

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082061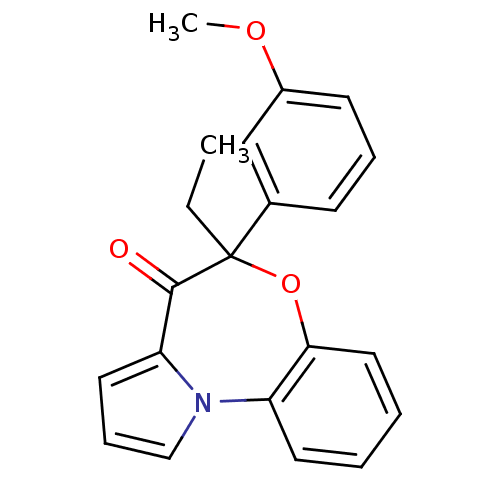 ((+/-)-5-ethyl-5-(3-methoxy-phenyl)-6-oxa-10b-aza-b...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082061 ((+/-)-5-ethyl-5-(3-methoxy-phenyl)-6-oxa-10b-aza-b...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082055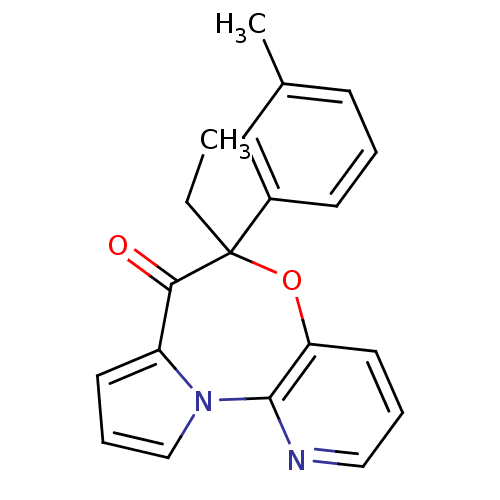 (5-Ethyl-5-m-tolyl-6-oxa-10,10b-diaza-benzo[e]azule...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082061 ((+/-)-5-ethyl-5-(3-methoxy-phenyl)-6-oxa-10b-aza-b...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082060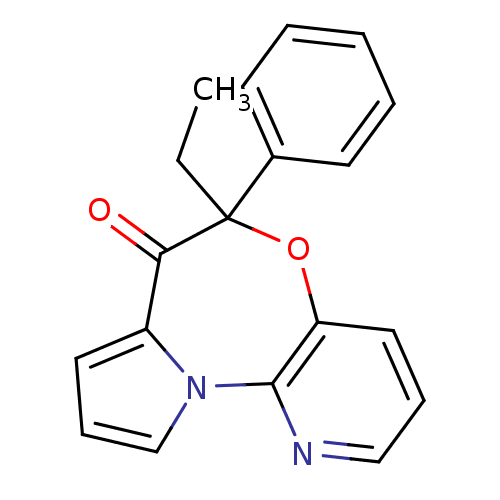 (5-Ethyl-5-phenyl-6-oxa-10,10b-diaza-benzo[e]azulen...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082062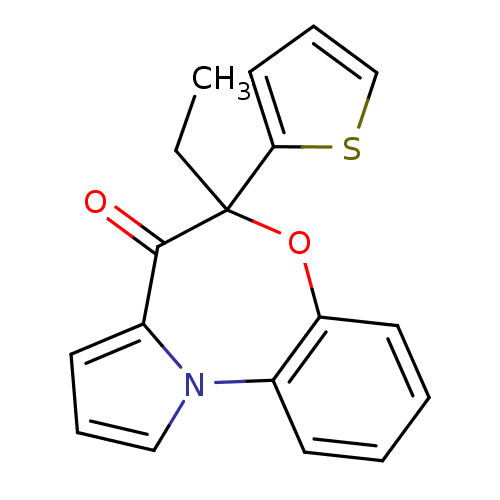 (5-Ethyl-5-thiophen-2-yl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082055 (5-Ethyl-5-m-tolyl-6-oxa-10,10b-diaza-benzo[e]azule...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082060 (5-Ethyl-5-phenyl-6-oxa-10,10b-diaza-benzo[e]azulen...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082055 (5-Ethyl-5-m-tolyl-6-oxa-10,10b-diaza-benzo[e]azule...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 44 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082056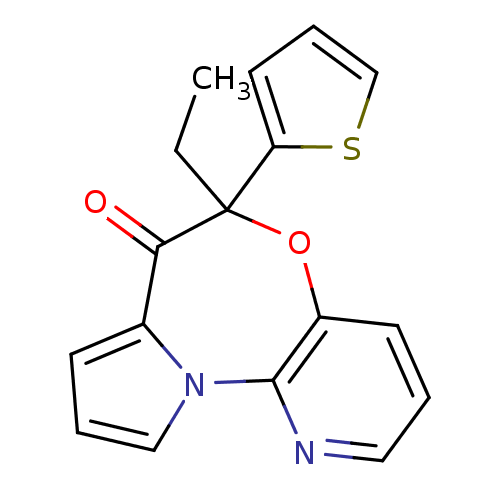 (5-Ethyl-5-thiophen-2-yl-6-oxa-10,10b-diaza-benzo[e...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 45 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082061 ((+/-)-5-ethyl-5-(3-methoxy-phenyl)-6-oxa-10b-aza-b...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082062 (5-Ethyl-5-thiophen-2-yl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082059 (5-Ethyl-5-(3-fluoro-phenyl)-6-oxa-10b-aza-benzo[e]...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082057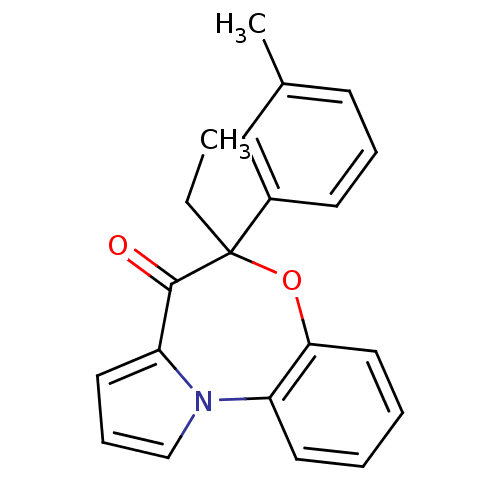 ((+/-)-5-ethyl-5-m-tolyl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082060 (5-Ethyl-5-phenyl-6-oxa-10,10b-diaza-benzo[e]azulen...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082056 (5-Ethyl-5-thiophen-2-yl-6-oxa-10,10b-diaza-benzo[e...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082056 (5-Ethyl-5-thiophen-2-yl-6-oxa-10,10b-diaza-benzo[e...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082056 (5-Ethyl-5-thiophen-2-yl-6-oxa-10,10b-diaza-benzo[e...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 93 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082059 (5-Ethyl-5-(3-fluoro-phenyl)-6-oxa-10b-aza-benzo[e]...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082057 ((+/-)-5-ethyl-5-m-tolyl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082062 (5-Ethyl-5-thiophen-2-yl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082059 (5-Ethyl-5-(3-fluoro-phenyl)-6-oxa-10b-aza-benzo[e]...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50052046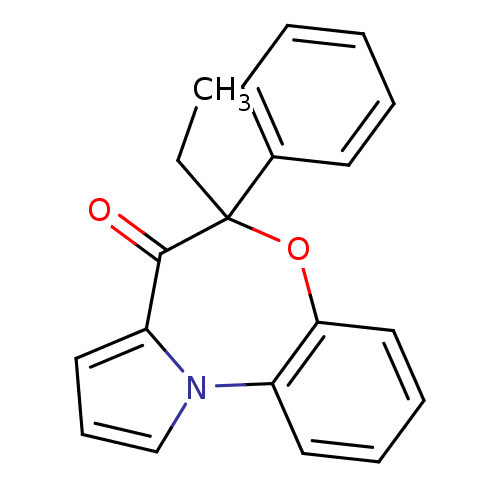 (5-Ethyl-5-phenyl-6-oxa-10b-aza-benzo[e]azulen-4-on...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082057 ((+/-)-5-ethyl-5-m-tolyl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082060 (5-Ethyl-5-phenyl-6-oxa-10,10b-diaza-benzo[e]azulen...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082062 (5-Ethyl-5-thiophen-2-yl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1434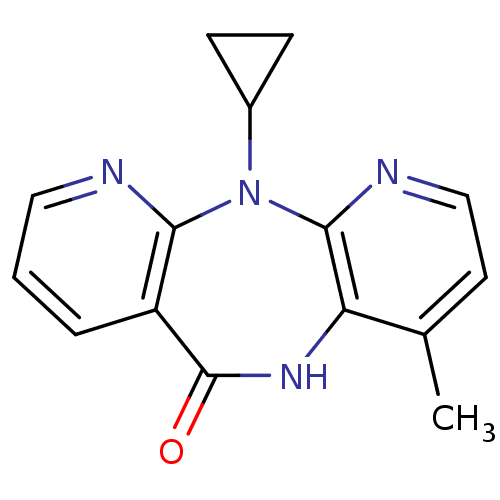 (11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082059 (5-Ethyl-5-(3-fluoro-phenyl)-6-oxa-10b-aza-benzo[e]...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082062 (5-Ethyl-5-thiophen-2-yl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082055 (5-Ethyl-5-m-tolyl-6-oxa-10,10b-diaza-benzo[e]azule...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082056 (5-Ethyl-5-thiophen-2-yl-6-oxa-10,10b-diaza-benzo[e...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082056 (5-Ethyl-5-thiophen-2-yl-6-oxa-10,10b-diaza-benzo[e...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 475 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50052046 (5-Ethyl-5-phenyl-6-oxa-10b-aza-benzo[e]azulen-4-on...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 750 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082063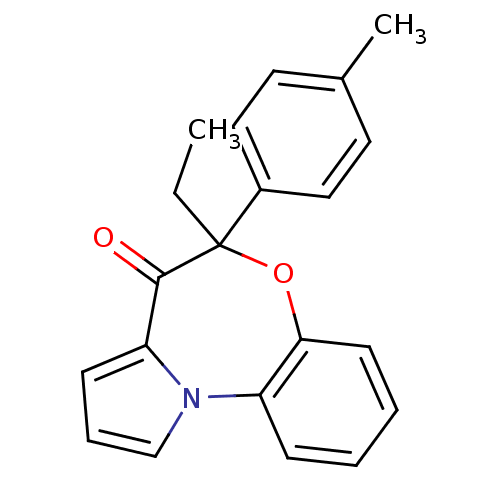 ((+/-)-6-ethyl-6-(p-tolyl)pyrrolo[2,1-d][1,5]benzox...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082062 (5-Ethyl-5-thiophen-2-yl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082057 ((+/-)-5-ethyl-5-m-tolyl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082055 (5-Ethyl-5-m-tolyl-6-oxa-10,10b-diaza-benzo[e]azule...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082059 (5-Ethyl-5-(3-fluoro-phenyl)-6-oxa-10b-aza-benzo[e]...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082060 (5-Ethyl-5-phenyl-6-oxa-10,10b-diaza-benzo[e]azulen...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082055 (5-Ethyl-5-m-tolyl-6-oxa-10,10b-diaza-benzo[e]azule...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082058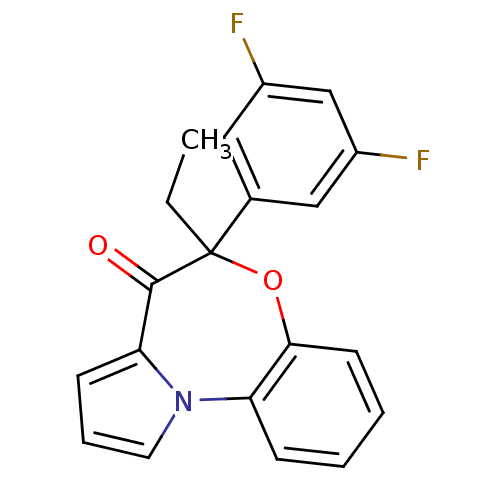 (5-(3,5-Difluoro-phenyl)-5-ethyl-6-oxa-10b-aza-benz...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 wild type reverse transcriptase (RT) | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082058 (5-(3,5-Difluoro-phenyl)-5-ethyl-6-oxa-10b-aza-benz...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082057 ((+/-)-5-ethyl-5-m-tolyl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082058 (5-(3,5-Difluoro-phenyl)-5-ethyl-6-oxa-10b-aza-benz...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082058 (5-(3,5-Difluoro-phenyl)-5-ethyl-6-oxa-10b-aza-benz...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50052046 (5-Ethyl-5-phenyl-6-oxa-10b-aza-benzo[e]azulen-4-on...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082057 ((+/-)-5-ethyl-5-m-tolyl-6-oxa-10b-aza-benzo[e]azul...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082059 (5-Ethyl-5-(3-fluoro-phenyl)-6-oxa-10b-aza-benzo[e]...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082060 (5-Ethyl-5-phenyl-6-oxa-10,10b-diaza-benzo[e]azulen...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082058 (5-(3,5-Difluoro-phenyl)-5-ethyl-6-oxa-10b-aza-benz...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 6.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1434 (11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB PubMed | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50052046 (5-Ethyl-5-phenyl-6-oxa-10b-aza-benzo[e]azulen-4-on...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 7.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution K103N | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1434 (11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB PubMed | 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution L100I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082061 ((+/-)-5-ethyl-5-(3-methoxy-phenyl)-6-oxa-10b-aza-b...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50052046 (5-Ethyl-5-phenyl-6-oxa-10b-aza-benzo[e]azulen-4-on...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50052046 (5-Ethyl-5-phenyl-6-oxa-10b-aza-benzo[e]azulen-4-on...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1434 (11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB PubMed | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution V106A | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082061 ((+/-)-5-ethyl-5-(3-methoxy-phenyl)-6-oxa-10b-aza-b...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50082058 (5-(3,5-Difluoro-phenyl)-5-ethyl-6-oxa-10b-aza-benz...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1434 (11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB PubMed | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y188L | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1434 (11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB PubMed | 3.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against HIV-1 mutant Reverse transcriptase containing the single amino acid substitution Y181I | J Med Chem 42: 4462-70 (1999) BindingDB Entry DOI: 10.7270/Q20R9NMJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||