| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Lutropin-choriogonadotropic hormone receptor |
|---|
| Ligand | BDBM50505793 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1824021 (CHEMBL4323785) |
|---|
| IC50 | 378±n/a nM |
|---|
| Citation |  Wortmann, L; Lindenthal, B; Muhn, P; Walter, A; Nubbemeyer, R; Heldmann, D; Sobek, L; Morandi, F; Schrey, AK; Moosmayer, D; G�nther, J; Kuhnke, J; Koppitz, M; L�cking, U; R�hn, U; Sch�fer, M; Nowak-Reppel, K; K�hne, R; Weinmann, H; Langer, G Discovery of BAY-298 and BAY-899: Tetrahydro-1,6-naphthyridine-Based, Potent, and Selective Antagonists of the Luteinizing Hormone Receptor Which Reduce Sex Hormone Levels in Vivo. J Med Chem62:10321-10341 (2019) [PubMed] Article Wortmann, L; Lindenthal, B; Muhn, P; Walter, A; Nubbemeyer, R; Heldmann, D; Sobek, L; Morandi, F; Schrey, AK; Moosmayer, D; G�nther, J; Kuhnke, J; Koppitz, M; L�cking, U; R�hn, U; Sch�fer, M; Nowak-Reppel, K; K�hne, R; Weinmann, H; Langer, G Discovery of BAY-298 and BAY-899: Tetrahydro-1,6-naphthyridine-Based, Potent, and Selective Antagonists of the Luteinizing Hormone Receptor Which Reduce Sex Hormone Levels in Vivo. J Med Chem62:10321-10341 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Lutropin-choriogonadotropic hormone receptor |
|---|
| Name: | Lutropin-choriogonadotropic hormone receptor |
|---|
| Synonyms: | LCGR | LGR2 | LH/CG-R | LHCGR | LHR | LHRHR | LSH-R | LSHR_HUMAN | Luteinizing hormone receptor |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 78662.77 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_562598 |
|---|
| Residue: | 699 |
|---|
| Sequence: | MKQRFSALQLLKLLLLLQPPLPRALREALCPEPCNCVPDGALRCPGPTAGLTRLSLAYLP
VKVIPSQAFRGLNEVIKIEISQIDSLERIEANAFDNLLNLSEILIQNTKNLRYIEPGAFI
NLPRLKYLSICNTGIRKFPDVTKVFSSESNFILEICDNLHITTIPGNAFQGMNNESVTLK
LYGNGFEEVQSHAFNGTTLTSLELKENVHLEKMHNGAFRGATGPKTLDISSTKLQALPSY
GLESIQRLIATSSYSLKKLPSRETFVNLLEATLTYPSHCCAFRNLPTKEQNFSHSISENF
SKQCESTVRKVNNKTLYSSMLAESELSGWDYEYGFCLPKTPRCAPEPDAFNPCEDIMGYD
FLRVLIWLINILAIMGNMTVLFVLLTSRYKLTVPRFLMCNLSFADFCMGLYLLLIASVDS
QTKGQYYNHAIDWQTGSGCSTAGFFTVFASELSVYTLTVITLERWHTITYAIHLDQKLRL
RHAILIMLGGWLFSSLIAMLPLVGVSNYMKVSICFPMDVETTLSQVYILTILILNVVAFF
IICACYIKIYFAVRNPELMATNKDTKIAKKMAILIFTDFTCMAPISFFAISAAFKVPLIT
VTNSKVLLVLFYPINSCANPFLYAIFTKTFQRDFFLLLSKFGCCKRRAELYRRKDFSAYT
SNCKNGFTGSNKPSQSTLKLSTLHCQGTALLDKTRYTEC
|
|
|
|---|
| BDBM50505793 |
|---|
| n/a |
|---|
| Name | BDBM50505793 |
|---|
| Synonyms: | CHEMBL4593222 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H18Cl2N2O2S |
|---|
| Mol. Mass. | 433.351 |
|---|
| SMILES | COc1ccc(cc1)C1N(CCc2sccc12)C(=O)Nc1cc(Cl)cc(Cl)c1 |
|---|
| Structure | 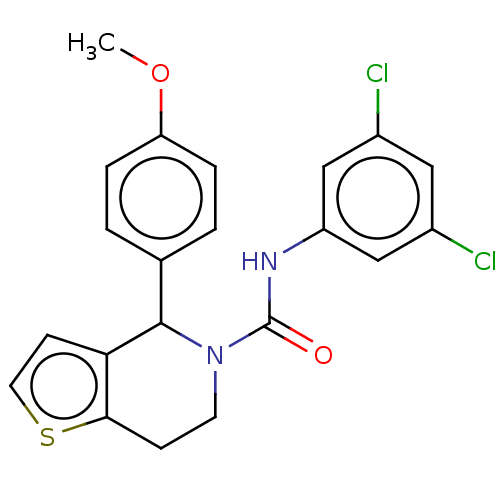
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wortmann, L; Lindenthal, B; Muhn, P; Walter, A; Nubbemeyer, R; Heldmann, D; Sobek, L; Morandi, F; Schrey, AK; Moosmayer, D; G�nther, J; Kuhnke, J; Koppitz, M; L�cking, U; R�hn, U; Sch�fer, M; Nowak-Reppel, K; K�hne, R; Weinmann, H; Langer, G Discovery of BAY-298 and BAY-899: Tetrahydro-1,6-naphthyridine-Based, Potent, and Selective Antagonists of the Luteinizing Hormone Receptor Which Reduce Sex Hormone Levels in Vivo. J Med Chem62:10321-10341 (2019) [PubMed] Article
Wortmann, L; Lindenthal, B; Muhn, P; Walter, A; Nubbemeyer, R; Heldmann, D; Sobek, L; Morandi, F; Schrey, AK; Moosmayer, D; G�nther, J; Kuhnke, J; Koppitz, M; L�cking, U; R�hn, U; Sch�fer, M; Nowak-Reppel, K; K�hne, R; Weinmann, H; Langer, G Discovery of BAY-298 and BAY-899: Tetrahydro-1,6-naphthyridine-Based, Potent, and Selective Antagonists of the Luteinizing Hormone Receptor Which Reduce Sex Hormone Levels in Vivo. J Med Chem62:10321-10341 (2019) [PubMed] Article