 Found 49 hits Enz. Inhib. hit(s) with all data for entry = 50034534
Found 49 hits Enz. Inhib. hit(s) with all data for entry = 50034534 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363942
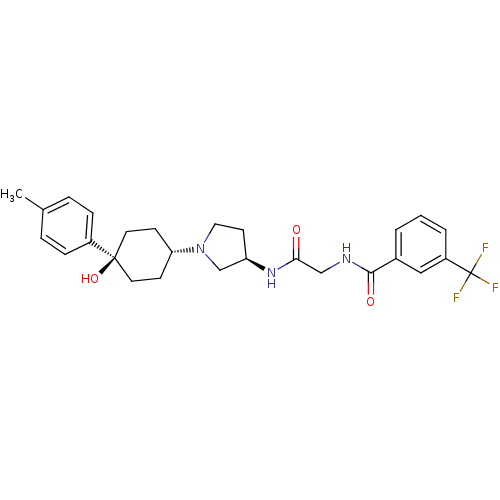
(CHEMBL1951766)Show SMILES Cc1ccc(cc1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-10.43,2.36,;-8.89,2.39,;-8.15,3.74,;-6.61,3.78,;-5.81,2.46,;-6.55,1.1,;-8.1,1.07,;-4.27,2.5,;-5.05,3.84,;-3.46,1.18,;-1.93,1.21,;-1.18,2.56,;-1.98,3.88,;-3.53,3.85,;.36,2.59,;1.28,1.37,;2.73,1.87,;2.7,3.41,;1.23,3.86,;4.03,4.2,;5.37,3.45,;5.39,1.91,;6.7,4.23,;8.04,3.48,;9.36,4.27,;9.34,5.81,;10.71,3.51,;10.72,1.97,;12.06,1.22,;13.38,2.01,;13.36,3.55,;12.02,4.3,;14.68,4.35,;16.03,3.6,;14.66,5.89,;16.01,5.13,)| Show InChI InChI=1S/C27H32F3N3O3/c1-18-5-7-20(8-6-18)26(36)12-9-23(10-13-26)33-14-11-22(17-33)32-24(34)16-31-25(35)19-3-2-4-21(15-19)27(28,29)30/h2-8,15,22-23,36H,9-14,16-17H2,1H3,(H,31,35)(H,32,34)/t22-,23-,26-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363942

(CHEMBL1951766)Show SMILES Cc1ccc(cc1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-10.43,2.36,;-8.89,2.39,;-8.15,3.74,;-6.61,3.78,;-5.81,2.46,;-6.55,1.1,;-8.1,1.07,;-4.27,2.5,;-5.05,3.84,;-3.46,1.18,;-1.93,1.21,;-1.18,2.56,;-1.98,3.88,;-3.53,3.85,;.36,2.59,;1.28,1.37,;2.73,1.87,;2.7,3.41,;1.23,3.86,;4.03,4.2,;5.37,3.45,;5.39,1.91,;6.7,4.23,;8.04,3.48,;9.36,4.27,;9.34,5.81,;10.71,3.51,;10.72,1.97,;12.06,1.22,;13.38,2.01,;13.36,3.55,;12.02,4.3,;14.68,4.35,;16.03,3.6,;14.66,5.89,;16.01,5.13,)| Show InChI InChI=1S/C27H32F3N3O3/c1-18-5-7-20(8-6-18)26(36)12-9-23(10-13-26)33-14-11-22(17-33)32-24(34)16-31-25(35)19-3-2-4-21(15-19)27(28,29)30/h2-8,15,22-23,36H,9-14,16-17H2,1H3,(H,31,35)(H,32,34)/t22-,23-,26-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363952
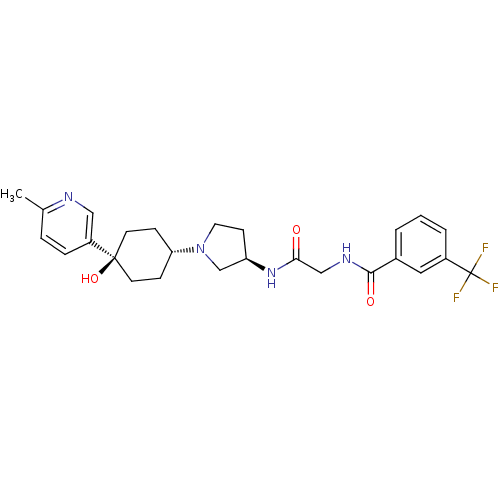
(CHEMBL1951777)Show SMILES Cc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-9.17,-25.25,;-7.63,-25.22,;-6.89,-23.87,;-5.35,-23.83,;-4.55,-25.15,;-5.29,-26.51,;-6.84,-26.54,;-3.01,-25.11,;-3.79,-23.77,;-2.2,-26.43,;-.67,-26.4,;.08,-25.05,;-.72,-23.73,;-2.27,-23.76,;1.62,-25.02,;2.54,-26.24,;3.99,-25.74,;3.96,-24.2,;2.49,-23.75,;5.29,-23.41,;6.63,-24.17,;6.65,-25.71,;7.96,-23.38,;9.3,-24.13,;10.62,-23.35,;10.6,-21.81,;11.97,-24.1,;11.98,-25.64,;13.32,-26.39,;14.64,-25.6,;14.62,-24.06,;13.28,-23.31,;15.94,-23.27,;17.29,-24.01,;15.92,-21.73,;17.27,-22.49,)| Show InChI InChI=1S/C26H31F3N4O3/c1-17-5-6-20(14-30-17)25(36)10-7-22(8-11-25)33-12-9-21(16-33)32-23(34)15-31-24(35)18-3-2-4-19(13-18)26(27,28)29/h2-6,13-14,21-22,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t21-,22-,25-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363944
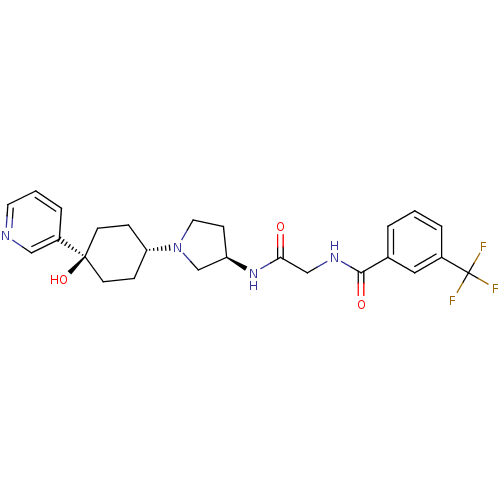
(CHEMBL1951769)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cccnc1 |r,wU:4.7,wD:10.13,1.0,(-5.98,-12.63,;-5.2,-13.97,;-4.4,-15.29,;-2.86,-15.26,;-2.12,-13.91,;-2.92,-12.59,;-4.46,-12.62,;-.58,-13.88,;.35,-15.1,;1.8,-14.6,;1.77,-13.06,;.3,-12.61,;3.09,-12.27,;4.44,-13.03,;4.46,-14.57,;5.76,-12.24,;7.1,-12.99,;8.43,-12.21,;8.41,-10.67,;9.77,-12.96,;9.78,-14.5,;11.12,-15.25,;12.45,-14.47,;12.43,-12.92,;11.08,-12.17,;13.75,-12.13,;15.09,-12.88,;13.72,-10.59,;15.07,-11.35,;-6.74,-14.01,;-7.54,-12.7,;-9.08,-12.73,;-9.83,-14.08,;-9.03,-15.4,;-7.49,-15.37,)| Show InChI InChI=1S/C25H29F3N4O3/c26-25(27,28)18-4-1-3-17(13-18)23(34)30-15-22(33)31-20-8-12-32(16-20)21-6-9-24(35,10-7-21)19-5-2-11-29-14-19/h1-5,11,13-14,20-21,35H,6-10,12,15-16H2,(H,30,34)(H,31,33)/t20-,21-,24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human CCR2-mediated Erk phosphorylation |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363954
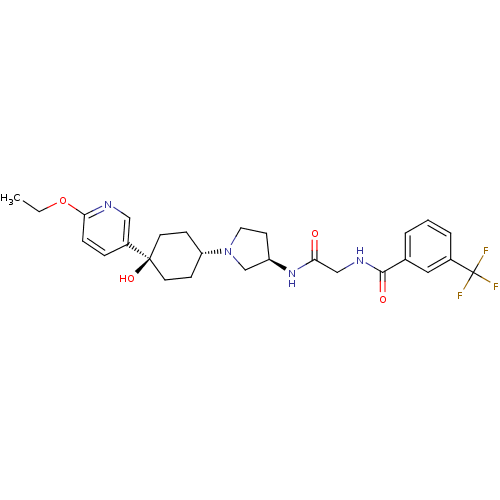
(CHEMBL1951779)Show SMILES CCOc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:13.17,wD:19.23,9.10,(-12.06,-40.23,;-10.52,-40.2,;-9.72,-41.52,;-8.18,-41.49,;-7.44,-40.14,;-5.9,-40.1,;-5.1,-41.42,;-5.85,-42.78,;-7.39,-42.81,;-3.56,-41.38,;-4.34,-40.04,;-2.75,-42.7,;-1.22,-42.67,;-.48,-41.32,;-1.27,-40,;-2.82,-40.03,;1.06,-41.29,;1.99,-42.51,;3.44,-42.01,;3.41,-40.47,;1.94,-40.02,;4.74,-39.68,;6.08,-40.44,;6.1,-41.98,;7.4,-39.65,;8.75,-40.4,;10.07,-39.62,;10.05,-38.08,;11.41,-40.37,;11.42,-41.91,;12.77,-42.66,;14.09,-41.88,;14.07,-40.33,;12.72,-39.58,;15.39,-39.54,;16.73,-40.28,;15.36,-38,;16.71,-38.76,)| Show InChI InChI=1S/C27H33F3N4O4/c1-2-38-24-7-6-20(15-31-24)26(37)11-8-22(9-12-26)34-13-10-21(17-34)33-23(35)16-32-25(36)18-4-3-5-19(14-18)27(28,29)30/h3-7,14-15,21-22,37H,2,8-13,16-17H2,1H3,(H,32,36)(H,33,35)/t21-,22-,26-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363945
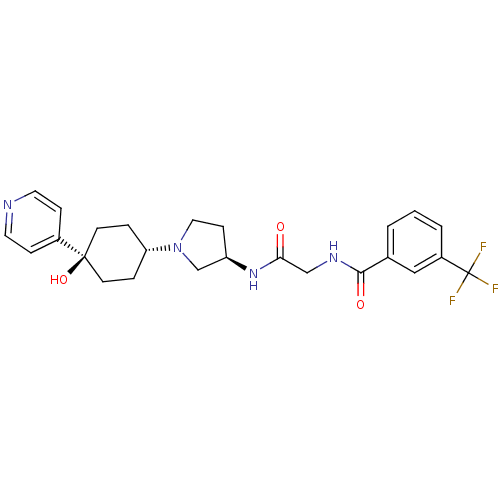
(CHEMBL1951770)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccncc1 |r,wU:4.7,wD:10.13,1.0,(-5.8,-20.26,;-5.02,-21.6,;-4.22,-22.92,;-2.68,-22.89,;-1.94,-21.54,;-2.74,-20.22,;-4.28,-20.25,;-.4,-21.51,;.52,-22.73,;1.98,-22.23,;1.95,-20.69,;.48,-20.24,;3.27,-19.9,;4.62,-20.66,;4.64,-22.2,;5.94,-19.87,;7.28,-20.62,;8.61,-19.84,;8.59,-18.3,;9.95,-20.59,;9.96,-22.13,;11.3,-22.88,;12.63,-22.1,;12.6,-20.55,;11.26,-19.8,;13.93,-19.76,;15.27,-20.51,;13.9,-18.22,;15.25,-18.98,;-6.56,-21.64,;-7.36,-20.33,;-8.9,-20.36,;-9.65,-21.71,;-8.85,-23.03,;-7.31,-23,)| Show InChI InChI=1S/C25H29F3N4O3/c26-25(27,28)19-3-1-2-17(14-19)23(34)30-15-22(33)31-20-8-13-32(16-20)21-4-9-24(35,10-5-21)18-6-11-29-12-7-18/h1-3,6-7,11-12,14,20-21,35H,4-5,8-10,13,15-16H2,(H,30,34)(H,31,33)/t20-,21-,24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363943
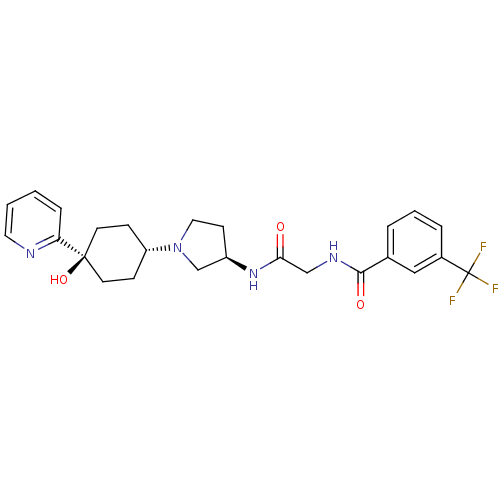
(CHEMBL1951768)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccccn1 |r,wU:4.7,wD:10.13,1.0,(-5.87,-4.88,;-5.09,-6.22,;-4.29,-7.54,;-2.75,-7.5,;-2.01,-6.16,;-2.81,-4.84,;-4.35,-4.87,;-.47,-6.13,;.45,-7.35,;1.91,-6.85,;1.88,-5.31,;.41,-4.86,;3.2,-4.52,;4.55,-5.27,;4.57,-6.81,;5.87,-4.49,;7.21,-5.24,;8.54,-4.45,;8.52,-2.91,;9.88,-5.21,;9.89,-6.75,;11.23,-7.5,;12.56,-6.71,;12.53,-5.16,;11.19,-4.42,;13.86,-4.37,;15.2,-5.12,;13.83,-2.83,;15.18,-3.59,;-6.63,-6.26,;-7.43,-4.94,;-8.97,-4.97,;-9.72,-6.33,;-8.92,-7.64,;-7.38,-7.62,)| Show InChI InChI=1S/C25H29F3N4O3/c26-25(27,28)18-5-3-4-17(14-18)23(34)30-15-22(33)31-19-9-13-32(16-19)20-7-10-24(35,11-8-20)21-6-1-2-12-29-21/h1-6,12,14,19-20,35H,7-11,13,15-16H2,(H,30,34)(H,31,33)/t19-,20-,24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363943

(CHEMBL1951768)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccccn1 |r,wU:4.7,wD:10.13,1.0,(-5.87,-4.88,;-5.09,-6.22,;-4.29,-7.54,;-2.75,-7.5,;-2.01,-6.16,;-2.81,-4.84,;-4.35,-4.87,;-.47,-6.13,;.45,-7.35,;1.91,-6.85,;1.88,-5.31,;.41,-4.86,;3.2,-4.52,;4.55,-5.27,;4.57,-6.81,;5.87,-4.49,;7.21,-5.24,;8.54,-4.45,;8.52,-2.91,;9.88,-5.21,;9.89,-6.75,;11.23,-7.5,;12.56,-6.71,;12.53,-5.16,;11.19,-4.42,;13.86,-4.37,;15.2,-5.12,;13.83,-2.83,;15.18,-3.59,;-6.63,-6.26,;-7.43,-4.94,;-8.97,-4.97,;-9.72,-6.33,;-8.92,-7.64,;-7.38,-7.62,)| Show InChI InChI=1S/C25H29F3N4O3/c26-25(27,28)18-5-3-4-17(14-18)23(34)30-15-22(33)31-19-9-13-32(16-19)20-7-10-24(35,11-8-20)21-6-1-2-12-29-21/h1-6,12,14,19-20,35H,7-11,13,15-16H2,(H,30,34)(H,31,33)/t19-,20-,24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363954

(CHEMBL1951779)Show SMILES CCOc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:13.17,wD:19.23,9.10,(-12.06,-40.23,;-10.52,-40.2,;-9.72,-41.52,;-8.18,-41.49,;-7.44,-40.14,;-5.9,-40.1,;-5.1,-41.42,;-5.85,-42.78,;-7.39,-42.81,;-3.56,-41.38,;-4.34,-40.04,;-2.75,-42.7,;-1.22,-42.67,;-.48,-41.32,;-1.27,-40,;-2.82,-40.03,;1.06,-41.29,;1.99,-42.51,;3.44,-42.01,;3.41,-40.47,;1.94,-40.02,;4.74,-39.68,;6.08,-40.44,;6.1,-41.98,;7.4,-39.65,;8.75,-40.4,;10.07,-39.62,;10.05,-38.08,;11.41,-40.37,;11.42,-41.91,;12.77,-42.66,;14.09,-41.88,;14.07,-40.33,;12.72,-39.58,;15.39,-39.54,;16.73,-40.28,;15.36,-38,;16.71,-38.76,)| Show InChI InChI=1S/C27H33F3N4O4/c1-2-38-24-7-6-20(15-31-24)26(37)11-8-22(9-12-26)34-13-10-21(17-34)33-23(35)16-32-25(36)18-4-3-5-19(14-18)27(28,29)30/h3-7,14-15,21-22,37H,2,8-13,16-17H2,1H3,(H,32,36)(H,33,35)/t21-,22-,26-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363944

(CHEMBL1951769)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cccnc1 |r,wU:4.7,wD:10.13,1.0,(-5.98,-12.63,;-5.2,-13.97,;-4.4,-15.29,;-2.86,-15.26,;-2.12,-13.91,;-2.92,-12.59,;-4.46,-12.62,;-.58,-13.88,;.35,-15.1,;1.8,-14.6,;1.77,-13.06,;.3,-12.61,;3.09,-12.27,;4.44,-13.03,;4.46,-14.57,;5.76,-12.24,;7.1,-12.99,;8.43,-12.21,;8.41,-10.67,;9.77,-12.96,;9.78,-14.5,;11.12,-15.25,;12.45,-14.47,;12.43,-12.92,;11.08,-12.17,;13.75,-12.13,;15.09,-12.88,;13.72,-10.59,;15.07,-11.35,;-6.74,-14.01,;-7.54,-12.7,;-9.08,-12.73,;-9.83,-14.08,;-9.03,-15.4,;-7.49,-15.37,)| Show InChI InChI=1S/C25H29F3N4O3/c26-25(27,28)18-4-1-3-17(13-18)23(34)30-15-22(33)31-20-8-12-32(16-20)21-6-9-24(35,10-7-21)19-5-2-11-29-14-19/h1-5,11,13-14,20-21,35H,6-10,12,15-16H2,(H,30,34)(H,31,33)/t20-,21-,24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363945

(CHEMBL1951770)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccncc1 |r,wU:4.7,wD:10.13,1.0,(-5.8,-20.26,;-5.02,-21.6,;-4.22,-22.92,;-2.68,-22.89,;-1.94,-21.54,;-2.74,-20.22,;-4.28,-20.25,;-.4,-21.51,;.52,-22.73,;1.98,-22.23,;1.95,-20.69,;.48,-20.24,;3.27,-19.9,;4.62,-20.66,;4.64,-22.2,;5.94,-19.87,;7.28,-20.62,;8.61,-19.84,;8.59,-18.3,;9.95,-20.59,;9.96,-22.13,;11.3,-22.88,;12.63,-22.1,;12.6,-20.55,;11.26,-19.8,;13.93,-19.76,;15.27,-20.51,;13.9,-18.22,;15.25,-18.98,;-6.56,-21.64,;-7.36,-20.33,;-8.9,-20.36,;-9.65,-21.71,;-8.85,-23.03,;-7.31,-23,)| Show InChI InChI=1S/C25H29F3N4O3/c26-25(27,28)19-3-1-2-17(14-19)23(34)30-15-22(33)31-20-8-13-32(16-20)21-4-9-24(35,10-5-21)18-6-11-29-12-7-18/h1-3,6-7,11-12,14,20-21,35H,4-5,8-10,13,15-16H2,(H,30,34)(H,31,33)/t20-,21-,24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363950
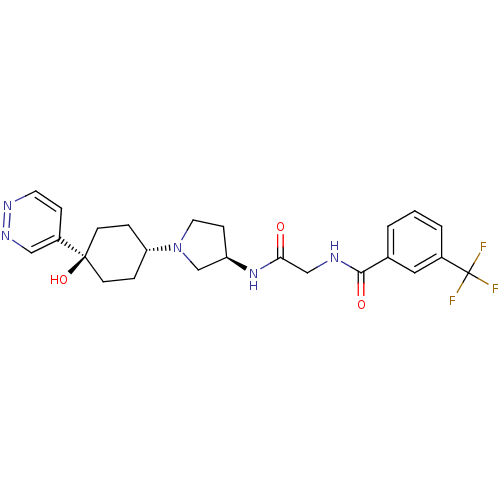
(CHEMBL1951775)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccnnc1 |r,wU:4.7,wD:10.13,1.0,(-4.71,-6.18,;-3.93,-7.52,;-3.12,-8.84,;-1.59,-8.8,;-.84,-7.46,;-1.64,-6.14,;-3.18,-6.17,;.7,-7.43,;1.62,-8.65,;3.08,-8.14,;3.05,-6.61,;1.57,-6.16,;4.37,-5.82,;5.71,-6.57,;5.73,-8.11,;7.04,-5.79,;8.38,-6.54,;9.7,-5.75,;9.69,-4.21,;11.05,-6.51,;11.06,-8.04,;12.4,-8.8,;13.73,-8.01,;13.7,-6.46,;12.36,-5.72,;15.02,-5.67,;16.37,-6.42,;15,-4.13,;16.35,-4.89,;-5.47,-7.55,;-6.27,-6.24,;-7.8,-6.27,;-8.55,-7.62,;-7.75,-8.94,;-6.21,-8.91,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(12-17)22(34)28-14-21(33)31-19-7-11-32(15-19)20-4-8-23(35,9-5-20)18-6-10-29-30-13-18/h1-3,6,10,12-13,19-20,35H,4-5,7-9,11,14-15H2,(H,28,34)(H,31,33)/t19-,20-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363952

(CHEMBL1951777)Show SMILES Cc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-9.17,-25.25,;-7.63,-25.22,;-6.89,-23.87,;-5.35,-23.83,;-4.55,-25.15,;-5.29,-26.51,;-6.84,-26.54,;-3.01,-25.11,;-3.79,-23.77,;-2.2,-26.43,;-.67,-26.4,;.08,-25.05,;-.72,-23.73,;-2.27,-23.76,;1.62,-25.02,;2.54,-26.24,;3.99,-25.74,;3.96,-24.2,;2.49,-23.75,;5.29,-23.41,;6.63,-24.17,;6.65,-25.71,;7.96,-23.38,;9.3,-24.13,;10.62,-23.35,;10.6,-21.81,;11.97,-24.1,;11.98,-25.64,;13.32,-26.39,;14.64,-25.6,;14.62,-24.06,;13.28,-23.31,;15.94,-23.27,;17.29,-24.01,;15.92,-21.73,;17.27,-22.49,)| Show InChI InChI=1S/C26H31F3N4O3/c1-17-5-6-20(14-30-17)25(36)10-7-22(8-11-25)33-12-9-21(16-33)32-23(34)15-31-24(35)18-3-2-4-19(13-18)26(27,28)29/h2-6,13-14,21-22,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t21-,22-,25-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363949
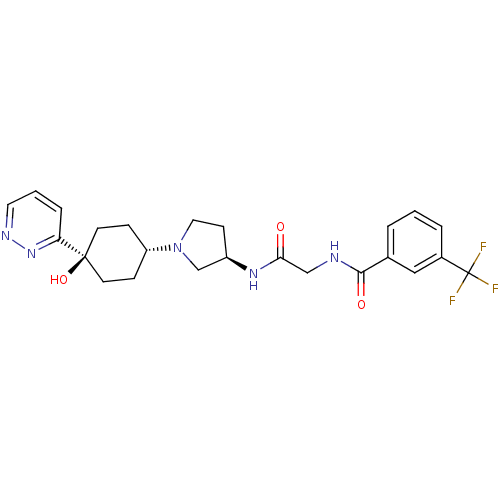
(CHEMBL1951774)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cccnn1 |r,wU:4.7,wD:10.13,1.0,(-4.96,2.61,;-4.18,1.27,;-3.38,-.05,;-1.84,-.01,;-1.1,1.33,;-1.9,2.65,;-3.44,2.62,;.44,1.36,;1.36,.14,;2.82,.64,;2.79,2.18,;1.32,2.63,;4.11,2.97,;5.46,2.22,;5.48,.68,;6.78,3,;8.12,2.25,;9.45,3.04,;9.43,4.58,;10.79,2.28,;10.8,.74,;12.14,-.01,;13.47,.78,;13.44,2.33,;12.1,3.07,;14.77,3.12,;16.11,2.37,;14.74,4.66,;16.09,3.9,;-5.72,1.23,;-6.52,2.55,;-8.06,2.52,;-8.81,1.16,;-8.01,-.15,;-6.47,-.13,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-4-1-3-16(13-17)22(34)28-14-21(33)30-18-8-12-32(15-18)19-6-9-23(35,10-7-19)20-5-2-11-29-31-20/h1-5,11,13,18-19,35H,6-10,12,14-15H2,(H,28,34)(H,30,33)/t18-,19-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human CCR2-mediated intracellular calcium mobilization by cell based assay |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363950

(CHEMBL1951775)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccnnc1 |r,wU:4.7,wD:10.13,1.0,(-4.71,-6.18,;-3.93,-7.52,;-3.12,-8.84,;-1.59,-8.8,;-.84,-7.46,;-1.64,-6.14,;-3.18,-6.17,;.7,-7.43,;1.62,-8.65,;3.08,-8.14,;3.05,-6.61,;1.57,-6.16,;4.37,-5.82,;5.71,-6.57,;5.73,-8.11,;7.04,-5.79,;8.38,-6.54,;9.7,-5.75,;9.69,-4.21,;11.05,-6.51,;11.06,-8.04,;12.4,-8.8,;13.73,-8.01,;13.7,-6.46,;12.36,-5.72,;15.02,-5.67,;16.37,-6.42,;15,-4.13,;16.35,-4.89,;-5.47,-7.55,;-6.27,-6.24,;-7.8,-6.27,;-8.55,-7.62,;-7.75,-8.94,;-6.21,-8.91,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(12-17)22(34)28-14-21(33)31-19-7-11-32(15-19)20-4-8-23(35,9-5-20)18-6-10-29-30-13-18/h1-3,6,10,12-13,19-20,35H,4-5,7-9,11,14-15H2,(H,28,34)(H,31,33)/t19-,20-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363949

(CHEMBL1951774)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cccnn1 |r,wU:4.7,wD:10.13,1.0,(-4.96,2.61,;-4.18,1.27,;-3.38,-.05,;-1.84,-.01,;-1.1,1.33,;-1.9,2.65,;-3.44,2.62,;.44,1.36,;1.36,.14,;2.82,.64,;2.79,2.18,;1.32,2.63,;4.11,2.97,;5.46,2.22,;5.48,.68,;6.78,3,;8.12,2.25,;9.45,3.04,;9.43,4.58,;10.79,2.28,;10.8,.74,;12.14,-.01,;13.47,.78,;13.44,2.33,;12.1,3.07,;14.77,3.12,;16.11,2.37,;14.74,4.66,;16.09,3.9,;-5.72,1.23,;-6.52,2.55,;-8.06,2.52,;-8.81,1.16,;-8.01,-.15,;-6.47,-.13,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-4-1-3-16(13-17)22(34)28-14-21(33)30-18-8-12-32(15-18)19-6-9-23(35,10-7-19)20-5-2-11-29-31-20/h1-5,11,13,18-19,35H,6-10,12,14-15H2,(H,28,34)(H,30,33)/t18-,19-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363947
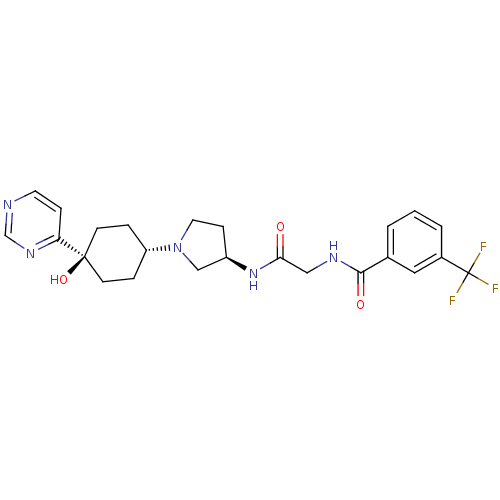
(CHEMBL1951772)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccncn1 |r,wU:4.7,wD:10.13,1.0,(-5.51,-34.96,;-4.74,-36.3,;-3.93,-37.62,;-2.4,-37.59,;-1.65,-36.24,;-2.45,-34.92,;-3.99,-34.95,;-.11,-36.21,;.81,-37.43,;2.27,-36.93,;2.24,-35.39,;.76,-34.94,;3.56,-34.6,;4.9,-35.36,;4.92,-36.9,;6.23,-34.57,;7.57,-35.32,;8.9,-34.54,;8.88,-33,;10.24,-35.29,;10.25,-36.83,;11.59,-37.58,;12.92,-36.8,;12.89,-35.25,;11.55,-34.5,;14.21,-34.46,;15.56,-35.21,;14.19,-32.92,;15.54,-33.68,;-6.28,-36.34,;-7.08,-35.03,;-8.61,-35.06,;-9.36,-36.41,;-8.56,-37.73,;-7.02,-37.7,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(12-17)22(34)29-13-21(33)31-18-7-11-32(14-18)19-4-8-23(35,9-5-19)20-6-10-28-15-30-20/h1-3,6,10,12,15,18-19,35H,4-5,7-9,11,13-14H2,(H,29,34)(H,31,33)/t18-,19-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363947

(CHEMBL1951772)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccncn1 |r,wU:4.7,wD:10.13,1.0,(-5.51,-34.96,;-4.74,-36.3,;-3.93,-37.62,;-2.4,-37.59,;-1.65,-36.24,;-2.45,-34.92,;-3.99,-34.95,;-.11,-36.21,;.81,-37.43,;2.27,-36.93,;2.24,-35.39,;.76,-34.94,;3.56,-34.6,;4.9,-35.36,;4.92,-36.9,;6.23,-34.57,;7.57,-35.32,;8.9,-34.54,;8.88,-33,;10.24,-35.29,;10.25,-36.83,;11.59,-37.58,;12.92,-36.8,;12.89,-35.25,;11.55,-34.5,;14.21,-34.46,;15.56,-35.21,;14.19,-32.92,;15.54,-33.68,;-6.28,-36.34,;-7.08,-35.03,;-8.61,-35.06,;-9.36,-36.41,;-8.56,-37.73,;-7.02,-37.7,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(12-17)22(34)29-13-21(33)31-18-7-11-32(14-18)19-4-8-23(35,9-5-19)20-6-10-28-15-30-20/h1-3,6,10,12,15,18-19,35H,4-5,7-9,11,13-14H2,(H,29,34)(H,31,33)/t18-,19-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363946
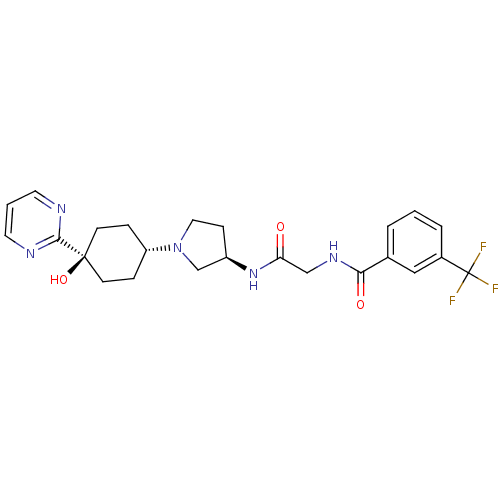
(CHEMBL1951771)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ncccn1 |r,wU:4.7,wD:10.13,1.0,(-5.61,-27.39,;-4.83,-28.74,;-4.02,-30.05,;-2.49,-30.02,;-1.74,-28.67,;-2.54,-27.35,;-4.09,-27.38,;-.2,-28.64,;.72,-29.87,;2.17,-29.36,;2.14,-27.82,;.67,-27.38,;3.47,-27.04,;4.81,-27.79,;4.83,-29.33,;6.14,-27,;7.48,-27.76,;8.8,-26.97,;8.78,-25.43,;10.15,-27.72,;10.16,-29.26,;11.5,-30.02,;12.82,-29.23,;12.8,-27.68,;11.46,-26.93,;14.12,-26.89,;15.47,-27.64,;14.1,-25.35,;15.45,-26.11,;-6.37,-28.77,;-7.17,-27.46,;-8.71,-27.49,;-9.45,-28.84,;-8.66,-30.16,;-7.11,-30.13,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-4-1-3-16(13-17)21(34)30-14-20(33)31-18-7-12-32(15-18)19-5-8-23(35,9-6-19)22-28-10-2-11-29-22/h1-4,10-11,13,18-19,35H,5-9,12,14-15H2,(H,30,34)(H,31,33)/t18-,19-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363946

(CHEMBL1951771)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ncccn1 |r,wU:4.7,wD:10.13,1.0,(-5.61,-27.39,;-4.83,-28.74,;-4.02,-30.05,;-2.49,-30.02,;-1.74,-28.67,;-2.54,-27.35,;-4.09,-27.38,;-.2,-28.64,;.72,-29.87,;2.17,-29.36,;2.14,-27.82,;.67,-27.38,;3.47,-27.04,;4.81,-27.79,;4.83,-29.33,;6.14,-27,;7.48,-27.76,;8.8,-26.97,;8.78,-25.43,;10.15,-27.72,;10.16,-29.26,;11.5,-30.02,;12.82,-29.23,;12.8,-27.68,;11.46,-26.93,;14.12,-26.89,;15.47,-27.64,;14.1,-25.35,;15.45,-26.11,;-6.37,-28.77,;-7.17,-27.46,;-8.71,-27.49,;-9.45,-28.84,;-8.66,-30.16,;-7.11,-30.13,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-4-1-3-16(13-17)21(34)30-14-20(33)31-18-7-12-32(15-18)19-5-8-23(35,9-6-19)22-28-10-2-11-29-22/h1-4,10-11,13,18-19,35H,5-9,12,14-15H2,(H,30,34)(H,31,33)/t18-,19-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10.3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363951
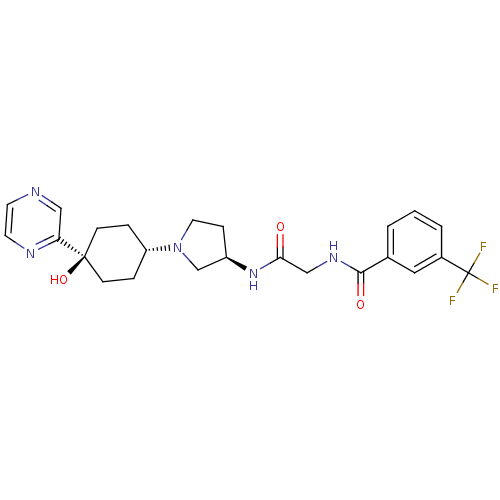
(CHEMBL1951776)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cnccn1 |r,wU:4.7,wD:10.13,1.0,(-5.05,-16.35,;-4.27,-17.69,;-3.46,-19.01,;-1.93,-18.98,;-1.18,-17.63,;-1.98,-16.31,;-3.53,-16.34,;.36,-17.6,;1.28,-18.82,;2.73,-18.32,;2.7,-16.78,;1.23,-16.33,;4.03,-15.99,;5.37,-16.75,;5.39,-18.29,;6.7,-15.96,;8.04,-16.71,;9.36,-15.93,;9.34,-14.39,;10.71,-16.68,;10.72,-18.22,;12.06,-18.97,;13.38,-18.18,;13.36,-16.64,;12.02,-15.89,;14.68,-15.85,;16.03,-16.59,;14.66,-14.31,;16.01,-15.07,;-5.81,-17.73,;-6.61,-16.41,;-8.15,-16.45,;-8.89,-17.8,;-8.1,-19.12,;-6.55,-19.09,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(12-17)22(34)30-14-21(33)31-18-6-11-32(15-18)19-4-7-23(35,8-5-19)20-13-28-9-10-29-20/h1-3,9-10,12-13,18-19,35H,4-8,11,14-15H2,(H,30,34)(H,31,33)/t18-,19-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10.4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363951

(CHEMBL1951776)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cnccn1 |r,wU:4.7,wD:10.13,1.0,(-5.05,-16.35,;-4.27,-17.69,;-3.46,-19.01,;-1.93,-18.98,;-1.18,-17.63,;-1.98,-16.31,;-3.53,-16.34,;.36,-17.6,;1.28,-18.82,;2.73,-18.32,;2.7,-16.78,;1.23,-16.33,;4.03,-15.99,;5.37,-16.75,;5.39,-18.29,;6.7,-15.96,;8.04,-16.71,;9.36,-15.93,;9.34,-14.39,;10.71,-16.68,;10.72,-18.22,;12.06,-18.97,;13.38,-18.18,;13.36,-16.64,;12.02,-15.89,;14.68,-15.85,;16.03,-16.59,;14.66,-14.31,;16.01,-15.07,;-5.81,-17.73,;-6.61,-16.41,;-8.15,-16.45,;-8.89,-17.8,;-8.1,-19.12,;-6.55,-19.09,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(12-17)22(34)30-14-21(33)31-18-6-11-32(15-18)19-4-7-23(35,8-5-19)20-13-28-9-10-29-20/h1-3,9-10,12-13,18-19,35H,4-8,11,14-15H2,(H,30,34)(H,31,33)/t18-,19-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10.9 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363948

(CHEMBL1951773)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cncnc1 |r,wU:4.7,wD:10.13,1.0,(-5.3,-43.56,;-4.53,-44.91,;-3.72,-46.22,;-2.19,-46.19,;-1.44,-44.84,;-2.24,-43.52,;-3.78,-43.55,;.1,-44.81,;1.02,-46.04,;2.48,-45.53,;2.45,-43.99,;.97,-43.55,;3.77,-43.21,;5.11,-43.96,;5.13,-45.5,;6.44,-43.17,;7.78,-43.93,;9.11,-43.14,;9.09,-41.6,;10.45,-43.89,;10.46,-45.43,;11.8,-46.19,;13.13,-45.4,;13.1,-43.85,;11.76,-43.1,;14.42,-43.06,;15.77,-43.81,;14.4,-41.52,;15.75,-42.28,;-6.07,-44.94,;-6.81,-46.3,;-8.35,-46.33,;-9.15,-45.01,;-8.4,-43.66,;-6.87,-43.63,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(10-17)22(34)30-13-21(33)31-19-6-9-32(14-19)20-4-7-23(35,8-5-20)18-11-28-15-29-12-18/h1-3,10-12,15,19-20,35H,4-9,13-14H2,(H,30,34)(H,31,33)/t19-,20-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Mus musculus) | BDBM50363942

(CHEMBL1951766)Show SMILES Cc1ccc(cc1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-10.43,2.36,;-8.89,2.39,;-8.15,3.74,;-6.61,3.78,;-5.81,2.46,;-6.55,1.1,;-8.1,1.07,;-4.27,2.5,;-5.05,3.84,;-3.46,1.18,;-1.93,1.21,;-1.18,2.56,;-1.98,3.88,;-3.53,3.85,;.36,2.59,;1.28,1.37,;2.73,1.87,;2.7,3.41,;1.23,3.86,;4.03,4.2,;5.37,3.45,;5.39,1.91,;6.7,4.23,;8.04,3.48,;9.36,4.27,;9.34,5.81,;10.71,3.51,;10.72,1.97,;12.06,1.22,;13.38,2.01,;13.36,3.55,;12.02,4.3,;14.68,4.35,;16.03,3.6,;14.66,5.89,;16.01,5.13,)| Show InChI InChI=1S/C27H32F3N3O3/c1-18-5-7-20(8-6-18)26(36)12-9-23(10-13-26)33-14-11-22(17-33)32-24(34)16-31-25(35)19-3-2-4-21(15-19)27(28,29)30/h2-8,15,22-23,36H,9-14,16-17H2,1H3,(H,31,35)(H,32,34)/t22-,23-,26-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from mouse CCR2 after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363948

(CHEMBL1951773)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cncnc1 |r,wU:4.7,wD:10.13,1.0,(-5.3,-43.56,;-4.53,-44.91,;-3.72,-46.22,;-2.19,-46.19,;-1.44,-44.84,;-2.24,-43.52,;-3.78,-43.55,;.1,-44.81,;1.02,-46.04,;2.48,-45.53,;2.45,-43.99,;.97,-43.55,;3.77,-43.21,;5.11,-43.96,;5.13,-45.5,;6.44,-43.17,;7.78,-43.93,;9.11,-43.14,;9.09,-41.6,;10.45,-43.89,;10.46,-45.43,;11.8,-46.19,;13.13,-45.4,;13.1,-43.85,;11.76,-43.1,;14.42,-43.06,;15.77,-43.81,;14.4,-41.52,;15.75,-42.28,;-6.07,-44.94,;-6.81,-46.3,;-8.35,-46.33,;-9.15,-45.01,;-8.4,-43.66,;-6.87,-43.63,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(10-17)22(34)30-13-21(33)31-19-6-9-32(14-19)20-4-7-23(35,8-5-20)18-11-28-15-29-12-18/h1-3,10-12,15,19-20,35H,4-9,13-14H2,(H,30,34)(H,31,33)/t19-,20-,23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363945

(CHEMBL1951770)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccncc1 |r,wU:4.7,wD:10.13,1.0,(-5.8,-20.26,;-5.02,-21.6,;-4.22,-22.92,;-2.68,-22.89,;-1.94,-21.54,;-2.74,-20.22,;-4.28,-20.25,;-.4,-21.51,;.52,-22.73,;1.98,-22.23,;1.95,-20.69,;.48,-20.24,;3.27,-19.9,;4.62,-20.66,;4.64,-22.2,;5.94,-19.87,;7.28,-20.62,;8.61,-19.84,;8.59,-18.3,;9.95,-20.59,;9.96,-22.13,;11.3,-22.88,;12.63,-22.1,;12.6,-20.55,;11.26,-19.8,;13.93,-19.76,;15.27,-20.51,;13.9,-18.22,;15.25,-18.98,;-6.56,-21.64,;-7.36,-20.33,;-8.9,-20.36,;-9.65,-21.71,;-8.85,-23.03,;-7.31,-23,)| Show InChI InChI=1S/C25H29F3N4O3/c26-25(27,28)19-3-1-2-17(14-19)23(34)30-15-22(33)31-20-8-13-32(16-20)21-4-9-24(35,10-5-21)18-6-11-29-12-7-18/h1-3,6-7,11-12,14,20-21,35H,4-5,8-10,13,15-16H2,(H,30,34)(H,31,33)/t20-,21-,24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363944

(CHEMBL1951769)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cccnc1 |r,wU:4.7,wD:10.13,1.0,(-5.98,-12.63,;-5.2,-13.97,;-4.4,-15.29,;-2.86,-15.26,;-2.12,-13.91,;-2.92,-12.59,;-4.46,-12.62,;-.58,-13.88,;.35,-15.1,;1.8,-14.6,;1.77,-13.06,;.3,-12.61,;3.09,-12.27,;4.44,-13.03,;4.46,-14.57,;5.76,-12.24,;7.1,-12.99,;8.43,-12.21,;8.41,-10.67,;9.77,-12.96,;9.78,-14.5,;11.12,-15.25,;12.45,-14.47,;12.43,-12.92,;11.08,-12.17,;13.75,-12.13,;15.09,-12.88,;13.72,-10.59,;15.07,-11.35,;-6.74,-14.01,;-7.54,-12.7,;-9.08,-12.73,;-9.83,-14.08,;-9.03,-15.4,;-7.49,-15.37,)| Show InChI InChI=1S/C25H29F3N4O3/c26-25(27,28)18-4-1-3-17(13-18)23(34)30-15-22(33)31-20-8-12-32(16-20)21-6-9-24(35,10-7-21)19-5-2-11-29-14-19/h1-5,11,13-14,20-21,35H,6-10,12,15-16H2,(H,30,34)(H,31,33)/t20-,21-,24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363943

(CHEMBL1951768)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccccn1 |r,wU:4.7,wD:10.13,1.0,(-5.87,-4.88,;-5.09,-6.22,;-4.29,-7.54,;-2.75,-7.5,;-2.01,-6.16,;-2.81,-4.84,;-4.35,-4.87,;-.47,-6.13,;.45,-7.35,;1.91,-6.85,;1.88,-5.31,;.41,-4.86,;3.2,-4.52,;4.55,-5.27,;4.57,-6.81,;5.87,-4.49,;7.21,-5.24,;8.54,-4.45,;8.52,-2.91,;9.88,-5.21,;9.89,-6.75,;11.23,-7.5,;12.56,-6.71,;12.53,-5.16,;11.19,-4.42,;13.86,-4.37,;15.2,-5.12,;13.83,-2.83,;15.18,-3.59,;-6.63,-6.26,;-7.43,-4.94,;-8.97,-4.97,;-9.72,-6.33,;-8.92,-7.64,;-7.38,-7.62,)| Show InChI InChI=1S/C25H29F3N4O3/c26-25(27,28)18-5-3-4-17(14-18)23(34)30-15-22(33)31-19-9-13-32(16-19)20-7-10-24(35,11-8-20)21-6-1-2-12-29-21/h1-6,12,14,19-20,35H,7-11,13,15-16H2,(H,30,34)(H,31,33)/t19-,20-,24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363946

(CHEMBL1951771)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ncccn1 |r,wU:4.7,wD:10.13,1.0,(-5.61,-27.39,;-4.83,-28.74,;-4.02,-30.05,;-2.49,-30.02,;-1.74,-28.67,;-2.54,-27.35,;-4.09,-27.38,;-.2,-28.64,;.72,-29.87,;2.17,-29.36,;2.14,-27.82,;.67,-27.38,;3.47,-27.04,;4.81,-27.79,;4.83,-29.33,;6.14,-27,;7.48,-27.76,;8.8,-26.97,;8.78,-25.43,;10.15,-27.72,;10.16,-29.26,;11.5,-30.02,;12.82,-29.23,;12.8,-27.68,;11.46,-26.93,;14.12,-26.89,;15.47,-27.64,;14.1,-25.35,;15.45,-26.11,;-6.37,-28.77,;-7.17,-27.46,;-8.71,-27.49,;-9.45,-28.84,;-8.66,-30.16,;-7.11,-30.13,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-4-1-3-16(13-17)21(34)30-14-20(33)31-18-7-12-32(15-18)19-5-8-23(35,9-6-19)22-28-10-2-11-29-22/h1-4,10-11,13,18-19,35H,5-9,12,14-15H2,(H,30,34)(H,31,33)/t18-,19-,23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363954

(CHEMBL1951779)Show SMILES CCOc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:13.17,wD:19.23,9.10,(-12.06,-40.23,;-10.52,-40.2,;-9.72,-41.52,;-8.18,-41.49,;-7.44,-40.14,;-5.9,-40.1,;-5.1,-41.42,;-5.85,-42.78,;-7.39,-42.81,;-3.56,-41.38,;-4.34,-40.04,;-2.75,-42.7,;-1.22,-42.67,;-.48,-41.32,;-1.27,-40,;-2.82,-40.03,;1.06,-41.29,;1.99,-42.51,;3.44,-42.01,;3.41,-40.47,;1.94,-40.02,;4.74,-39.68,;6.08,-40.44,;6.1,-41.98,;7.4,-39.65,;8.75,-40.4,;10.07,-39.62,;10.05,-38.08,;11.41,-40.37,;11.42,-41.91,;12.77,-42.66,;14.09,-41.88,;14.07,-40.33,;12.72,-39.58,;15.39,-39.54,;16.73,-40.28,;15.36,-38,;16.71,-38.76,)| Show InChI InChI=1S/C27H33F3N4O4/c1-2-38-24-7-6-20(15-31-24)26(37)11-8-22(9-12-26)34-13-10-21(17-34)33-23(35)16-32-25(36)18-4-3-5-19(14-18)27(28,29)30/h3-7,14-15,21-22,37H,2,8-13,16-17H2,1H3,(H,32,36)(H,33,35)/t21-,22-,26-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363952

(CHEMBL1951777)Show SMILES Cc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-9.17,-25.25,;-7.63,-25.22,;-6.89,-23.87,;-5.35,-23.83,;-4.55,-25.15,;-5.29,-26.51,;-6.84,-26.54,;-3.01,-25.11,;-3.79,-23.77,;-2.2,-26.43,;-.67,-26.4,;.08,-25.05,;-.72,-23.73,;-2.27,-23.76,;1.62,-25.02,;2.54,-26.24,;3.99,-25.74,;3.96,-24.2,;2.49,-23.75,;5.29,-23.41,;6.63,-24.17,;6.65,-25.71,;7.96,-23.38,;9.3,-24.13,;10.62,-23.35,;10.6,-21.81,;11.97,-24.1,;11.98,-25.64,;13.32,-26.39,;14.64,-25.6,;14.62,-24.06,;13.28,-23.31,;15.94,-23.27,;17.29,-24.01,;15.92,-21.73,;17.27,-22.49,)| Show InChI InChI=1S/C26H31F3N4O3/c1-17-5-6-20(14-30-17)25(36)10-7-22(8-11-25)33-12-9-21(16-33)32-23(34)15-31-24(35)18-3-2-4-19(13-18)26(27,28)29/h2-6,13-14,21-22,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t21-,22-,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363951

(CHEMBL1951776)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cnccn1 |r,wU:4.7,wD:10.13,1.0,(-5.05,-16.35,;-4.27,-17.69,;-3.46,-19.01,;-1.93,-18.98,;-1.18,-17.63,;-1.98,-16.31,;-3.53,-16.34,;.36,-17.6,;1.28,-18.82,;2.73,-18.32,;2.7,-16.78,;1.23,-16.33,;4.03,-15.99,;5.37,-16.75,;5.39,-18.29,;6.7,-15.96,;8.04,-16.71,;9.36,-15.93,;9.34,-14.39,;10.71,-16.68,;10.72,-18.22,;12.06,-18.97,;13.38,-18.18,;13.36,-16.64,;12.02,-15.89,;14.68,-15.85,;16.03,-16.59,;14.66,-14.31,;16.01,-15.07,;-5.81,-17.73,;-6.61,-16.41,;-8.15,-16.45,;-8.89,-17.8,;-8.1,-19.12,;-6.55,-19.09,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(12-17)22(34)30-14-21(33)31-18-6-11-32(15-18)19-4-7-23(35,8-5-19)20-13-28-9-10-29-20/h1-3,9-10,12-13,18-19,35H,4-8,11,14-15H2,(H,30,34)(H,31,33)/t18-,19-,23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363950

(CHEMBL1951775)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccnnc1 |r,wU:4.7,wD:10.13,1.0,(-4.71,-6.18,;-3.93,-7.52,;-3.12,-8.84,;-1.59,-8.8,;-.84,-7.46,;-1.64,-6.14,;-3.18,-6.17,;.7,-7.43,;1.62,-8.65,;3.08,-8.14,;3.05,-6.61,;1.57,-6.16,;4.37,-5.82,;5.71,-6.57,;5.73,-8.11,;7.04,-5.79,;8.38,-6.54,;9.7,-5.75,;9.69,-4.21,;11.05,-6.51,;11.06,-8.04,;12.4,-8.8,;13.73,-8.01,;13.7,-6.46,;12.36,-5.72,;15.02,-5.67,;16.37,-6.42,;15,-4.13,;16.35,-4.89,;-5.47,-7.55,;-6.27,-6.24,;-7.8,-6.27,;-8.55,-7.62,;-7.75,-8.94,;-6.21,-8.91,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(12-17)22(34)28-14-21(33)31-19-7-11-32(15-19)20-4-8-23(35,9-5-20)18-6-10-29-30-13-18/h1-3,6,10,12-13,19-20,35H,4-5,7-9,11,14-15H2,(H,28,34)(H,31,33)/t19-,20-,23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363949

(CHEMBL1951774)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1cccnn1 |r,wU:4.7,wD:10.13,1.0,(-4.96,2.61,;-4.18,1.27,;-3.38,-.05,;-1.84,-.01,;-1.1,1.33,;-1.9,2.65,;-3.44,2.62,;.44,1.36,;1.36,.14,;2.82,.64,;2.79,2.18,;1.32,2.63,;4.11,2.97,;5.46,2.22,;5.48,.68,;6.78,3,;8.12,2.25,;9.45,3.04,;9.43,4.58,;10.79,2.28,;10.8,.74,;12.14,-.01,;13.47,.78,;13.44,2.33,;12.1,3.07,;14.77,3.12,;16.11,2.37,;14.74,4.66,;16.09,3.9,;-5.72,1.23,;-6.52,2.55,;-8.06,2.52,;-8.81,1.16,;-8.01,-.15,;-6.47,-.13,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-4-1-3-16(13-17)22(34)28-14-21(33)30-18-8-12-32(15-18)19-6-9-23(35,10-7-19)20-5-2-11-29-31-20/h1-5,11,13,18-19,35H,6-10,12,14-15H2,(H,28,34)(H,30,33)/t18-,19-,23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363947

(CHEMBL1951772)Show SMILES O[C@]1(CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccncn1 |r,wU:4.7,wD:10.13,1.0,(-5.51,-34.96,;-4.74,-36.3,;-3.93,-37.62,;-2.4,-37.59,;-1.65,-36.24,;-2.45,-34.92,;-3.99,-34.95,;-.11,-36.21,;.81,-37.43,;2.27,-36.93,;2.24,-35.39,;.76,-34.94,;3.56,-34.6,;4.9,-35.36,;4.92,-36.9,;6.23,-34.57,;7.57,-35.32,;8.9,-34.54,;8.88,-33,;10.24,-35.29,;10.25,-36.83,;11.59,-37.58,;12.92,-36.8,;12.89,-35.25,;11.55,-34.5,;14.21,-34.46,;15.56,-35.21,;14.19,-32.92,;15.54,-33.68,;-6.28,-36.34,;-7.08,-35.03,;-8.61,-35.06,;-9.36,-36.41,;-8.56,-37.73,;-7.02,-37.7,)| Show InChI InChI=1S/C24H28F3N5O3/c25-24(26,27)17-3-1-2-16(12-17)22(34)29-13-21(33)31-18-7-11-32(14-18)19-4-8-23(35,9-5-19)20-6-10-28-15-30-20/h1-3,6,10,12,15,18-19,35H,4-5,7-9,11,13-14H2,(H,29,34)(H,31,33)/t18-,19-,23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Mus musculus) | BDBM50363942

(CHEMBL1951766)Show SMILES Cc1ccc(cc1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-10.43,2.36,;-8.89,2.39,;-8.15,3.74,;-6.61,3.78,;-5.81,2.46,;-6.55,1.1,;-8.1,1.07,;-4.27,2.5,;-5.05,3.84,;-3.46,1.18,;-1.93,1.21,;-1.18,2.56,;-1.98,3.88,;-3.53,3.85,;.36,2.59,;1.28,1.37,;2.73,1.87,;2.7,3.41,;1.23,3.86,;4.03,4.2,;5.37,3.45,;5.39,1.91,;6.7,4.23,;8.04,3.48,;9.36,4.27,;9.34,5.81,;10.71,3.51,;10.72,1.97,;12.06,1.22,;13.38,2.01,;13.36,3.55,;12.02,4.3,;14.68,4.35,;16.03,3.6,;14.66,5.89,;16.01,5.13,)| Show InChI InChI=1S/C27H32F3N3O3/c1-18-5-7-20(8-6-18)26(36)12-9-23(10-13-26)33-14-11-22(17-33)32-24(34)16-31-25(35)19-3-2-4-21(15-19)27(28,29)30/h2-8,15,22-23,36H,9-14,16-17H2,1H3,(H,31,35)(H,32,34)/t22-,23-,26-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 156 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at mouse CCR2 assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363955
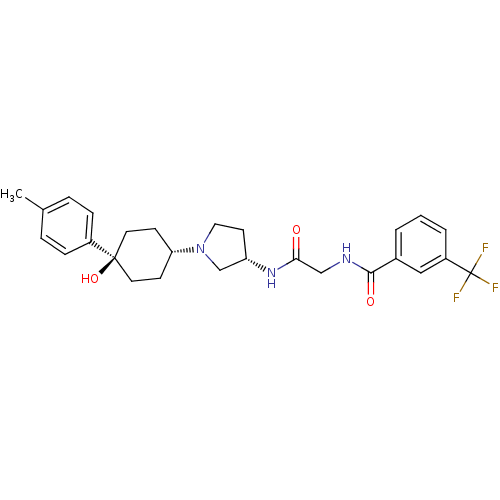
(CHEMBL1951767)Show SMILES Cc1ccc(cc1)[C@]1(O)CC[C@@H](CC1)N1CC[C@@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:17.21,11.15,wD:7.8,(19.13,3.03,;20.67,3.06,;21.42,4.41,;22.95,4.45,;23.75,3.13,;23.01,1.77,;21.47,1.74,;25.29,3.17,;24.52,4.51,;26.1,1.85,;27.63,1.88,;28.38,3.23,;27.58,4.55,;26.04,4.52,;29.92,3.26,;30.84,2.04,;32.3,2.54,;32.27,4.08,;30.79,4.53,;33.59,4.87,;34.93,4.11,;34.95,2.57,;36.26,4.9,;37.6,4.15,;38.93,4.93,;38.91,6.47,;40.27,4.18,;40.28,2.64,;41.62,1.89,;42.95,2.68,;42.92,4.22,;41.58,4.97,;44.24,5.01,;45.59,4.27,;44.22,6.55,;45.57,5.79,)| Show InChI InChI=1S/C27H32F3N3O3/c1-18-5-7-20(8-6-18)26(36)12-9-23(10-13-26)33-14-11-22(17-33)32-24(34)16-31-25(35)19-3-2-4-21(15-19)27(28,29)30/h2-8,15,22-23,36H,9-14,16-17H2,1H3,(H,31,35)(H,32,34)/t22-,23-,26-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363942

(CHEMBL1951766)Show SMILES Cc1ccc(cc1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-10.43,2.36,;-8.89,2.39,;-8.15,3.74,;-6.61,3.78,;-5.81,2.46,;-6.55,1.1,;-8.1,1.07,;-4.27,2.5,;-5.05,3.84,;-3.46,1.18,;-1.93,1.21,;-1.18,2.56,;-1.98,3.88,;-3.53,3.85,;.36,2.59,;1.28,1.37,;2.73,1.87,;2.7,3.41,;1.23,3.86,;4.03,4.2,;5.37,3.45,;5.39,1.91,;6.7,4.23,;8.04,3.48,;9.36,4.27,;9.34,5.81,;10.71,3.51,;10.72,1.97,;12.06,1.22,;13.38,2.01,;13.36,3.55,;12.02,4.3,;14.68,4.35,;16.03,3.6,;14.66,5.89,;16.01,5.13,)| Show InChI InChI=1S/C27H32F3N3O3/c1-18-5-7-20(8-6-18)26(36)12-9-23(10-13-26)33-14-11-22(17-33)32-24(34)16-31-25(35)19-3-2-4-21(15-19)27(28,29)30/h2-8,15,22-23,36H,9-14,16-17H2,1H3,(H,31,35)(H,32,34)/t22-,23-,26-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.32E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of dofetidine from human Erg |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50363953

(CHEMBL1951778 | CHEMBL1963131)Show SMILES COc1ccc(cn1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:12.16,wD:18.22,8.9,(-10.53,-32.06,;-9.74,-33.38,;-8.2,-33.35,;-7.45,-32,;-5.92,-31.97,;-5.12,-33.28,;-5.86,-34.64,;-7.4,-34.67,;-3.58,-33.25,;-4.36,-31.9,;-2.77,-34.56,;-1.24,-34.53,;-.49,-33.18,;-1.29,-31.87,;-2.83,-31.89,;1.05,-33.15,;1.97,-34.38,;3.43,-33.87,;3.4,-32.33,;1.92,-31.89,;4.72,-31.55,;6.06,-32.3,;6.08,-33.84,;7.39,-31.51,;8.73,-32.27,;10.05,-31.48,;10.04,-29.94,;11.4,-32.23,;11.41,-33.77,;12.75,-34.53,;14.08,-33.74,;14.05,-32.19,;12.71,-31.45,;15.37,-31.4,;16.72,-32.15,;15.35,-29.86,;16.7,-30.62,)| Show InChI InChI=1S/C26H31F3N4O4/c1-37-23-6-5-19(14-30-23)25(36)10-7-21(8-11-25)33-12-9-20(16-33)32-22(34)15-31-24(35)17-3-2-4-18(13-17)26(27,28)29/h2-6,13-14,20-21,36H,7-12,15-16H2,1H3,(H,31,35)(H,32,34)/t20-,21-,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Erg by patch clamp assay |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data