| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostacyclin receptor |
|---|
| Ligand | BDBM50244809 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_512422 (CHEMBL980763) |
|---|
| IC50 | 6340±n/a nM |
|---|
| Citation |  Debenham, SD; Chan, A; Lau, FW; Liu, W; Wood, HB; Lemme, K; Colwell, L; Habulihaz, B; Akiyama, TE; Einstein, M; Doebber, TW; Sharma, N; Wang, CF; Wu, M; Berger, JP; Meinke, PT Highly functionalized 7-azaindoles as selective PPAR gamma modulators. Bioorg Med Chem Lett18:4798-801 (2008) [PubMed] Article Debenham, SD; Chan, A; Lau, FW; Liu, W; Wood, HB; Lemme, K; Colwell, L; Habulihaz, B; Akiyama, TE; Einstein, M; Doebber, TW; Sharma, N; Wang, CF; Wu, M; Berger, JP; Meinke, PT Highly functionalized 7-azaindoles as selective PPAR gamma modulators. Bioorg Med Chem Lett18:4798-801 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostacyclin receptor |
|---|
| Name: | Prostacyclin receptor |
|---|
| Synonyms: | PGI receptor | PI2R_HUMAN | PRIPR | PTGIR | Prostacyclin (IP) Receptor | Prostacyclin receptor | Prostaglandin I | Prostaglandin I2 | Prostaglandin I2 receptor | Prostanoid IP receptor |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 40968.22 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | The membranes prepared from human platelet were used in binding assay. |
|---|
| Residue: | 386 |
|---|
| Sequence: | MADSCRNLTYVRGSVGPATSTLMFVAGVVGNGLALGILSARRPARPSAFAVLVTGLAATD
LLGTSFLSPAVFVAYARNSSLLGLARGGPALCDAFAFAMTFFGLASMLILFAMAVERCLA
LSHPYLYAQLDGPRCARLALPAIYAFCVLFCALPLLGLGQHQQYCPGSWCFLRMRWAQPG
GAAFSLAYAGLVALLVAAIFLCNGSVTLSLCRMYRQQKRHQGSLGPRPRTGEDEVDHLIL
LALMTVVMAVCSLPLTIRCFTQAVAPDSSSEMGDLLAFRFYAFNPILDPWVFILFRKAVF
QRLKLWVCCLCLGPAHGDSQTPLSQLASGRRDPRAPSAPVGKEGSCVPLSAWGEGQVEPL
PPTQQSSGSAVGTSSKAEASVACSLC
|
|
|
|---|
| BDBM50244809 |
|---|
| n/a |
|---|
| Name | BDBM50244809 |
|---|
| Synonyms: | (2R)-2-[3-[[3-(4-Chlorobenzoyl)-2-methyl-6-(trifluoromethoxy)-1H-indol-1-yl]methyl]phenoxy]propanoicAcid | (R)-2-(3-((3-(4-chlorobenzoyl)-2-methyl-6-(trifluoromethoxy)-1H-indol-1-yl)methyl)phenoxy)propanoic acid | CHEMBL461571 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H21ClF3NO5 |
|---|
| Mol. Mass. | 531.908 |
|---|
| SMILES | C[C@@H](Oc1cccc(Cn2c(C)c(C(=O)c3ccc(Cl)cc3)c3ccc(OC(F)(F)F)cc23)c1)C(O)=O |r| |
|---|
| Structure | 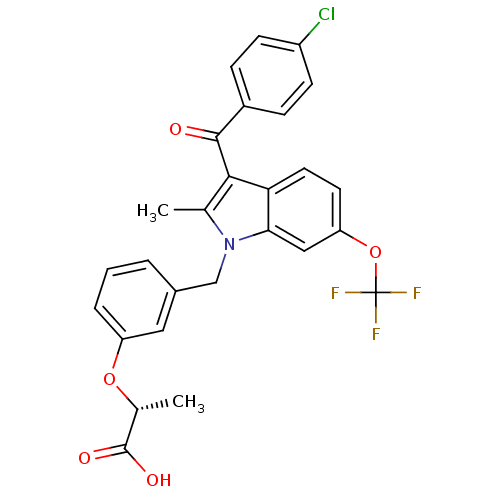
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Debenham, SD; Chan, A; Lau, FW; Liu, W; Wood, HB; Lemme, K; Colwell, L; Habulihaz, B; Akiyama, TE; Einstein, M; Doebber, TW; Sharma, N; Wang, CF; Wu, M; Berger, JP; Meinke, PT Highly functionalized 7-azaindoles as selective PPAR gamma modulators. Bioorg Med Chem Lett18:4798-801 (2008) [PubMed] Article
Debenham, SD; Chan, A; Lau, FW; Liu, W; Wood, HB; Lemme, K; Colwell, L; Habulihaz, B; Akiyama, TE; Einstein, M; Doebber, TW; Sharma, N; Wang, CF; Wu, M; Berger, JP; Meinke, PT Highly functionalized 7-azaindoles as selective PPAR gamma modulators. Bioorg Med Chem Lett18:4798-801 (2008) [PubMed] Article