| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2B6 |
|---|
| Ligand | BDBM50133595 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1526169 (CHEMBL3636694) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Ruggeri, RB; Buckbinder, L; Bagley, SW; Carpino, PA; Conn, EL; Dowling, MS; Fernando, DP; Jiao, W; Kung, DW; Orr, ST; Qi, Y; Rocke, BN; Smith, A; Warmus, JS; Zhang, Y; Bowles, D; Widlicka, DW; Eng, H; Ryder, T; Sharma, R; Wolford, A; Okerberg, C; Walters, K; Maurer, TS; Zhang, Y; Bonin, PD; Spath, SN; Xing, G; Hepworth, D; Ahn, K; Kalgutkar, AS Discovery of 2-(6-(5-Chloro-2-methoxyphenyl)-4-oxo-2-thioxo-3,4-dihydropyrimidin-1(2H)-yl)acetamide (PF-06282999): A Highly Selective Mechanism-Based Myeloperoxidase Inhibitor for the Treatment of Cardiovascular Diseases. J Med Chem58:8513-28 (2015) [PubMed] Article Ruggeri, RB; Buckbinder, L; Bagley, SW; Carpino, PA; Conn, EL; Dowling, MS; Fernando, DP; Jiao, W; Kung, DW; Orr, ST; Qi, Y; Rocke, BN; Smith, A; Warmus, JS; Zhang, Y; Bowles, D; Widlicka, DW; Eng, H; Ryder, T; Sharma, R; Wolford, A; Okerberg, C; Walters, K; Maurer, TS; Zhang, Y; Bonin, PD; Spath, SN; Xing, G; Hepworth, D; Ahn, K; Kalgutkar, AS Discovery of 2-(6-(5-Chloro-2-methoxyphenyl)-4-oxo-2-thioxo-3,4-dihydropyrimidin-1(2H)-yl)acetamide (PF-06282999): A Highly Selective Mechanism-Based Myeloperoxidase Inhibitor for the Treatment of Cardiovascular Diseases. J Med Chem58:8513-28 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2B6 |
|---|
| Name: | Cytochrome P450 2B6 |
|---|
| Synonyms: | CP2B6_HUMAN | CYP2B6 | Cytochrome P450 2B6 (CYP2B6) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 56289.75 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P20813 |
|---|
| Residue: | 491 |
|---|
| Sequence: | MELSVLLFLALLTGLLLLLVQRHPNTHDRLPPGPRPLPLLGNLLQMDRRGLLKSFLRFRE
KYGDVFTVHLGPRPVVMLCGVEAIREALVDKAEAFSGRGKIAMVDPFFRGYGVIFANGNR
WKVLRRFSVTTMRDFGMGKRSVEERIQEEAQCLIEELRKSKGALMDPTFLFQSITANIIC
SIVFGKRFHYQDQEFLKMLNLFYQTFSLISSVFGQLFELFSGFLKYFPGAHRQVYKNLQE
INAYIGHSVEKHRETLDPSAPKDLIDTYLLHMEKEKSNAHSEFSHQNLNLNTLSLFFAGT
ETTSTTLRYGFLLMLKYPHVAERVYREIEQVIGPHRPPELHDRAKMPYTEAVIYEIQRFS
DLLPMGVPHIVTQHTSFRGYIIPKDTEVFLILSTALHDPHYFEKPDAFNPDHFLDANGAL
KKTEAFIPFSLGKRICLGEGIARAELFLFFTTILQNFSMASPVAPEDIDLTPQECGVGKI
PPTYQIRFLPR
|
|
|
|---|
| BDBM50133595 |
|---|
| n/a |
|---|
| Name | BDBM50133595 |
|---|
| Synonyms: | CHEMBL3633460 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H12ClN3O3S |
|---|
| Mol. Mass. | 325.771 |
|---|
| SMILES | COc1ccc(Cl)cc1-c1cc(=O)[nH]c(=S)n1CC(N)=O |
|---|
| Structure | 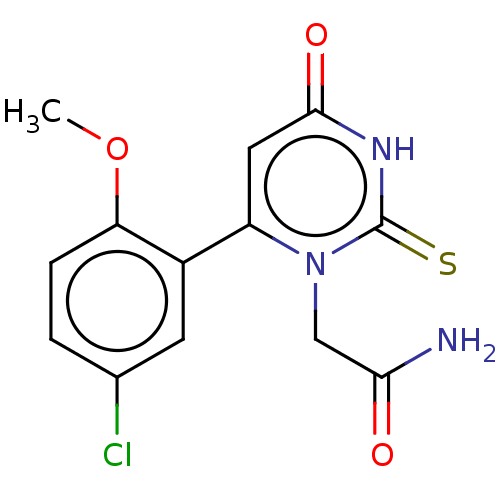
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ruggeri, RB; Buckbinder, L; Bagley, SW; Carpino, PA; Conn, EL; Dowling, MS; Fernando, DP; Jiao, W; Kung, DW; Orr, ST; Qi, Y; Rocke, BN; Smith, A; Warmus, JS; Zhang, Y; Bowles, D; Widlicka, DW; Eng, H; Ryder, T; Sharma, R; Wolford, A; Okerberg, C; Walters, K; Maurer, TS; Zhang, Y; Bonin, PD; Spath, SN; Xing, G; Hepworth, D; Ahn, K; Kalgutkar, AS Discovery of 2-(6-(5-Chloro-2-methoxyphenyl)-4-oxo-2-thioxo-3,4-dihydropyrimidin-1(2H)-yl)acetamide (PF-06282999): A Highly Selective Mechanism-Based Myeloperoxidase Inhibitor for the Treatment of Cardiovascular Diseases. J Med Chem58:8513-28 (2015) [PubMed] Article
Ruggeri, RB; Buckbinder, L; Bagley, SW; Carpino, PA; Conn, EL; Dowling, MS; Fernando, DP; Jiao, W; Kung, DW; Orr, ST; Qi, Y; Rocke, BN; Smith, A; Warmus, JS; Zhang, Y; Bowles, D; Widlicka, DW; Eng, H; Ryder, T; Sharma, R; Wolford, A; Okerberg, C; Walters, K; Maurer, TS; Zhang, Y; Bonin, PD; Spath, SN; Xing, G; Hepworth, D; Ahn, K; Kalgutkar, AS Discovery of 2-(6-(5-Chloro-2-methoxyphenyl)-4-oxo-2-thioxo-3,4-dihydropyrimidin-1(2H)-yl)acetamide (PF-06282999): A Highly Selective Mechanism-Based Myeloperoxidase Inhibitor for the Treatment of Cardiovascular Diseases. J Med Chem58:8513-28 (2015) [PubMed] Article