 Found 32 hits of Enzyme Inhibition Constant Data
Found 32 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308556
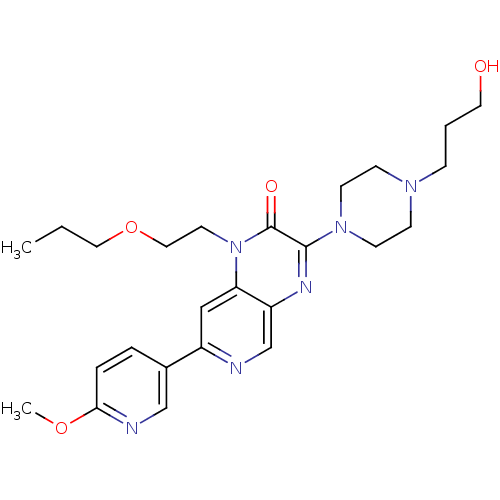
(3-(4-(3-hydroxypropyl)piperazin-1-yl)-1-(2-propoxy...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C25H34N6O4/c1-3-14-35-15-12-31-22-16-20(19-5-6-23(34-2)27-17-19)26-18-21(22)28-24(25(31)33)30-10-8-29(9-11-30)7-4-13-32/h5-6,16-18,32H,3-4,7-15H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308557
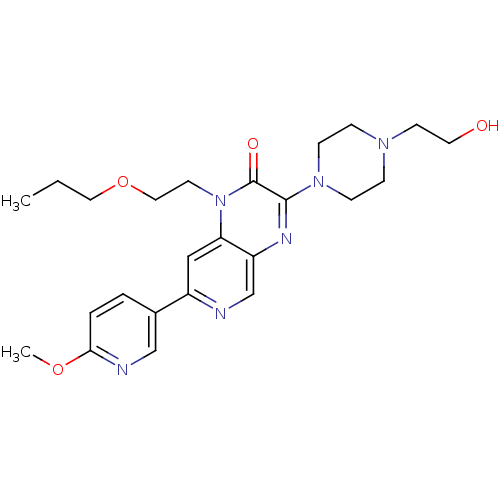
(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308563
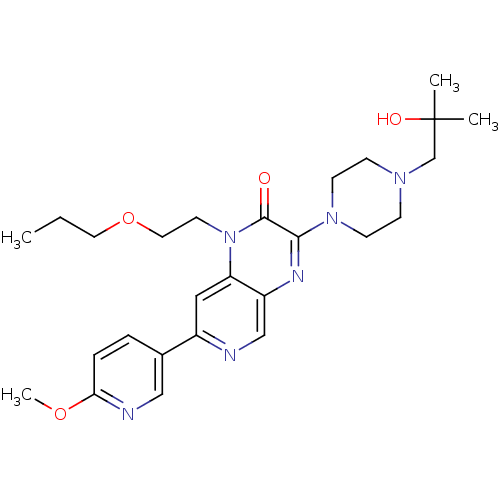
(3-(4-(2-hydroxy-2-methylpropyl)piperazin-1-yl)-1-(...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CC(C)(C)O)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C26H36N6O4/c1-5-13-36-14-12-32-22-15-20(19-6-7-23(35-4)28-16-19)27-17-21(22)29-24(25(32)33)31-10-8-30(9-11-31)18-26(2,3)34/h6-7,15-17,34H,5,8-14,18H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308558

(3-(4-ethylpiperazin-1-yl)-1-(2-propoxyethyl)-7-(6-...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CC)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O3/c1-4-13-33-14-12-30-21-15-19(18-6-7-22(32-3)26-16-18)25-17-20(21)27-23(24(30)31)29-10-8-28(5-2)9-11-29/h6-7,15-17H,4-5,8-14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308564

((R)-3-(4-(2-hydroxypropyl)piperazin-1-yl)-1-(2-iso...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(C[C@@H](C)O)CC2)c1=O)-c1ccc(OC)nc1 |r| Show InChI InChI=1S/C25H34N6O4/c1-4-12-35-13-11-31-22-14-20(19-5-6-23(34-3)27-15-19)26-16-21(22)28-24(25(31)33)30-9-7-29(8-10-30)17-18(2)32/h5-6,14-16,18,32H,4,7-13,17H2,1-3H3/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50300957
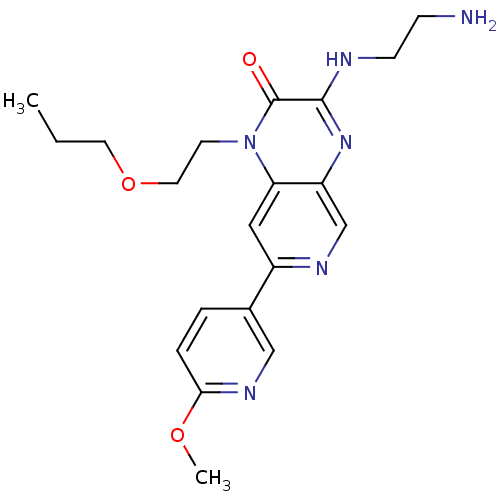
(3-(2-aminoethylamino)-7-(6-methoxypyridin-3-yl)-1-...)Show SMILES CCCOCCn1c2cc(ncc2nc(NCCN)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C20H26N6O3/c1-3-9-29-10-8-26-17-11-15(14-4-5-18(28-2)24-12-14)23-13-16(17)25-19(20(26)27)22-7-6-21/h4-5,11-13H,3,6-10,21H2,1-2H3,(H,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308561
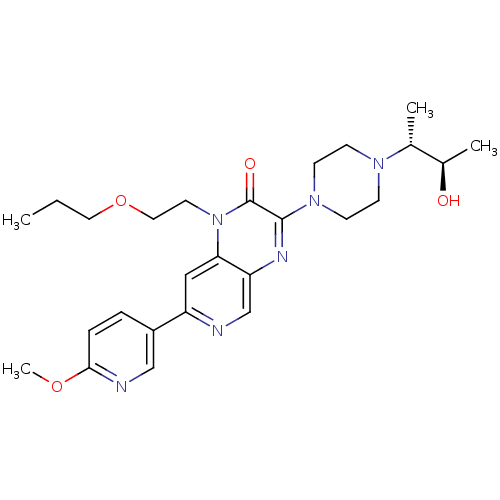
((+/-)-3-(4-((2R,3R)-3-hydroxybutan-2-yl)piperazin-...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CC2)[C@H](C)[C@@H](C)O)c1=O)-c1ccc(OC)nc1 |r| Show InChI InChI=1S/C26H36N6O4/c1-5-13-36-14-12-32-23-15-21(20-6-7-24(35-4)28-16-20)27-17-22(23)29-25(26(32)34)31-10-8-30(9-11-31)18(2)19(3)33/h6-7,15-19,33H,5,8-14H2,1-4H3/t18-,19-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308553

((S)-3-(4-(2-hydroxypropyl)piperazin-1-yl)-1-(2-iso...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(C[C@H](C)O)CC2)c1=O)-c1ccc(OC)nc1 |r| Show InChI InChI=1S/C25H34N6O4/c1-4-12-35-13-11-31-22-14-20(19-5-6-23(34-3)27-15-19)26-16-21(22)28-24(25(31)33)30-9-7-29(8-10-30)17-18(2)32/h5-6,14-16,18,32H,4,7-13,17H2,1-3H3/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308562

((+/-)-3-(4-((2S,3R)-3-hydroxybutan-2-yl)piperazin-...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CC2)[C@@H](C)[C@@H](C)O)c1=O)-c1ccc(OC)nc1 |r| Show InChI InChI=1S/C26H36N6O4/c1-5-13-36-14-12-32-23-15-21(20-6-7-24(35-4)28-16-20)27-17-22(23)29-25(26(32)34)31-10-8-30(9-11-31)18(2)19(3)33/h6-7,15-19,33H,5,8-14H2,1-4H3/t18-,19+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308555

(3-(4-(2-hydroxyethyl)-1,4-diazepan-1-yl)-1-(2-isop...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C25H34N6O4/c1-3-14-35-15-12-31-22-16-20(19-5-6-23(34-2)27-17-19)26-18-21(22)28-24(25(31)33)30-8-4-7-29(9-10-30)11-13-32/h5-6,16-18,32H,3-4,7-15H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.720 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308554
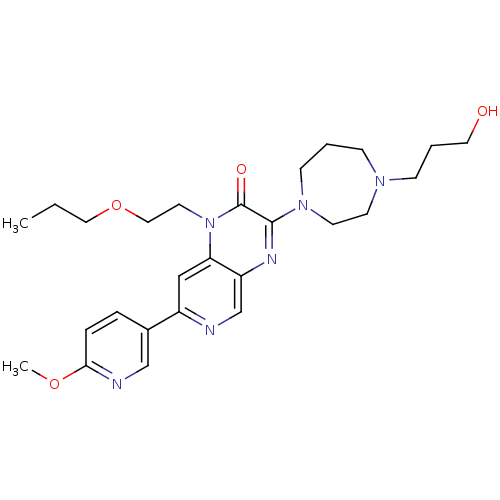
(3-(4-(3-hydroxypropyl)-1,4-diazepan-1-yl)-1-(2-pro...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCCN(CCCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C26H36N6O4/c1-3-15-36-16-13-32-23-17-21(20-6-7-24(35-2)28-18-20)27-19-22(23)29-25(26(32)34)31-10-4-8-30(11-12-31)9-5-14-33/h6-7,17-19,33H,3-5,8-16H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50300958
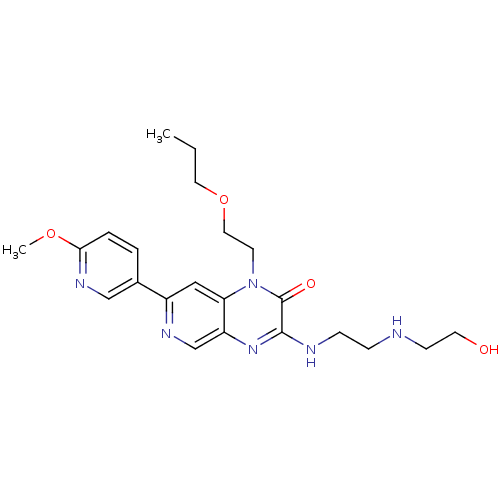
(3-(2-(2-hydroxyethylamino)ethylamino)-7-(6-methoxy...)Show SMILES CCCOCCn1c2cc(ncc2nc(NCCNCCO)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C22H30N6O4/c1-3-11-32-12-9-28-19-13-17(16-4-5-20(31-2)26-14-16)25-15-18(19)27-21(22(28)30)24-7-6-23-8-10-29/h4-5,13-15,23,29H,3,6-12H2,1-2H3,(H,24,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308559
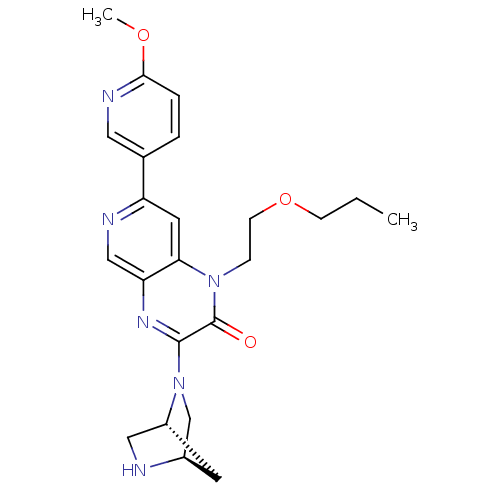
(3-((1R,4R)-2,5-diazabicyclo[2.2.1]heptan-2-yl)-1-(...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2C[C@H]3C[C@@H]2CN3)c1=O)-c1ccc(OC)nc1 |r| Show InChI InChI=1S/C23H28N6O3/c1-3-7-32-8-6-28-20-10-18(15-4-5-21(31-2)26-11-15)25-13-19(20)27-22(23(28)30)29-14-16-9-17(29)12-24-16/h4-5,10-11,13,16-17,24H,3,6-9,12,14H2,1-2H3/t16-,17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50308560
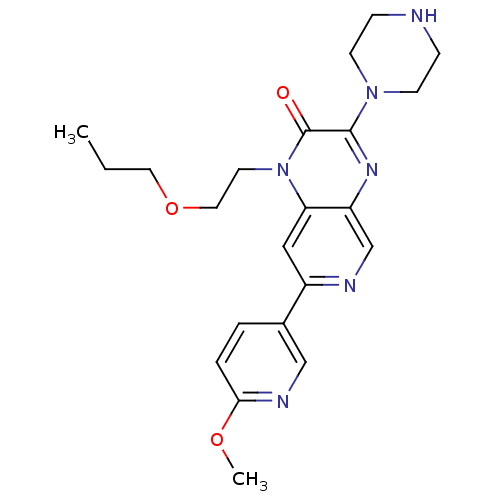
(3-(2-(2-hydroxyethylamino)ethylamino)-1-(2-propoxy...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCNCC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C22H28N6O3/c1-3-11-31-12-10-28-19-13-17(16-4-5-20(30-2)25-14-16)24-15-18(19)26-21(22(28)29)27-8-6-23-7-9-27/h4-5,13-15,23H,3,6-12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human platelet PDE5 assessed as reduction in conversion of [3H]cGMP to [3H]GMP after 2 hr by scintillation proximity assay |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50308562

((+/-)-3-(4-((2S,3R)-3-hydroxybutan-2-yl)piperazin-...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CC2)[C@@H](C)[C@@H](C)O)c1=O)-c1ccc(OC)nc1 |r| Show InChI InChI=1S/C26H36N6O4/c1-5-13-36-14-12-32-23-15-21(20-6-7-24(35-4)28-16-20)27-17-22(23)29-25(26(32)34)31-10-8-30(9-11-31)18(2)19(3)33/h6-7,15-19,33H,5,8-14H2,1-4H3/t18-,19+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch-clamp method |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50308564

((R)-3-(4-(2-hydroxypropyl)piperazin-1-yl)-1-(2-iso...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(C[C@@H](C)O)CC2)c1=O)-c1ccc(OC)nc1 |r| Show InChI InChI=1S/C25H34N6O4/c1-4-12-35-13-11-31-22-14-20(19-5-6-23(34-3)27-15-19)26-16-21(22)28-24(25(31)33)30-9-7-29(8-10-30)17-18(2)32/h5-6,14-16,18,32H,4,7-13,17H2,1-3H3/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch-clamp method |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50308558

(3-(4-ethylpiperazin-1-yl)-1-(2-propoxyethyl)-7-(6-...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CC)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O3/c1-4-13-33-14-12-30-21-15-19(18-6-7-22(32-3)26-16-18)25-17-20(21)27-23(24(30)31)29-10-8-28(5-2)9-11-29/h6-7,15-17H,4-5,8-14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch-clamp method |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50308560

(3-(2-(2-hydroxyethylamino)ethylamino)-1-(2-propoxy...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCNCC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C22H28N6O3/c1-3-11-31-12-10-28-19-13-17(16-4-5-20(30-2)25-14-16)24-15-18(19)26-21(22(28)29)27-8-6-23-7-9-27/h4-5,13-15,23H,3,6-12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 670 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch-clamp method |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.79E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch-clamp method |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
High affinity cAMP-specific and IBMX-insensitive 3',5'-cyclic phosphodiesterase 8B
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE8B |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
Dual specificity calcium/calmodulin-dependent 3',5'-cyclic nucleotide phosphodiesterase 1A
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE1A |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 7B
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE7B |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-inhibited 3',5'-cyclic phosphodiesterase 3B
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE3B |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
Dual specificity calcium/calmodulin-dependent 3',5'-cyclic nucleotide phosphodiesterase 1B
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE1B |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cGMP-inhibited 3',5'-cyclic phosphodiesterase 3A
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE3A |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
Dual specificity calcium/calmodulin-dependent 3',5'-cyclic nucleotide phosphodiesterase 1C
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE1C |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4D
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE4D |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
High affinity cAMP-specific and IBMX-insensitive 3',5'-cyclic phosphodiesterase 8A
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE8A |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE4C |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4B
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE4B |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A
(Homo sapiens (Human)) | BDBM50308557

(3-(4-(2-hydroxyethyl)piperazin-1-yl)-1-(2-propoxye...)Show SMILES CCCOCCn1c2cc(ncc2nc(N2CCN(CCO)CC2)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C24H32N6O4/c1-3-13-34-14-11-30-21-15-19(18-4-5-22(33-2)26-16-18)25-17-20(21)27-23(24(30)32)29-8-6-28(7-9-29)10-12-31/h4-5,15-17,31H,3,6-14H2,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of PDE4A |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50300958

(3-(2-(2-hydroxyethylamino)ethylamino)-7-(6-methoxy...)Show SMILES CCCOCCn1c2cc(ncc2nc(NCCNCCO)c1=O)-c1ccc(OC)nc1 Show InChI InChI=1S/C22H30N6O4/c1-3-11-32-12-9-28-19-13-17(16-4-5-20(31-2)26-14-16)25-15-18(19)27-21(22(28)30)24-7-6-23-8-10-29/h4-5,13-15,23,29H,3,6-12H2,1-2H3,(H,24,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch-clamp method |
J Med Chem 53: 2656-60 (2010)
Article DOI: 10.1021/jm901781q
BindingDB Entry DOI: 10.7270/Q2VM4CCK |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data