

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50038843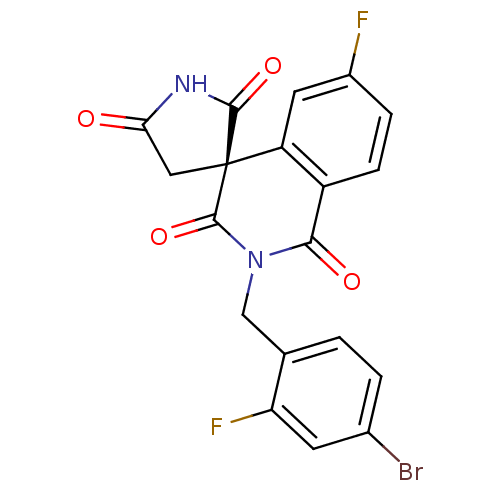 ((4R)-2-(4-bromo-2-fluorobenzyl)-6-fluoro-1H,2'H,5'...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 6.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of N-terminal 6His-tagged human aldehyde reductase expressed in Escherichia coli BL21(DE3) mediated D-glucuronate reduction | Bioorg Med Chem 18: 2485-90 (2010) Article DOI: 10.1016/j.bmc.2010.02.050 BindingDB Entry DOI: 10.7270/Q2K35TTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16314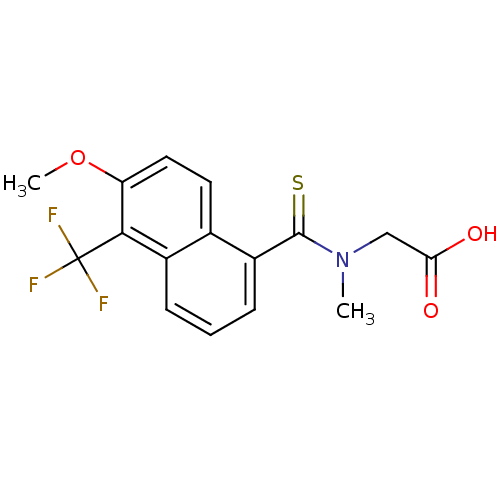 (2-{[6-methoxy-5-(trifluoromethyl)naphthalen-1-yl]-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 720 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of N-terminal 6His-tagged human aldehyde reductase expressed in Escherichia coli BL21(DE3) mediated D-glucuronate reduction | Bioorg Med Chem 18: 2485-90 (2010) Article DOI: 10.1016/j.bmc.2010.02.050 BindingDB Entry DOI: 10.7270/Q2K35TTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16512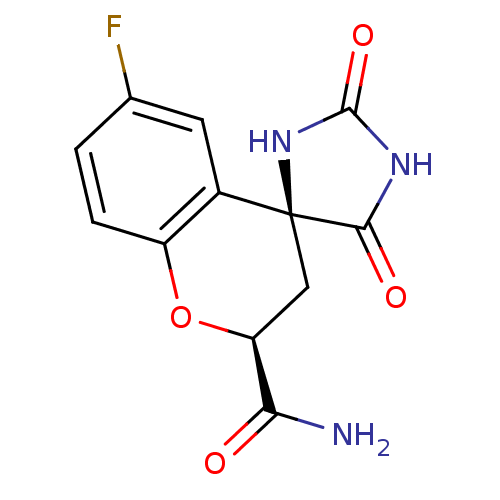 ((2S,4S)-6-fluoro-2',5'-dioxo-2,3-dihydrospiro[1-be...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University (Parkville Campus) Curated by ChEMBL | Assay Description Inhibitory concentration against human ALR1 Aldehyde reductase using DL-glyceraldehyde | J Med Chem 48: 5536-42 (2005) Article DOI: 10.1021/jm050412o BindingDB Entry DOI: 10.7270/Q2K35T6F | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16512 ((2S,4S)-6-fluoro-2',5'-dioxo-2,3-dihydrospiro[1-be...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Rational Drug Design Laboratories Curated by ChEMBL | Assay Description Tested for in vitro inhibition activity against human aldehyde reductase (AHR) | J Med Chem 43: 2479-83 (2000) BindingDB Entry DOI: 10.7270/Q2HH6JBT | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50241817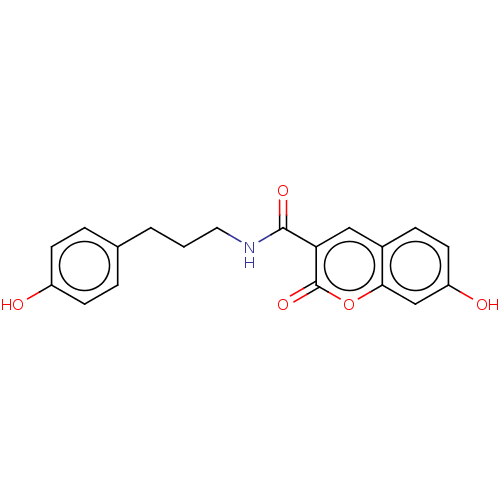 (CHEMBL4081954) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of recombinant human AKR1A1 using pyridine-3-aldehyde as substrate | J Med Chem 60: 8441-8455 (2017) Article DOI: 10.1021/acs.jmedchem.7b00830 BindingDB Entry DOI: 10.7270/Q2NZ89S7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50241828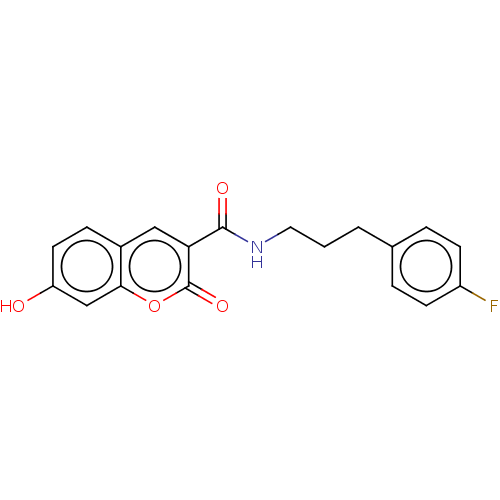 (CHEMBL4089817) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of recombinant human AKR1A1 using pyridine-3-aldehyde as substrate | J Med Chem 60: 8441-8455 (2017) Article DOI: 10.1021/acs.jmedchem.7b00830 BindingDB Entry DOI: 10.7270/Q2NZ89S7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16314 (2-{[6-methoxy-5-(trifluoromethyl)naphthalen-1-yl]-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.94E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16462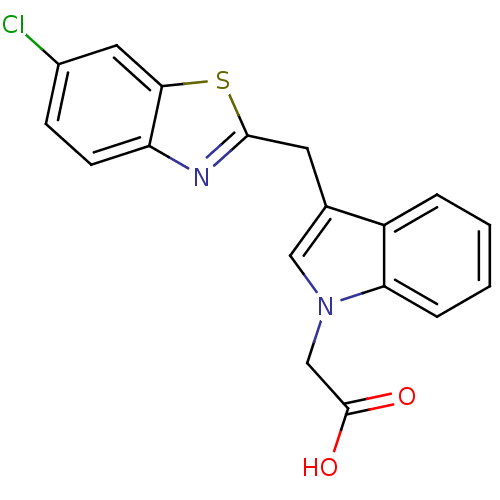 (2-{3-[(6-chloro-1,3-benzothiazol-2-yl)methyl]-1H-i...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50049730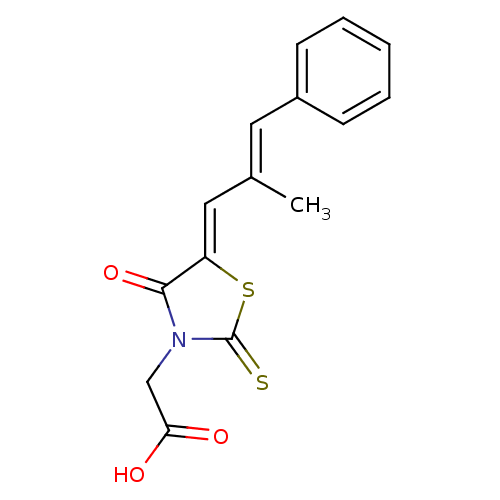 (2-(5-(2-methyl-3-phenylallylidene)-4-oxo-2-thioxot...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of N-terminal 6His-tagged human aldehyde reductase expressed in Escherichia coli BL21(DE3) mediated D-glucuronate reduction | Bioorg Med Chem 18: 2485-90 (2010) Article DOI: 10.1016/j.bmc.2010.02.050 BindingDB Entry DOI: 10.7270/Q2K35TTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50049730 (2-(5-(2-methyl-3-phenylallylidene)-4-oxo-2-thioxot...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16483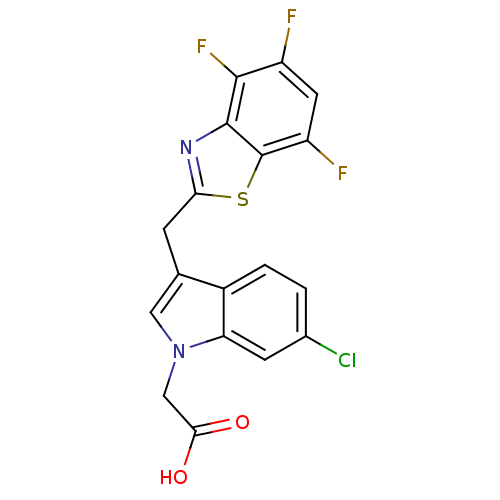 (2-{6-chloro-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16452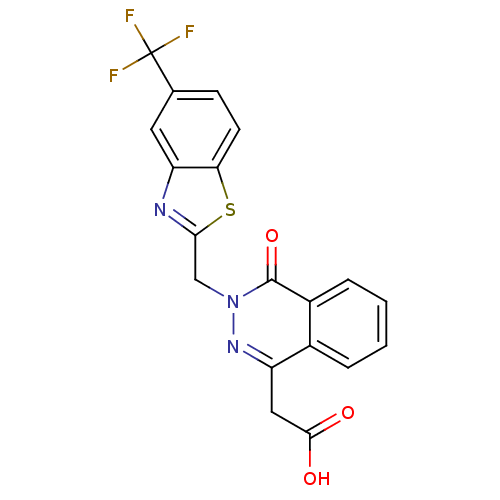 ((4-oxo-3-{[5-(trifluoromethyl)-1,3-benzothiazol-2-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of N-terminal 6His-tagged human aldehyde reductase expressed in Escherichia coli BL21(DE3) mediated D-glucuronate reduction | Bioorg Med Chem 18: 2485-90 (2010) Article DOI: 10.1016/j.bmc.2010.02.050 BindingDB Entry DOI: 10.7270/Q2K35TTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16461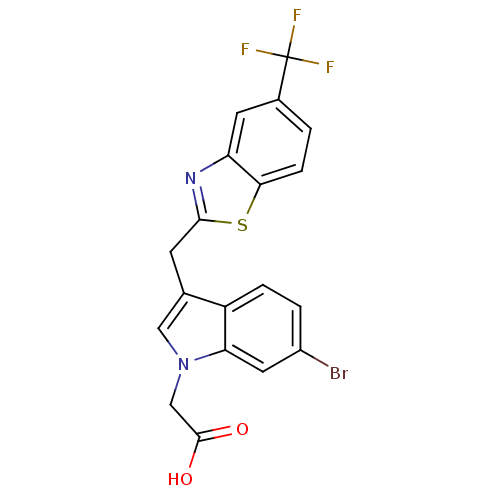 (2-(6-bromo-3-{[5-(trifluoromethyl)-1,3-benzothiazo...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16465 (2-{3-[(6-fluoro-1,3-benzothiazol-2-yl)methyl]-1H-i...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16312 ((4S)-6-fluoro-2,3-dihydrospiro[1-benzopyran-4,4'-i...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of N-terminal 6His-tagged human aldehyde reductase expressed in Escherichia coli BL21(DE3) mediated D-glucuronate reduction | Bioorg Med Chem 18: 2485-90 (2010) Article DOI: 10.1016/j.bmc.2010.02.050 BindingDB Entry DOI: 10.7270/Q2K35TTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16475 (2-{5-bromo-3-[(4,5,7-trifluoro-1,3-benzothiazol-2-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50363067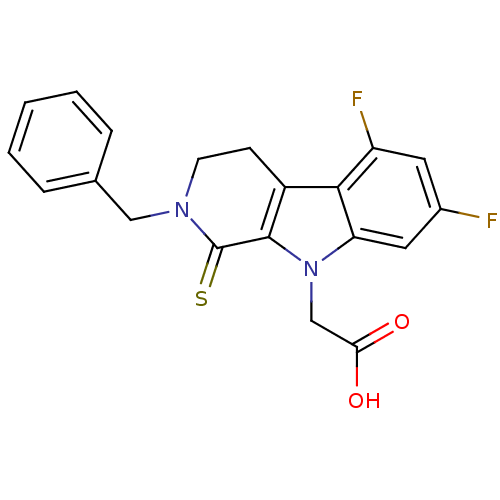 (CHEMBL1944858) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16478 (2-[5-(benzyloxy)-3-[(4,5,7-trifluoro-1,3-benzothia...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16460 (2-(5-chloro-3-{[5-(trifluoromethyl)-1,3-benzothiaz...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16468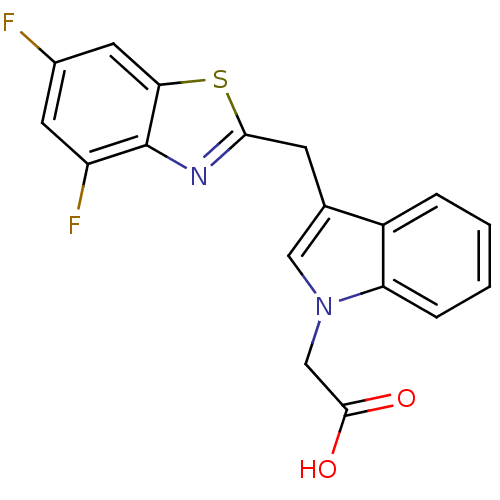 (2-{3-[(4,6-difluoro-1,3-benzothiazol-2-yl)methyl]-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16312 ((4S)-6-fluoro-2,3-dihydrospiro[1-benzopyran-4,4'-i...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Rational Drug Design Laboratories Curated by ChEMBL | Assay Description Tested for in vitro inhibition activity against human aldehyde reductase (AHR) | J Med Chem 43: 2479-83 (2000) BindingDB Entry DOI: 10.7270/Q2HH6JBT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16312 ((4S)-6-fluoro-2,3-dihydrospiro[1-benzopyran-4,4'-i...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University (Parkville Campus) Curated by ChEMBL | Assay Description Inhibitory concentration against human ALR1 Aldehyde reductase using DL-glyceraldehyde | J Med Chem 48: 5536-42 (2005) Article DOI: 10.1021/jm050412o BindingDB Entry DOI: 10.7270/Q2K35T6F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16458 (2-(3-{[5-(trifluoromethyl)-1,3-benzothiazol-2-yl]m...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16486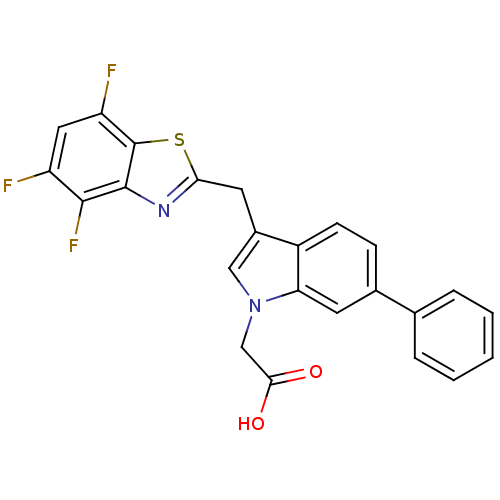 (2-{6-phenyl-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16464 (2-{3-[(5-fluoro-1,3-benzothiazol-2-yl)methyl]-1H-i...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16495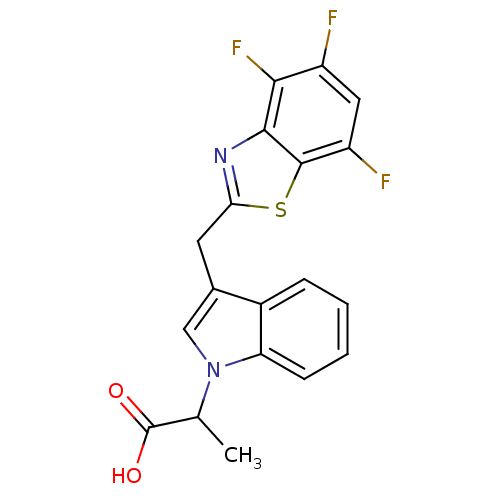 (2-{3-[(4,5,7-trifluoro-1,3-benzothiazol-2-yl)methy...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16488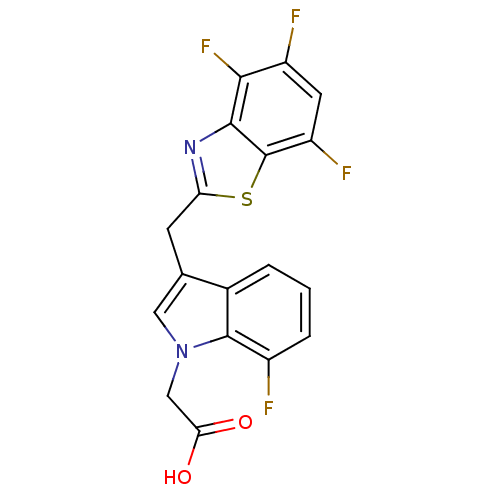 (2-{7-fluoro-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16482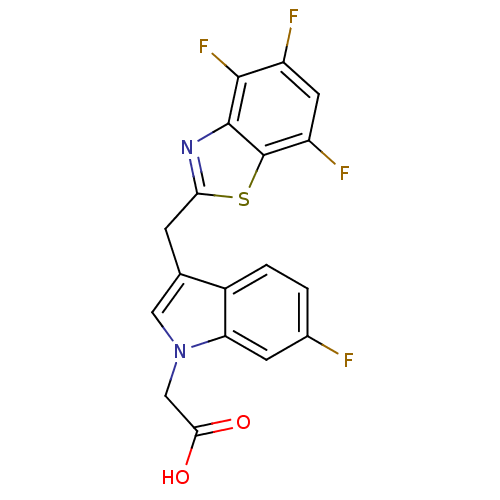 (2-{6-fluoro-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50363054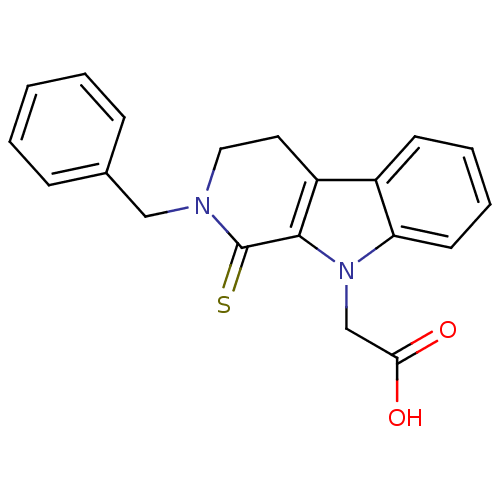 (CHEMBL1946959) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16496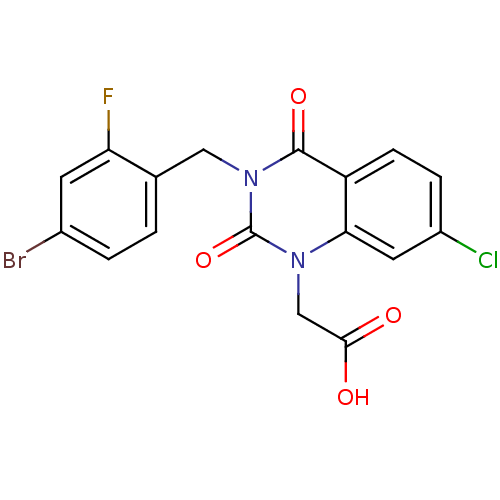 (2-{3-[(4-bromo-2-fluorophenyl)methyl]-7-chloro-2,4...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents | Article PubMed | n/a | n/a | 9.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50363066 (CHEMBL1944857) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16480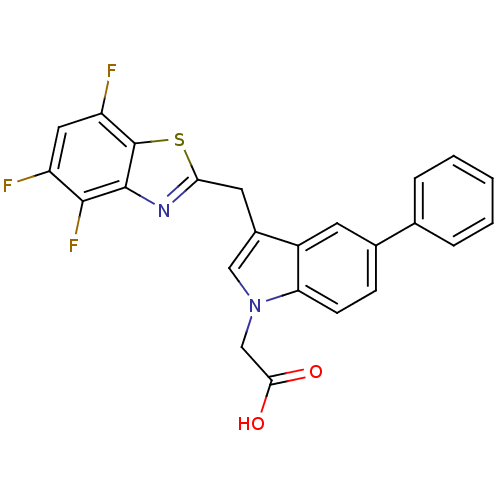 (2-{5-phenyl-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16459 (2-(5-methyl-3-{[5-(trifluoromethyl)-1,3-benzothiaz...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.02E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16493 (2-{3-[2-(4,5,7-trifluoro-1,3-benzothiazol-2-yl)eth...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50363060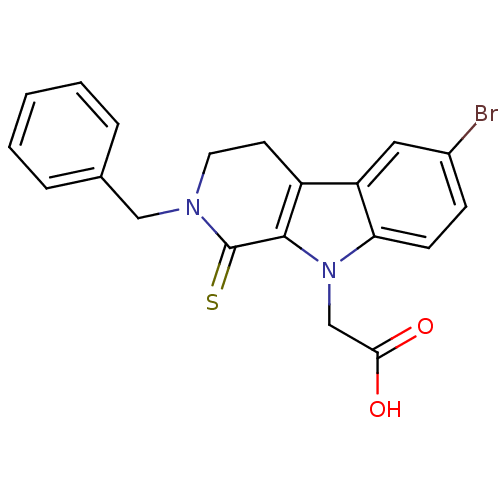 (CHEMBL1946965) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50363063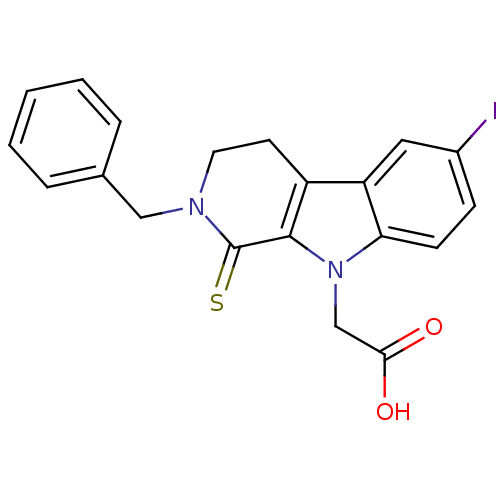 (CHEMBL1944854) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16473 (2-{5-chloro-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16479 (2-{5-phenoxy-3-[(4,5,7-trifluoro-1,3-benzothiazol-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16463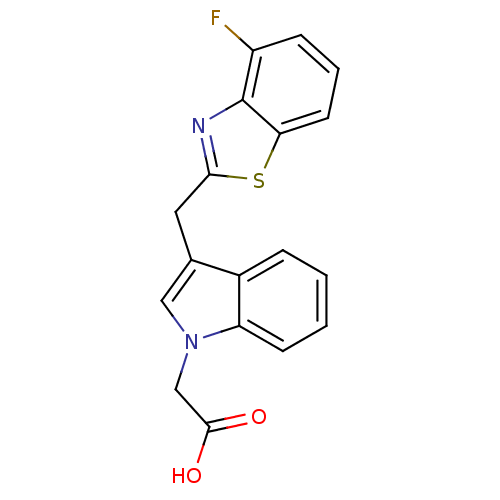 (2-{3-[(4-fluoro-1,3-benzothiazol-2-yl)methyl]-1H-i...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50363064 (CHEMBL1944855) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16485 (2-{6-methoxy-3-[(4,5,7-trifluoro-1,3-benzothiazol-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16470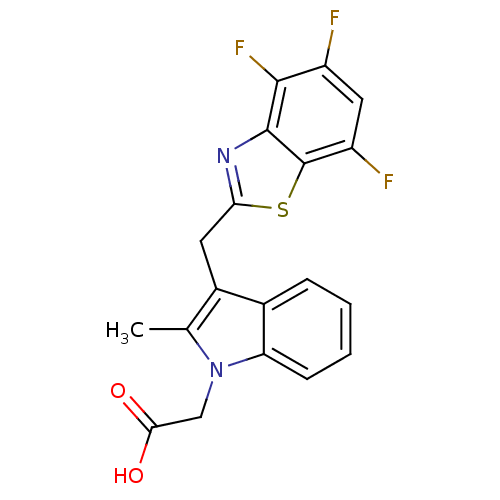 (2-{2-methyl-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16484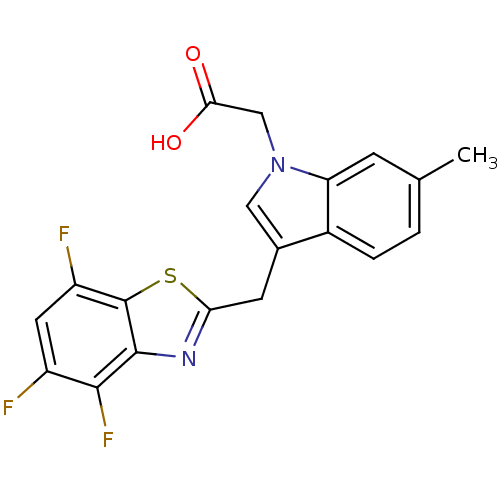 (2-{6-methyl-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16467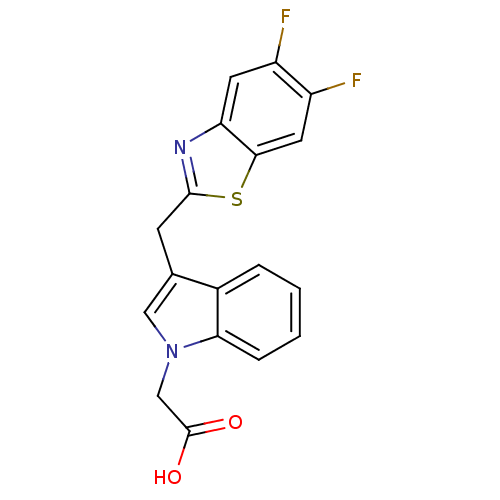 (2-{3-[(5,6-difluoro-1,3-benzothiazol-2-yl)methyl]-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16472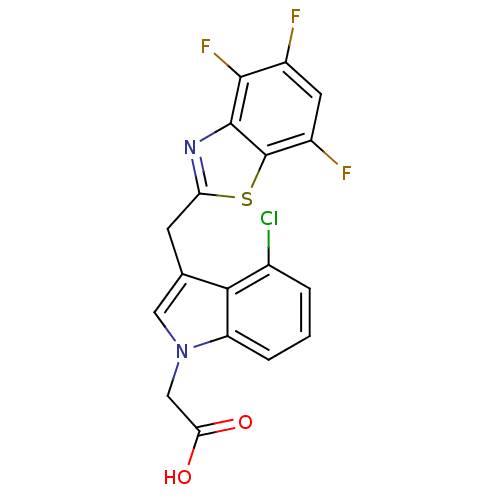 (2-{4-chloro-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50363059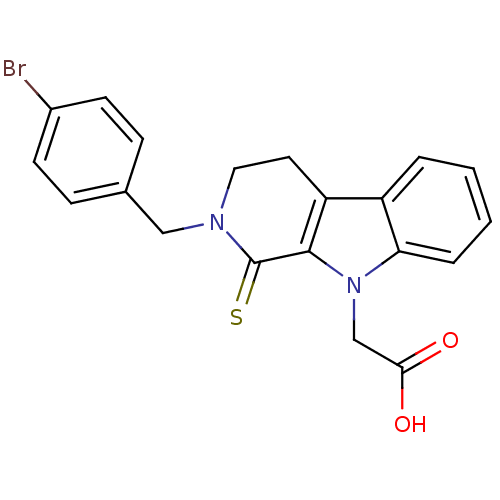 (CHEMBL1946964) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50363062 (CHEMBL1944853) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16471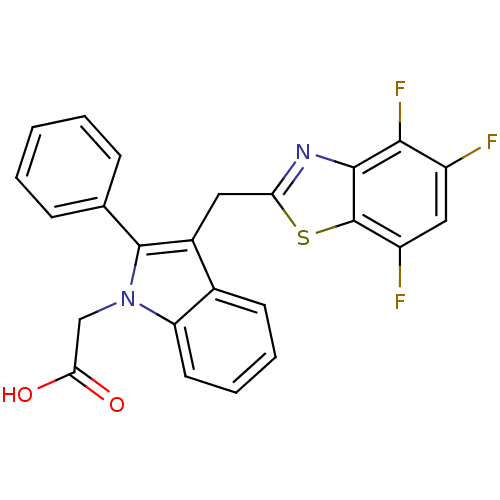 (2-{2-phenyl-3-[(4,5,7-trifluoro-1,3-benzothiazol-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM50363065 (CHEMBL1944856) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toyama Curated by ChEMBL | Assay Description Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometry | Bioorg Med Chem 20: 356-67 (2011) Article DOI: 10.1016/j.bmc.2011.10.073 BindingDB Entry DOI: 10.7270/Q2N58MTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member A1 (Homo sapiens (Human)) | BDBM16490 (2-{7-bromo-3-[(4,5,7-trifluoro-1,3-benzothiazol-2-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute for Diabetes Discovery | Assay Description The activity of the test enzyme was determined spectrophotometrically by monitoring the change in absorbance at 340 nm, which is due to the disappear... | J Med Chem 48: 3141-52 (2005) Article DOI: 10.1021/jm0492094 BindingDB Entry DOI: 10.7270/Q2J38QSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 72 total ) | Next | Last >> |