| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50318666 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_633293 (CHEMBL1119997) |
|---|
| IC50 | 5420±n/a nM |
|---|
| Citation |  Yeung, BK; Zou, B; Rottmann, M; Lakshminarayana, SB; Ang, SH; Leong, SY; Tan, J; Wong, J; Keller-Maerki, S; Fischli, C; Goh, A; Schmitt, EK; Krastel, P; Francotte, E; Kuhen, K; Plouffe, D; Henson, K; Wagner, T; Winzeler, EA; Petersen, F; Brun, R; Dartois, V; Diagana, TT; Keller, TH Spirotetrahydro beta-carbolines (spiroindolones): a new class of potent and orally efficacious compounds for the treatment of malaria. J Med Chem53:5155-64 (2010) [PubMed] Article Yeung, BK; Zou, B; Rottmann, M; Lakshminarayana, SB; Ang, SH; Leong, SY; Tan, J; Wong, J; Keller-Maerki, S; Fischli, C; Goh, A; Schmitt, EK; Krastel, P; Francotte, E; Kuhen, K; Plouffe, D; Henson, K; Wagner, T; Winzeler, EA; Petersen, F; Brun, R; Dartois, V; Diagana, TT; Keller, TH Spirotetrahydro beta-carbolines (spiroindolones): a new class of potent and orally efficacious compounds for the treatment of malaria. J Med Chem53:5155-64 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50318666 |
|---|
| n/a |
|---|
| Name | BDBM50318666 |
|---|
| Synonyms: | (1R,3S)-5',7-Dichloro-6-fluoro-3-methyl-2,3,4,9-tetrahydrospiro-[beta-carboline-1,3'-indol]-2'(1'H)-one | CHEMBL1082723 | NITD609 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H14Cl2FN3O |
|---|
| Mol. Mass. | 390.238 |
|---|
| SMILES | C[C@H]1Cc2c([nH]c3cc(Cl)c(F)cc23)[C@@]2(N1)C(=O)Nc1ccc(Cl)cc21 |r| |
|---|
| Structure | 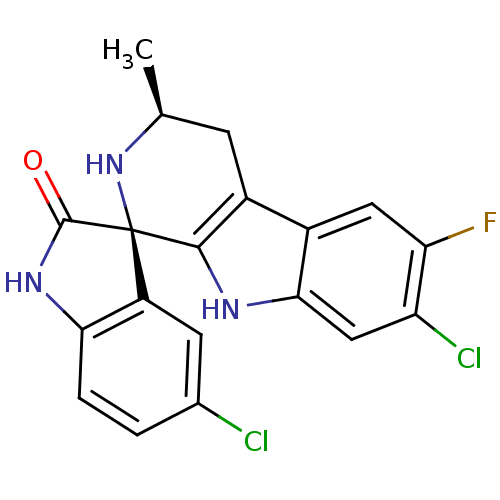
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yeung, BK; Zou, B; Rottmann, M; Lakshminarayana, SB; Ang, SH; Leong, SY; Tan, J; Wong, J; Keller-Maerki, S; Fischli, C; Goh, A; Schmitt, EK; Krastel, P; Francotte, E; Kuhen, K; Plouffe, D; Henson, K; Wagner, T; Winzeler, EA; Petersen, F; Brun, R; Dartois, V; Diagana, TT; Keller, TH Spirotetrahydro beta-carbolines (spiroindolones): a new class of potent and orally efficacious compounds for the treatment of malaria. J Med Chem53:5155-64 (2010) [PubMed] Article
Yeung, BK; Zou, B; Rottmann, M; Lakshminarayana, SB; Ang, SH; Leong, SY; Tan, J; Wong, J; Keller-Maerki, S; Fischli, C; Goh, A; Schmitt, EK; Krastel, P; Francotte, E; Kuhen, K; Plouffe, D; Henson, K; Wagner, T; Winzeler, EA; Petersen, F; Brun, R; Dartois, V; Diagana, TT; Keller, TH Spirotetrahydro beta-carbolines (spiroindolones): a new class of potent and orally efficacious compounds for the treatment of malaria. J Med Chem53:5155-64 (2010) [PubMed] Article