 Found 17 hits of Enzyme Inhibition Constant Data
Found 17 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50362540
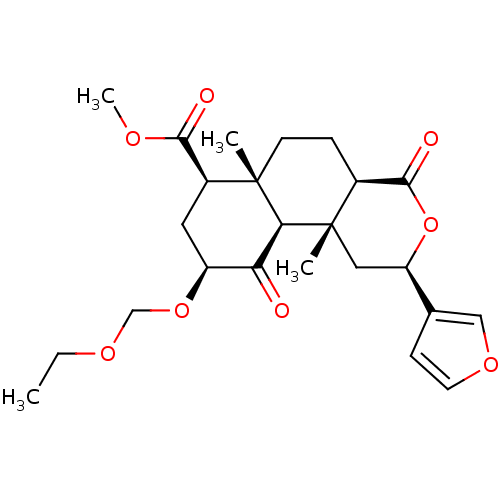
(CHEMBL1941031)Show SMILES CCOCO[C@H]1C[C@@H](C(=O)OC)[C@]2(C)CC[C@H]3C(=O)O[C@H](C[C@]3(C)[C@H]2C1=O)c1ccoc1 |r| Show InChI InChI=1S/C24H32O8/c1-5-29-13-31-17-10-16(21(26)28-4)23(2)8-6-15-22(27)32-18(14-7-9-30-12-14)11-24(15,3)20(23)19(17)25/h7,9,12,15-18,20H,5-6,8,10-11,13H2,1-4H3/t15-,16-,17-,18+,20-,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from human KOPR expressed in CHO cells |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50159165
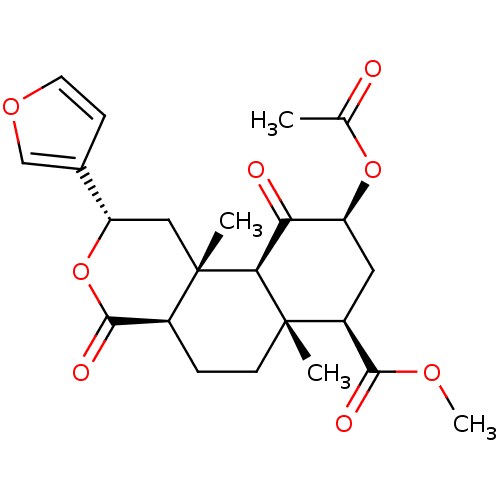
((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...)Show SMILES COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@@H](C[C@]21C)c1ccoc1 |r| Show InChI InChI=1S/C23H28O8/c1-12(24)30-16-9-15(20(26)28-4)22(2)7-5-14-21(27)31-17(13-6-8-29-11-13)10-23(14,3)19(22)18(16)25/h6,8,11,14-17,19H,5,7,9-10H2,1-4H3/t14-,15-,16-,17-,19-,22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from human KOPR expressed in CHO cells |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50362539
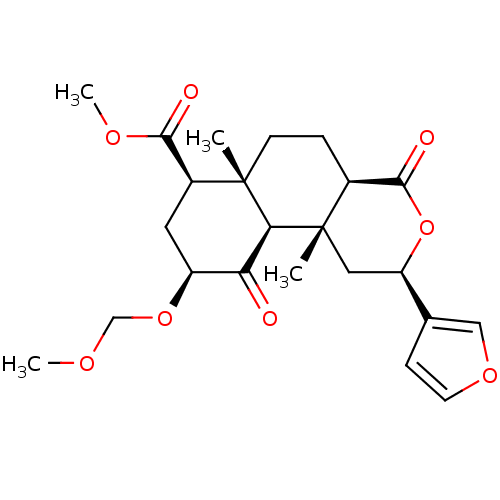
(CHEMBL1941030)Show SMILES COCO[C@H]1C[C@@H](C(=O)OC)[C@]2(C)CC[C@H]3C(=O)O[C@H](C[C@]3(C)[C@H]2C1=O)c1ccoc1 |r| Show InChI InChI=1S/C23H30O8/c1-22-7-5-14-21(26)31-17(13-6-8-29-11-13)10-23(14,2)19(22)18(24)16(30-12-27-3)9-15(22)20(25)28-4/h6,8,11,14-17,19H,5,7,9-10,12H2,1-4H3/t14-,15-,16-,17+,19-,22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from human KOPR expressed in CHO cells |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50000296
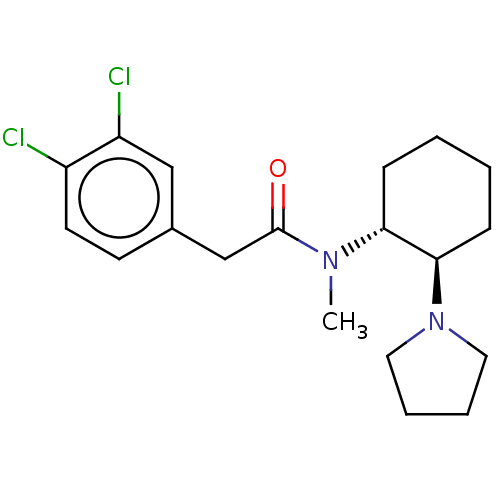
(CHEMBL441765 | CHEMBL482811 | U-50488H | US1149237...)Show SMILES CN([C@@H]1CCCC[C@H]1N1CCCC1)C(=O)Cc1ccc(Cl)c(Cl)c1 |r| Show InChI InChI=1S/C19H26Cl2N2O/c1-22(19(24)13-14-8-9-15(20)16(21)12-14)17-6-2-3-7-18(17)23-10-4-5-11-23/h8-9,12,17-18H,2-7,10-11,13H2,1H3/t17-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from human KOPR expressed in CHO cells |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50362542
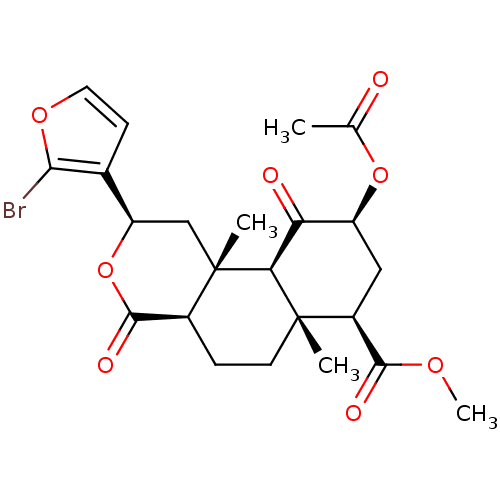
(CHEMBL1941033)Show SMILES COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@H](C[C@]21C)c1ccoc1Br |r| Show InChI InChI=1S/C23H27BrO8/c1-11(25)31-15-9-14(20(27)29-4)22(2)7-5-13-21(28)32-16(12-6-8-30-19(12)24)10-23(13,3)18(22)17(15)26/h6,8,13-16,18H,5,7,9-10H2,1-4H3/t13-,14-,15-,16+,18-,22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from human KOPR expressed in CHO cells |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50362543
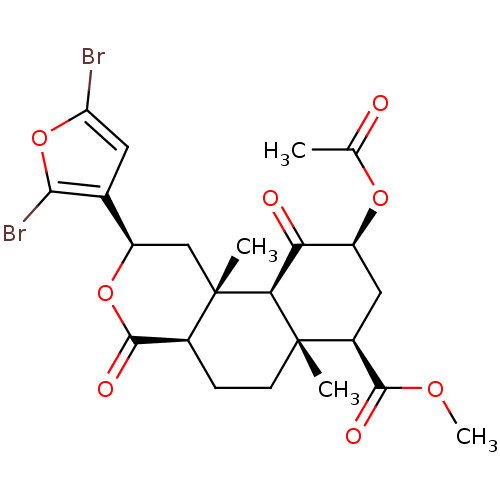
(CHEMBL1941034)Show SMILES COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@H](C[C@]21C)c1cc(Br)oc1Br |r| Show InChI InChI=1S/C23H26Br2O8/c1-10(26)31-14-8-13(20(28)30-4)22(2)6-5-12-21(29)32-15(11-7-16(24)33-19(11)25)9-23(12,3)18(22)17(14)27/h7,12-15,18H,5-6,8-9H2,1-4H3/t12-,13-,14-,15+,18-,22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from human KOPR expressed in CHO cells |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50362541
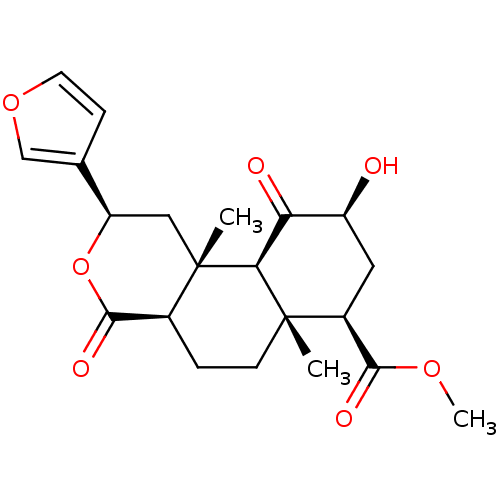
(CHEMBL1941032)Show SMILES COC(=O)[C@@H]1C[C@H](O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@H](C[C@]21C)c1ccoc1 |r| Show InChI InChI=1S/C21H26O7/c1-20-6-4-12-19(25)28-15(11-5-7-27-10-11)9-21(12,2)17(20)16(23)14(22)8-13(20)18(24)26-3/h5,7,10,12-15,17,22H,4,6,8-9H2,1-3H3/t12-,13-,14-,15+,17-,20-,21-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 63 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from human KOPR expressed in CHO cells |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50362543

(CHEMBL1941034)Show SMILES COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@H](C[C@]21C)c1cc(Br)oc1Br |r| Show InChI InChI=1S/C23H26Br2O8/c1-10(26)31-14-8-13(20(28)30-4)22(2)6-5-12-21(29)32-15(11-7-16(24)33-19(11)25)9-23(12,3)18(22)17(14)27/h7,12-15,18H,5-6,8-9H2,1-4H3/t12-,13-,14-,15+,18-,22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 318 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from rat MOPR expressed in CHO cells |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50105085
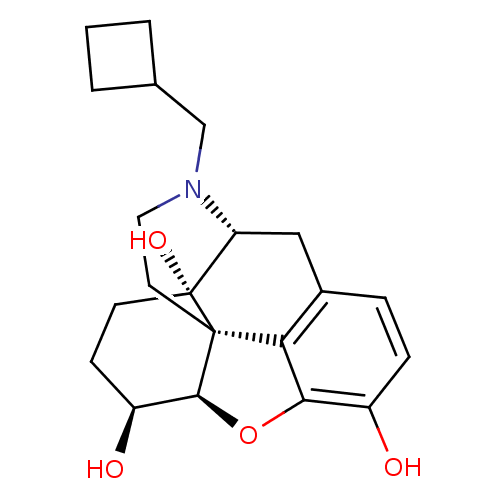
(17-cyclobutylmethyl-4,5alpha-epoxymorphinan-3,6alp...)Show SMILES O[C@H]1CC[C@@]2(O)[C@H]3Cc4ccc(O)c5O[C@@H]1[C@]2(CCN3CC1CCC1)c45 |r| Show InChI InChI=1S/C21H27NO4/c23-14-5-4-13-10-16-21(25)7-6-15(24)19-20(21,17(13)18(14)26-19)8-9-22(16)11-12-2-1-3-12/h4-5,12,15-16,19,23-25H,1-3,6-11H2/t15-,16+,19-,20-,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | n/a | n/a | 65 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human KOPR expressed in CHO cells assessed as [35S]GTPgammaS binding after 60 mins by liquid scintillation counting |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50266337
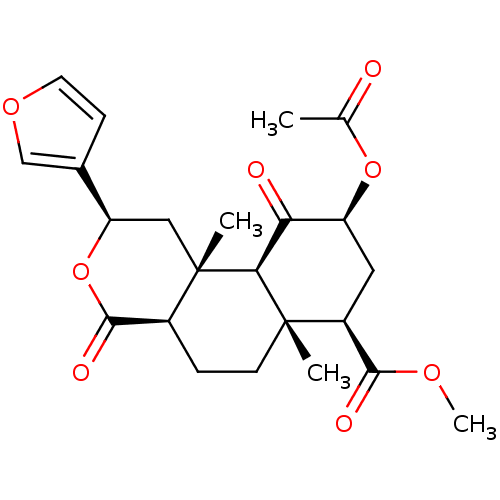
(12-epi-Salvinorin A | CHEMBL458235)Show SMILES COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@H](C[C@]21C)c1ccoc1 |r| Show InChI InChI=1S/C23H28O8/c1-12(24)30-16-9-15(20(26)28-4)22(2)7-5-14-21(27)31-17(13-6-8-29-11-13)10-23(14,3)19(22)18(16)25/h6,8,11,14-17,19H,5,7,9-10H2,1-4H3/t14-,15-,16-,17+,19-,22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 382 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human KOPR expressed in U2OS cells assessed as beta-arrestin2 recruitment after 90 mins by DiscoveRx PathHunter assay |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50362540

(CHEMBL1941031)Show SMILES CCOCO[C@H]1C[C@@H](C(=O)OC)[C@]2(C)CC[C@H]3C(=O)O[C@H](C[C@]3(C)[C@H]2C1=O)c1ccoc1 |r| Show InChI InChI=1S/C24H32O8/c1-5-29-13-31-17-10-16(21(26)28-4)23(2)8-6-15-22(27)32-18(14-7-9-30-12-14)11-24(15,3)20(23)19(17)25/h7,9,12,15-18,20H,5-6,8,10-11,13H2,1-4H3/t15-,16-,17-,18+,20-,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 123 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human KOPR expressed in U2OS cells assessed as beta-arrestin2 recruitment after 90 mins by DiscoveRx PathHunter assay |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50105085

(17-cyclobutylmethyl-4,5alpha-epoxymorphinan-3,6alp...)Show SMILES O[C@H]1CC[C@@]2(O)[C@H]3Cc4ccc(O)c5O[C@@H]1[C@]2(CCN3CC1CCC1)c45 |r| Show InChI InChI=1S/C21H27NO4/c23-14-5-4-13-10-16-21(25)7-6-15(24)19-20(21,17(13)18(14)26-19)8-9-22(16)11-12-2-1-3-12/h4-5,12,15-16,19,23-25H,1-3,6-11H2/t15-,16+,19-,20-,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | n/a | n/a | 250 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human KOPR expressed in U2OS cells assessed as beta-arrestin2 recruitment after 90 mins by DiscoveRx PathHunter assay |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50362540

(CHEMBL1941031)Show SMILES CCOCO[C@H]1C[C@@H](C(=O)OC)[C@]2(C)CC[C@H]3C(=O)O[C@H](C[C@]3(C)[C@H]2C1=O)c1ccoc1 |r| Show InChI InChI=1S/C24H32O8/c1-5-29-13-31-17-10-16(21(26)28-4)23(2)8-6-15-22(27)32-18(14-7-9-30-12-14)11-24(15,3)20(23)19(17)25/h7,9,12,15-18,20H,5-6,8,10-11,13H2,1-4H3/t15-,16-,17-,18+,20-,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 9.5 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human KOPR expressed in CHO cells assessed as [35S]GTPgammaS binding after 60 mins by liquid scintillation counting |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50000296

(CHEMBL441765 | CHEMBL482811 | U-50488H | US1149237...)Show SMILES CN([C@@H]1CCCC[C@H]1N1CCCC1)C(=O)Cc1ccc(Cl)c(Cl)c1 |r| Show InChI InChI=1S/C19H26Cl2N2O/c1-22(19(24)13-14-8-9-15(20)16(21)12-14)17-6-2-3-7-18(17)23-10-4-5-11-23/h8-9,12,17-18H,2-7,10-11,13H2,1H3/t17-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.70 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human KOPR expressed in CHO cells assessed as [35S]GTPgammaS binding after 60 mins by liquid scintillation counting |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50159165

((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...)Show SMILES COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@@H](C[C@]21C)c1ccoc1 |r| Show InChI InChI=1S/C23H28O8/c1-12(24)30-16-9-15(20(26)28-4)22(2)7-5-14-21(27)31-17(13-6-8-29-11-13)10-23(14,3)19(22)18(16)25/h6,8,11,14-17,19H,5,7,9-10H2,1-4H3/t14-,15-,16-,17-,19-,22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.90 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human KOPR expressed in CHO cells assessed as [35S]GTPgammaS binding after 60 mins by liquid scintillation counting |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50159165

((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...)Show SMILES COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@@H](C[C@]21C)c1ccoc1 |r| Show InChI InChI=1S/C23H28O8/c1-12(24)30-16-9-15(20(26)28-4)22(2)7-5-14-21(27)31-17(13-6-8-29-11-13)10-23(14,3)19(22)18(16)25/h6,8,11,14-17,19H,5,7,9-10H2,1-4H3/t14-,15-,16-,17-,19-,22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 31 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human KOPR expressed in U2OS cells assessed as beta-arrestin2 recruitment after 90 mins by DiscoveRx PathHunter assay |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50266337

(12-epi-Salvinorin A | CHEMBL458235)Show SMILES COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@H](C[C@]21C)c1ccoc1 |r| Show InChI InChI=1S/C23H28O8/c1-12(24)30-16-9-15(20(26)28-4)22(2)7-5-14-21(27)31-17(13-6-8-29-11-13)10-23(14,3)19(22)18(16)25/h6,8,11,14-17,19H,5,7,9-10H2,1-4H3/t14-,15-,16-,17+,19-,22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 41 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human KOPR expressed in CHO cells assessed as [35S]GTPgammaS binding after 60 mins by liquid scintillation counting |
Bioorg Med Chem Lett 22: 1023-6 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.128
BindingDB Entry DOI: 10.7270/Q2S182X2 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data