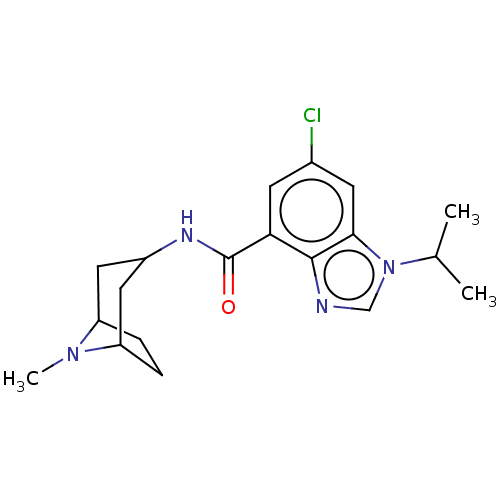 (CHEMBL142852 | RS-33800) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of [3H]-BRL 43694 from NG-108-15 cell membranes | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A/3B (Rattus norvegicus-RAT) | BDBM50031475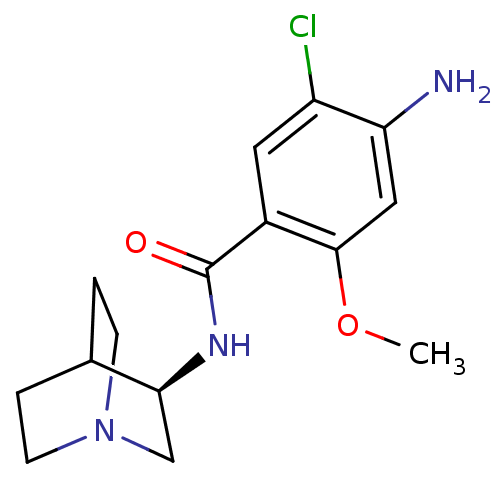 (4-Amino-N-(1-aza-bicyclo[2.2.2]oct-3-yl)-5-chloro-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of racemic [3H]zacopride from rat cortex | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50031475 (4-Amino-N-(1-aza-bicyclo[2.2.2]oct-3-yl)-5-chloro-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of [3H](R)-zacopride from ondansetron-treated NG-108-15 cell membranes | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50214494 (CHEMBL142852 | RS-33800) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of [3H](R)-zacopride from ondansetron-treated NG-108-15 cell membranes | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 4 (GUINEA PIG) | BDBM50214492 (CHEMBL145725 | RS-16566) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 4 receptor by displacement of [3H]GR-113808 from guinea pig brain striatum. | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 4 (GUINEA PIG) | BDBM50214494 (CHEMBL142852 | RS-33800) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | 158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 4 receptor by displacement of [3H]GR-113808 from guinea pig brain striatum. | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 4 (GUINEA PIG) | BDBM50056419 (4-Amino-N-(1-aza-bicyclo[2.2.2]oct-3-yl)-5-chloro-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | 437 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 4 receptor by displacement of [3H]GR-113808 from guinea pig brain striatum. | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50214493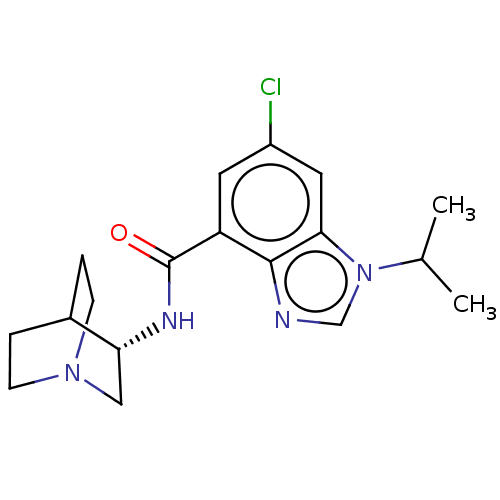 (CHEMBL145033 | RS-16456) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | 602 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of [3H](R)-zacopride from ondansetron-treated NG-108-15 cell membranes | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50214493 (CHEMBL145033 | RS-16456) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | 603 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of [3H](R)-zacopride from ondansetron-treated NG-108-15 cell membranes | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 4 (GUINEA PIG) | BDBM50031475 (4-Amino-N-(1-aza-bicyclo[2.2.2]oct-3-yl)-5-chloro-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | 2.82E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 4 receptor by displacement of [3H]GR-113808 from guinea pig brain striatum. | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50056419 (4-Amino-N-(1-aza-bicyclo[2.2.2]oct-3-yl)-5-chloro-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | 5.01E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity for 5-hydroxytryptamine 3 receptor by displacement of [3H](R)-zacopride from ondansetron-treated NG-108-15 cell membranes | Citation and Details BindingDB Entry DOI: 10.7270/Q2QZ2D49 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||