| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Urokinase-type plasminogen activator |
|---|
| Ligand | BDBM14148 |
|---|
| Substrate/Competitor | BDBM13949 |
|---|
| Meas. Tech. | Enzyme Assay and Determination of the Inhibition Constants |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 295.15±n/a K |
|---|
| Ki | 110±n/a nM |
|---|
| Citation |  Katz, BA; Sprengeler, PA; Luong, C; Verner, E; Elrod, K; Kirtley, M; Janc, J; Spencer, JR; Breitenbucher, JG; Hui, H; McGee, D; Allen, D; Martelli, A; Mackman, RL Engineering inhibitors highly selective for the S1 sites of Ser190 trypsin-like serine protease drug targets. Chem Biol8:1107-21 (2001) [PubMed] Article Katz, BA; Sprengeler, PA; Luong, C; Verner, E; Elrod, K; Kirtley, M; Janc, J; Spencer, JR; Breitenbucher, JG; Hui, H; McGee, D; Allen, D; Martelli, A; Mackman, RL Engineering inhibitors highly selective for the S1 sites of Ser190 trypsin-like serine protease drug targets. Chem Biol8:1107-21 (2001) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Urokinase-type plasminogen activator |
|---|
| Name: | Urokinase-type plasminogen activator |
|---|
| Synonyms: | 3.4.21.73 | PLAU | U-plasminogen activator | UROK_HUMAN | Urokinase | Urokinase-type plasminogen activator (uPA) | Urokinase-type plasminogen activator chain B | Urokinase-type plasminogen activator long chain A | Urokinase-type plasminogen activator short chain A | Urokinase-type plasminogen activator/surface receptor | uPA |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48528.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00749 |
|---|
| Residue: | 431 |
|---|
| Sequence: | MRALLARLLLCVLVVSDSKGSNELHQVPSNCDCLNGGTCVSNKYFSNIHWCNCPKKFGGQ
HCEIDKSKTCYEGNGHFYRGKASTDTMGRPCLPWNSATVLQQTYHAHRSDALQLGLGKHN
YCRNPDNRRRPWCYVQVGLKLLVQECMVHDCADGKKPSSPPEELKFQCGQKTLRPRFKII
GGEFTTIENQPWFAAIYRRHRGGSVTYVCGGSLISPCWVISATHCFIDYPKKEDYIVYLG
RSRLNSNTQGEMKFEVENLILHKDYSADTLAHHNDIALLKIRSKEGRCAQPSRTIQTICL
PSMYNDPQFGTSCEITGFGKENSTDYLYPEQLKMTVVKLISHRECQQPHYYGSEVTTKML
CAADPQWKTDSCQGDSGGPLVCSLQGRMTLTGIVSWGRGCALKDKPGVYTRVSHFLPWIR
SHTKEENGLAL
|
|
|
|---|
| BDBM14148 |
|---|
| BDBM13949 |
|---|
| Name | BDBM14148 |
|---|
| Synonyms: | 2-{5-[amino(iminiumyl)methyl]-6-fluoro-1H-1,3-benzodiazol-2-yl}-6-(2-methylpropoxy)benzen-1-olate | APC-10950 | CA-10 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H19FN4O2 |
|---|
| Mol. Mass. | 342.3675 |
|---|
| SMILES | CC(C)COc1cccc(-c2nc3cc(F)c(cc3[nH]2)C(N)=[NH2+])c1[O-] |
|---|
| Structure | 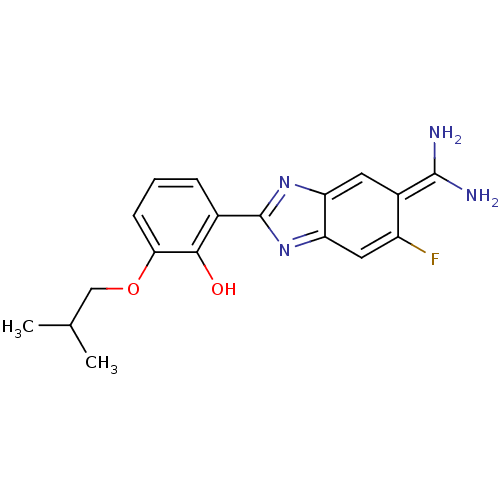
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Katz, BA; Sprengeler, PA; Luong, C; Verner, E; Elrod, K; Kirtley, M; Janc, J; Spencer, JR; Breitenbucher, JG; Hui, H; McGee, D; Allen, D; Martelli, A; Mackman, RL Engineering inhibitors highly selective for the S1 sites of Ser190 trypsin-like serine protease drug targets. Chem Biol8:1107-21 (2001) [PubMed] Article
Katz, BA; Sprengeler, PA; Luong, C; Verner, E; Elrod, K; Kirtley, M; Janc, J; Spencer, JR; Breitenbucher, JG; Hui, H; McGee, D; Allen, D; Martelli, A; Mackman, RL Engineering inhibitors highly selective for the S1 sites of Ser190 trypsin-like serine protease drug targets. Chem Biol8:1107-21 (2001) [PubMed] Article