| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor delta |
|---|
| Ligand | BDBM28661 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2160009 (CHEMBL5044759) |
|---|
| EC50 | 0.500000±n/a nM |
|---|
| Citation |  Feng, Z; Xiang, J; Liu, H; Li, J; Xu, X; Sun, G; Zheng, R; Zhang, S; Liu, J; Yang, S; Xu, Q; Wen, X; Yuan, H; Sun, H; Dai, L Design, Synthesis, and Biological Evaluation of Triazolone Derivatives as Potent PPAR?/? Dual Agonists for the Treatment of Nonalcoholic Steatohepatitis. J Med Chem65:2571-2592 (2022) [PubMed] Article Feng, Z; Xiang, J; Liu, H; Li, J; Xu, X; Sun, G; Zheng, R; Zhang, S; Liu, J; Yang, S; Xu, Q; Wen, X; Yuan, H; Sun, H; Dai, L Design, Synthesis, and Biological Evaluation of Triazolone Derivatives as Potent PPAR?/? Dual Agonists for the Treatment of Nonalcoholic Steatohepatitis. J Med Chem65:2571-2592 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor delta |
|---|
| Name: | Peroxisome proliferator-activated receptor delta |
|---|
| Synonyms: | NR1C2 | NUC1 | NUCI | Nuclear hormone receptor 1 | Nuclear receptor subfamily 1 group C member 2 | PPAR delta | PPAR-beta | PPARB | PPARD | PPARD_HUMAN | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor beta | Peroxisome proliferator-activated receptor delta |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 49910.45 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q03181 |
|---|
| Residue: | 441 |
|---|
| Sequence: | MEQPQEEAPEVREEEEKEEVAEAEGAPELNGGPQHALPSSSYTDLSRSSSPPSLLDQLQM
GCDGASCGSLNMECRVCGDKASGFHYGVHACEGCKGFFRRTIRMKLEYEKCERSCKIQKK
NRNKCQYCRFQKCLALGMSHNAIRFGRMPEAEKRKLVAGLTANEGSQYNPQVADLKAFSK
HIYNAYLKNFNMTKKKARSILTGKASHTAPFVIHDIETLWQAEKGLVWKQLVNGLPPYKE
ISVHVFYRCQCTTVETVRELTEFAKSIPSFSSLFLNDQVTLLKYGVHEAIFAMLASIVNK
DGLLVANGSGFVTREFLRSLRKPFSDIIEPKFEFAVKFNALELDDSDLALFIAAIILCGD
RPGLMNVPRVEAIQDTILRALEFHLQANHPDAQYLFPKLLQKMADLRQLVTEHAQMMQRI
KKTETETSLHPLLQEIYKDMY
|
|
|
|---|
| BDBM28661 |
|---|
| n/a |
|---|
| Name | BDBM28661 |
|---|
| Synonyms: | 2-{2-methyl-4-[({4-methyl-2-[4-(trifluoromethyl)phenyl]-1,3-thiazol-5-yl}methyl)sulfanyl]phenoxy}acetic acid | CHEMBL38943 | GW 501516 | GW-501516 | GW1516 | GW501516 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H18F3NO3S2 |
|---|
| Mol. Mass. | 453.498 |
|---|
| SMILES | Cc1nc(sc1CSc1ccc(OCC(O)=O)c(C)c1)-c1ccc(cc1)C(F)(F)F |
|---|
| Structure | 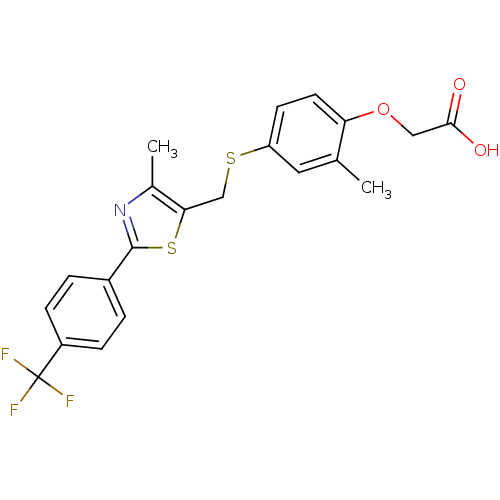
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Feng, Z; Xiang, J; Liu, H; Li, J; Xu, X; Sun, G; Zheng, R; Zhang, S; Liu, J; Yang, S; Xu, Q; Wen, X; Yuan, H; Sun, H; Dai, L Design, Synthesis, and Biological Evaluation of Triazolone Derivatives as Potent PPAR?/? Dual Agonists for the Treatment of Nonalcoholic Steatohepatitis. J Med Chem65:2571-2592 (2022) [PubMed] Article
Feng, Z; Xiang, J; Liu, H; Li, J; Xu, X; Sun, G; Zheng, R; Zhang, S; Liu, J; Yang, S; Xu, Q; Wen, X; Yuan, H; Sun, H; Dai, L Design, Synthesis, and Biological Evaluation of Triazolone Derivatives as Potent PPAR?/? Dual Agonists for the Treatment of Nonalcoholic Steatohepatitis. J Med Chem65:2571-2592 (2022) [PubMed] Article