

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 1A2 (Homo sapiens (Human)) | BDBM50156671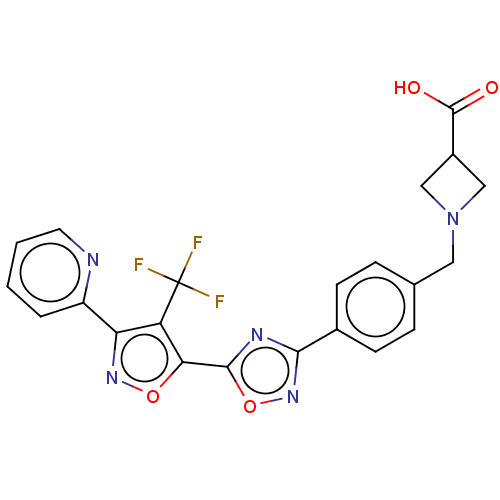 (CHEMBL3794145) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Inhibition of CYP1A2 (unknown origin) | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C19 (Homo sapiens (Human)) | BDBM50156671 (CHEMBL3794145) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Inhibition of CYP2C19 (unknown origin) | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM50156671 (CHEMBL3794145) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Inhibition of CYP3A4 (unknown origin) | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C8 (Homo sapiens (Human)) | BDBM50156671 (CHEMBL3794145) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Inhibition of CYP2C8 (unknown origin) | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50156671 (CHEMBL3794145) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Inhibition of CYP2D6 (unknown origin) | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2B6 (Homo sapiens (Human)) | BDBM50156671 (CHEMBL3794145) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Inhibition of CYP2B6 (unknown origin) | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50156671 (CHEMBL3794145) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Inhibition of CYP2C9 (unknown origin) | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 3 (Homo sapiens (Human)) | BDBM50156675 (CHEMBL3793394) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 8.20E+3 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P3 receptor expressed in EDG3-Ga15-bla HEK293T cell membranes assessed as stimulation of [35S]GTPgamma binding incubated ... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 3 (Homo sapiens (Human)) | BDBM50156676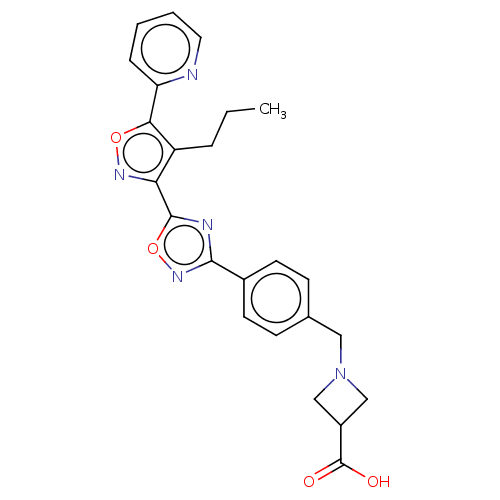 (CHEMBL3793185) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P3 receptor expressed in EDG3-Ga15-bla HEK293T cell membranes assessed as stimulation of [35S]GTPgamma binding incubated ... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 1 (Homo sapiens (Human)) | BDBM50156676 (CHEMBL3793185) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.820 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P1 receptor expressed in CHO cell membranes assessed as stimulation of [35S]GTPgamma binding incubated for 45 mins by liq... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 1 (Homo sapiens (Human)) | BDBM50156675 (CHEMBL3793394) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P1 receptor expressed in CHO cell membranes assessed as stimulation of [35S]GTPgamma binding incubated for 45 mins by liq... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 1 (Homo sapiens (Human)) | BDBM50156671 (CHEMBL3794145) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.710 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P1 receptor expressed in CHO cell membranes assessed as stimulation of [35S]GTPgamma binding incubated for 45 mins by liq... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 3 (Homo sapiens (Human)) | BDBM50156672 (CHEMBL3794064) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P3 receptor expressed in EDG3-Ga15-bla HEK293T cell membranes assessed as stimulation of [35S]GTPgamma binding incubated ... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 1 (Homo sapiens (Human)) | BDBM50156677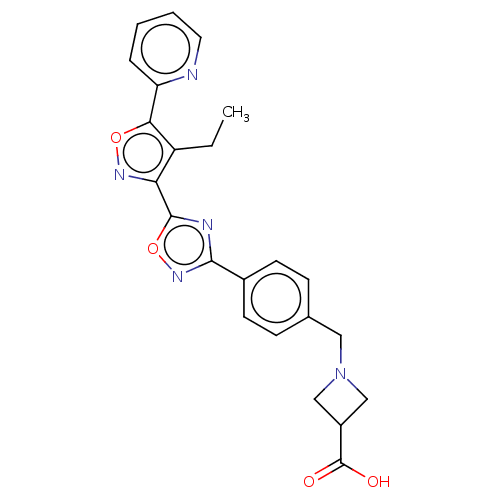 (CHEMBL3792704) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem | Article PubMed | n/a | n/a | n/a | n/a | 3.40 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P1 receptor expressed in CHO cell membranes assessed as stimulation of [35S]GTPgamma binding incubated for 45 mins by liq... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 3 (Homo sapiens (Human)) | BDBM50156677 (CHEMBL3792704) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P3 receptor expressed in EDG3-Ga15-bla HEK293T cell membranes assessed as stimulation of [35S]GTPgamma binding incubated ... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 3 (Homo sapiens (Human)) | BDBM50156674 (CHEMBL3793145) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 630 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P3 receptor expressed in EDG3-Ga15-bla HEK293T cell membranes assessed as stimulation of [35S]GTPgamma binding incubated ... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 1 (Homo sapiens (Human)) | BDBM50156672 (CHEMBL3794064) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.470 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P1 receptor expressed in CHO cell membranes assessed as stimulation of [35S]GTPgamma binding incubated for 45 mins by liq... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 3 (Homo sapiens (Human)) | BDBM50156671 (CHEMBL3794145) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P3 receptor expressed in EDG3-Ga15-bla HEK293T cell membranes assessed as stimulation of [35S]GTPgamma binding incubated ... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sphingosine 1-phosphate receptor 1 (Homo sapiens (Human)) | BDBM50156674 (CHEMBL3793145) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.210 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development Curated by ChEMBL | Assay Description Agonist activity at human S1P1 receptor expressed in CHO cell membranes assessed as stimulation of [35S]GTPgamma binding incubated for 45 mins by liq... | Bioorg Med Chem Lett 26: 2470-4 (2016) Article DOI: 10.1016/j.bmcl.2016.03.105 BindingDB Entry DOI: 10.7270/Q23T9K3M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||