

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50379273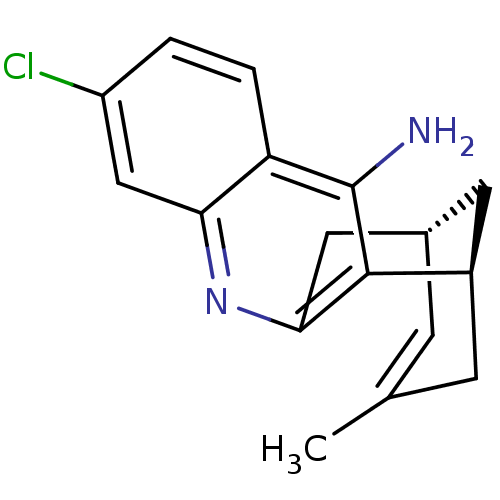 (CHEMBL1994202 | US9238626, (-)-Huprine Y HCl) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50379273 (CHEMBL1994202 | US9238626, (-)-Huprine Y HCl) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.430 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.690 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM202364 (US9238626, (-)-(Ib) HCl) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM202363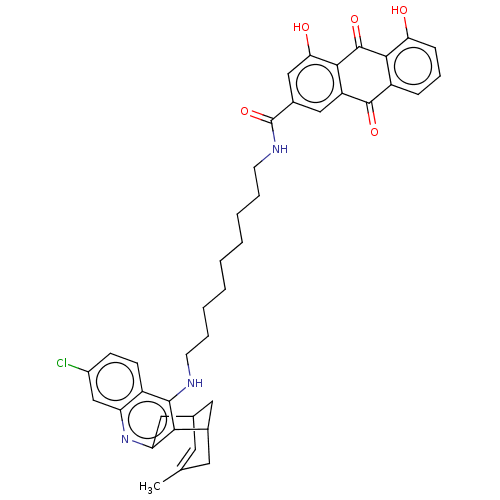 (US9238626, (+/-)-(Ib) HCl) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 3.60 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM202362 (US9238626, (+/-)-(Ia) HCl) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM202366 (US9238626, (+/-)-(Ic) HCl) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 7.60 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM202364 (US9238626, (-)-(Ib) HCl) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 9.40 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50379274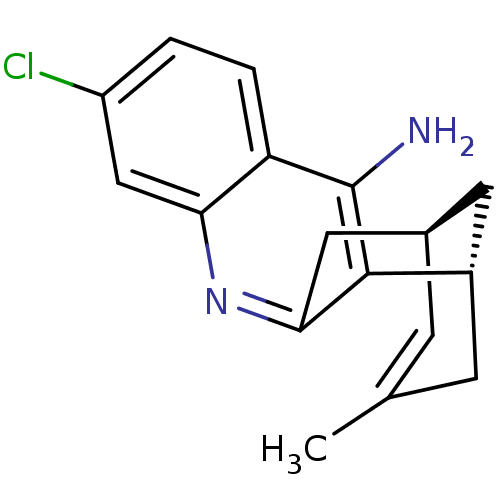 (CHEMBL2011499 | US9238626, (+)-Huprine Y HCl) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 13.6 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM202367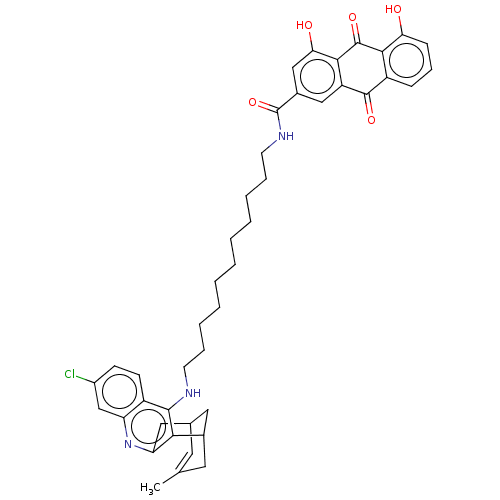 (US9238626, (+/-)-(Id) HCl) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 16.3 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM202367 (US9238626, (+/-)-(Id) HCl) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 17.4 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM202368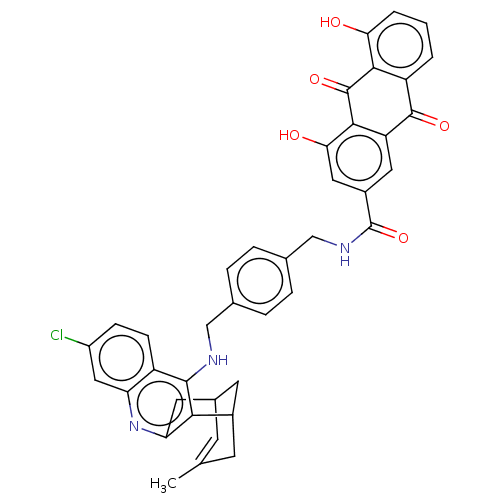 (US9238626, (+/-)-(Ie) HCl) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 18.2 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM202363 (US9238626, (+/-)-(Ib) HCl) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 32.6 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM202366 (US9238626, (+/-)-(Ic) HCl) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 46.7 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM202368 (US9238626, (+/-)-(Ie) HCl) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 60 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM202365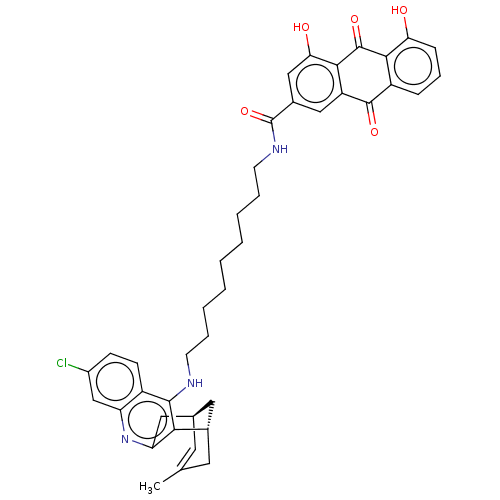 (US9238626, (+)-(Ib) HCl) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description β-Secretase (BACE-1, Sigma) inhibition studies were performed by employing a peptide mimicking APP sequence as substrate (methoxycoumarin-Ser-... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM202364 (US9238626, (-)-(Ib) HCl) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description β-Secretase (BACE-1, Sigma) inhibition studies were performed by employing a peptide mimicking APP sequence as substrate (methoxycoumarin-Ser-... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM202362 (US9238626, (+/-)-(Ia) HCl) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 88.6 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM202365 (US9238626, (+)-(Ib) HCl) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 98.9 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM202363 (US9238626, (+/-)-(Ib) HCl) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description β-Secretase (BACE-1, Sigma) inhibition studies were performed by employing a peptide mimicking APP sequence as substrate (methoxycoumarin-Ser-... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50379274 (CHEMBL2011499 | US9238626, (+)-Huprine Y HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 170 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 181 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50379273 (CHEMBL1994202 | US9238626, (-)-Huprine Y HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 222 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM202365 (US9238626, (+)-(Ib) HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 265 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM202362 (US9238626, (+/-)-(Ia) HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 350 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50379274 (CHEMBL2011499 | US9238626, (+)-Huprine Y HCl) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 373 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM202368 (US9238626, (+/-)-(Ie) HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 510 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM202364 (US9238626, (-)-(Ib) HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 513 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM202363 (US9238626, (+/-)-(Ib) HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 620 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM32021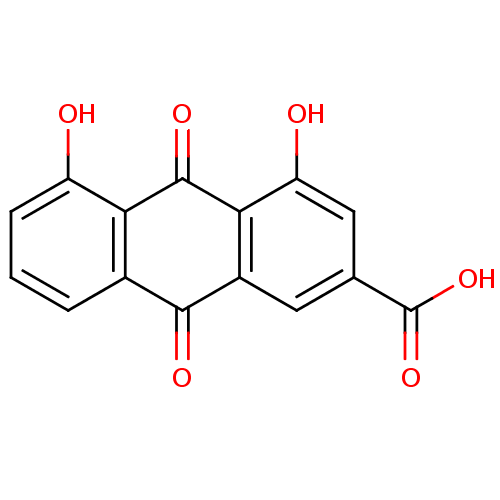 (4,5-bis(oxidanyl)-9,10-bis(oxidanylidene)anthracen...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Patents Similars | US Patent | n/a | n/a | 637 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM202367 (US9238626, (+/-)-(Id) HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 645 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM202362 (US9238626, (+/-)-(Ia) HCl) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 980 | n/a | n/a | n/a | n/a | n/a | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description β-Secretase (BACE-1, Sigma) inhibition studies were performed by employing a peptide mimicking APP sequence as substrate (methoxycoumarin-Ser-... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM202366 (US9238626, (+/-)-(Ic) HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM202366 (US9238626, (+/-)-(Ic) HCl) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.19E+3 | n/a | n/a | n/a | n/a | n/a | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description β-Secretase (BACE-1, Sigma) inhibition studies were performed by employing a peptide mimicking APP sequence as substrate (methoxycoumarin-Ser-... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM202367 (US9238626, (+/-)-(Id) HCl) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description β-Secretase (BACE-1, Sigma) inhibition studies were performed by employing a peptide mimicking APP sequence as substrate (methoxycoumarin-Ser-... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM202368 (US9238626, (+/-)-(Ie) HCl) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 2.02E+3 | n/a | n/a | n/a | n/a | n/a | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description β-Secretase (BACE-1, Sigma) inhibition studies were performed by employing a peptide mimicking APP sequence as substrate (methoxycoumarin-Ser-... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM202365 (US9238626, (+)-(Ib) HCl) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 2.93E+3 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50379273 (CHEMBL1994202 | US9238626, (-)-Huprine Y HCl) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | >5.00E+3 | n/a | n/a | n/a | n/a | n/a | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description β-Secretase (BACE-1, Sigma) inhibition studies were performed by employing a peptide mimicking APP sequence as substrate (methoxycoumarin-Ser-... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50379274 (CHEMBL2011499 | US9238626, (+)-Huprine Y HCl) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | >5.00E+3 | n/a | n/a | n/a | n/a | n/a | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description β-Secretase (BACE-1, Sigma) inhibition studies were performed by employing a peptide mimicking APP sequence as substrate (methoxycoumarin-Ser-... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM32021 (4,5-bis(oxidanyl)-9,10-bis(oxidanylidene)anthracen...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Patents Similars | US Patent | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description AChE inhibitory activity of compounds (Ia)-(Ie) and (-)-(Ib) and (+)-(Ib) was evaluated spectrophotometrically at 25° C. by the method of Ellma... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM32021 (4,5-bis(oxidanyl)-9,10-bis(oxidanylidene)anthracen...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Patents Similars | US Patent | n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | 8.0 | 25 |
UNIVERSITAT DE BARCELONA; PONTIFICIA UNIVERSIDAD CATÓLICA DE CHILE US Patent | Assay Description BChE inhibitory activity determinations were carried out similarly by the method of Ellman et al., using 0.02 unit/mL of human serum BChE and 300 u... | US Patent US9238626 (2016) BindingDB Entry DOI: 10.7270/Q2T152FZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||