| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mu-type opioid receptor |
|---|
| Ligand | BDBM50123273 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_149156 (CHEMBL757699) |
|---|
| Ki | 1.5±n/a nM |
|---|
| Citation |  Ananthan, S; Kezar, HS; Carter, RL; Saini, SK; Rice, KC; Wells, JL; Davis, P; Xu, H; Dersch, CM; Bilsky, EJ; Porreca, F; Rothman, RB Synthesis, opioid receptor binding, and biological activities of naltrexone-derived pyrido- and pyrimidomorphinans. J Med Chem42:3527-38 (1999) [PubMed] Article Ananthan, S; Kezar, HS; Carter, RL; Saini, SK; Rice, KC; Wells, JL; Davis, P; Xu, H; Dersch, CM; Bilsky, EJ; Porreca, F; Rothman, RB Synthesis, opioid receptor binding, and biological activities of naltrexone-derived pyrido- and pyrimidomorphinans. J Med Chem42:3527-38 (1999) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mu-type opioid receptor |
|---|
| Name: | Mu-type opioid receptor |
|---|
| Synonyms: | MOR-1 | MUOR1 | Mu-type opioid receptor (MOR) | OPIATE Mu | OPRM_RAT | Opiate non-selective | Opioid receptor B | Oprm1 | Ror-b |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44503.11 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Competition binding assays were carried out using membrane preparations from transfected HN9.10 cells that constitutively expressed the mu opioid receptor. |
|---|
| Residue: | 398 |
|---|
| Sequence: | MDSSTGPGNTSDCSDPLAQASCSPAPGSWLNLSHVDGNQSDPCGLNRTGLGGNDSLCPQT
GSPSMVTAITIMALYSIVCVVGLFGNFLVMYVIVRYTKMKTATNIYIFNLALADALATST
LPFQSVNYLMGTWPFGTILCKIVISIDYYNMFTSIFTLCTMSVDRYIAVCHPVKALDFRT
PRNAKIVNVCNWILSSAIGLPVMFMATTKYRQGSIDCTLTFSHPTWYWENLLKICVFIFA
FIMPVLIITVCYGLMILRLKSVRMLSGSKEKDRNLRRITRMVLVVVAVFIVCWTPIHIYV
IIKALITIPETTFQTVSWHFCIALGYTNSCLNPVLYAFLDENFKRCFREFCIPTSSTIEQ
QNSTRVRQNTREHPSTANTVDRTNHQLENLEAETAPLP
|
|
|
|---|
| BDBM50123273 |
|---|
| n/a |
|---|
| Name | BDBM50123273 |
|---|
| Synonyms: | 19-cyclopropylmethyl-(2S,10R)-11-oxa-8,19-diazahexacyclo[10.9.1.01,10.02,18.04,9.016,22]docosa-4(9),5,7,12,14,16(22)-hexaene-2,13-diol | 19-cyclopropylmethyl-11-oxa-8,19-diazahexacyclo[10.9.1.01,10.02,18.04,9.016,22]docosa-4(9),5,7,12,14,16(22)-hexaene-2,13-diol | CHEMBL346173 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H24N2O3 |
|---|
| Mol. Mass. | 376.4483 |
|---|
| SMILES | Oc1ccc2C[C@H]3N(CC4CC4)CC[C@@]45[C@@H](Oc1c24)c1ncccc1C[C@@]35O |
|---|
| Structure | 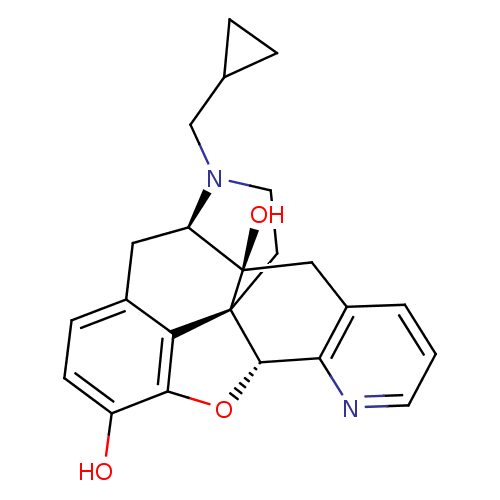
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ananthan, S; Kezar, HS; Carter, RL; Saini, SK; Rice, KC; Wells, JL; Davis, P; Xu, H; Dersch, CM; Bilsky, EJ; Porreca, F; Rothman, RB Synthesis, opioid receptor binding, and biological activities of naltrexone-derived pyrido- and pyrimidomorphinans. J Med Chem42:3527-38 (1999) [PubMed] Article
Ananthan, S; Kezar, HS; Carter, RL; Saini, SK; Rice, KC; Wells, JL; Davis, P; Xu, H; Dersch, CM; Bilsky, EJ; Porreca, F; Rothman, RB Synthesis, opioid receptor binding, and biological activities of naltrexone-derived pyrido- and pyrimidomorphinans. J Med Chem42:3527-38 (1999) [PubMed] Article