| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50305452 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_817424 (CHEMBL2027647) |
|---|
| IC50 | 4600±n/a nM |
|---|
| Citation |  Jia, L; Simpson, RD; Yuan, J; Xu, Z; Zhao, W; Cacatian, S; Tice, CM; Guo, J; Ishchenko, A; Singh, SB; Wu, Z; McKeever, BM; Bukhtiyarov, Y; Johnson, JA; Doe, CP; Harrison, RK; McGeehan, GM; Dillard, LW; Baldwin, JJ; Claremon, DA Discovery of VTP-27999, an Alkyl Amine Renin Inhibitor with Potential for Clinical Utility. ACS Med Chem Lett2:747-751 (2011) [PubMed] Article Jia, L; Simpson, RD; Yuan, J; Xu, Z; Zhao, W; Cacatian, S; Tice, CM; Guo, J; Ishchenko, A; Singh, SB; Wu, Z; McKeever, BM; Bukhtiyarov, Y; Johnson, JA; Doe, CP; Harrison, RK; McGeehan, GM; Dillard, LW; Baldwin, JJ; Claremon, DA Discovery of VTP-27999, an Alkyl Amine Renin Inhibitor with Potential for Clinical Utility. ACS Med Chem Lett2:747-751 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50305452 |
|---|
| n/a |
|---|
| Name | BDBM50305452 |
|---|
| Synonyms: | CHEMBL591578 | methyl 2-((R)-(3-chlorophenyl)((R)-1-((S)-1-cyclohexyl-3-(methylamino)propan-2-ylcarbamoyl)piperidin-3-yl)methoxy)ethylcarbamate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H43ClN4O4 |
|---|
| Mol. Mass. | 523.108 |
|---|
| SMILES | CNC[C@H](CC1CCCCC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| |
|---|
| Structure | 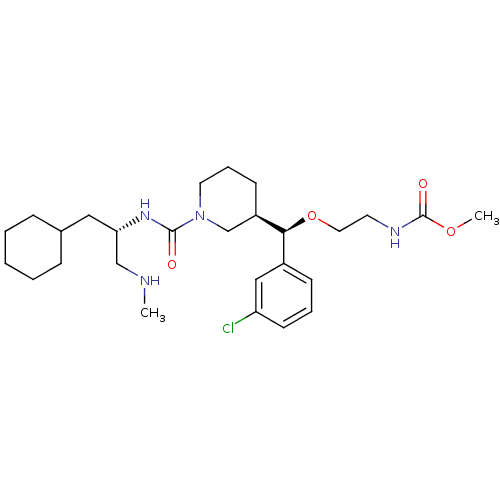
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jia, L; Simpson, RD; Yuan, J; Xu, Z; Zhao, W; Cacatian, S; Tice, CM; Guo, J; Ishchenko, A; Singh, SB; Wu, Z; McKeever, BM; Bukhtiyarov, Y; Johnson, JA; Doe, CP; Harrison, RK; McGeehan, GM; Dillard, LW; Baldwin, JJ; Claremon, DA Discovery of VTP-27999, an Alkyl Amine Renin Inhibitor with Potential for Clinical Utility. ACS Med Chem Lett2:747-751 (2011) [PubMed] Article
Jia, L; Simpson, RD; Yuan, J; Xu, Z; Zhao, W; Cacatian, S; Tice, CM; Guo, J; Ishchenko, A; Singh, SB; Wu, Z; McKeever, BM; Bukhtiyarov, Y; Johnson, JA; Doe, CP; Harrison, RK; McGeehan, GM; Dillard, LW; Baldwin, JJ; Claremon, DA Discovery of VTP-27999, an Alkyl Amine Renin Inhibitor with Potential for Clinical Utility. ACS Med Chem Lett2:747-751 (2011) [PubMed] Article