| Reaction Details |
|---|
 | Report a problem with these data |
| Target | cAMP-dependent protein kinase catalytic subunit alpha |
|---|
| Ligand | BDBM15211 |
|---|
| Substrate/Competitor | BDBM27221 |
|---|
| Meas. Tech. | Fluorescence Polarization-Based Binding Displacement Kinase Assay |
|---|
| pH | 7.5±n/a |
|---|
| Temperature | 303.15±n/a K |
|---|
| Kd | 23±2 nM |
|---|
| Citation |  Lavogina, D; Lust, M; Viil, I; König, N; Raidaru, G; Rogozina, J; Enkvist, E; Uri, A; Bossemeyer, D Structural analysis of ARC-type inhibitor (ARC-1034) binding to protein kinase A catalytic subunit and rational design of bisubstrate analogue inhibitors of basophilic protein kinases. J Med Chem52:308-21 (2009) [PubMed] Article Lavogina, D; Lust, M; Viil, I; König, N; Raidaru, G; Rogozina, J; Enkvist, E; Uri, A; Bossemeyer, D Structural analysis of ARC-type inhibitor (ARC-1034) binding to protein kinase A catalytic subunit and rational design of bisubstrate analogue inhibitors of basophilic protein kinases. J Med Chem52:308-21 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Inhibition_Run data, Solution Info, Assay Method |
|---|
| |
| cAMP-dependent protein kinase catalytic subunit alpha |
|---|
| Name: | cAMP-dependent protein kinase catalytic subunit alpha |
|---|
| Synonyms: | KAPCA_MOUSE | PKA C-alpha | Pkaca | Prkaca | cAMP-Dependent Protein Kinase (PKA) | cAMP-dependent protein kinase catalytic subunit alpha | cAMP-dependent protein kinase, alpha-catalytic subunit |
|---|
| Type: | Enzyme Catalytic Unit |
|---|
| Mol. Mass.: | 40579.18 |
|---|
| Organism: | Mus musculus (mouse) |
|---|
| Description: | n/a |
|---|
| Residue: | 351 |
|---|
| Sequence: | MGNAAAAKKGSEQESVKEFLAKAKEDFLKKWETPSQNTAQLDQFDRIKTLGTGSFGRVML
VKHKESGNHYAMKILDKQKVVKLKQIEHTLNEKRILQAVNFPFLVKLEFSFKDNSNLYMV
MEYVAGGEMFSHLRRIGRFSEPHARFYAAQIVLTFEYLHSLDLIYRDLKPENLLIDQQGY
IQVTDFGFAKRVKGRTWTLCGTPEYLAPEIILSKGYNKAVDWWALGVLIYEMAAGYPPFF
ADQPIQIYEKIVSGKVRFPSHFSSDLKDLLRNLLQVDLTKRFGNLKNGVNDIKNHKWFAT
TDWIAIYQRKVEAPFIPKFKGPGDTSNFDDYEEEEIRVSINEKCGKEFTEF
|
|
|
|---|
| BDBM15211 |
|---|
| BDBM27221 |
|---|
| Name | BDBM15211 |
|---|
| Synonyms: | CHEMBL104264 | H-89 | H89 | HT-89 (H-89) | N-(2-{[(2E)-3-(4-bromophenyl)prop-2-en-1-yl]amino}ethyl)isoquinoline-5-sulfonamide | N-[2-[[(E)-3-(4-bromophenyl)prop-2-enyl]amino]ethyl]isoquinoline-5-sulfonamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H20BrN3O2S |
|---|
| Mol. Mass. | 446.361 |
|---|
| SMILES | Brc1ccc(\C=C\CNCCNS(=O)(=O)c2cccc3cnccc23)cc1 |
|---|
| Structure | 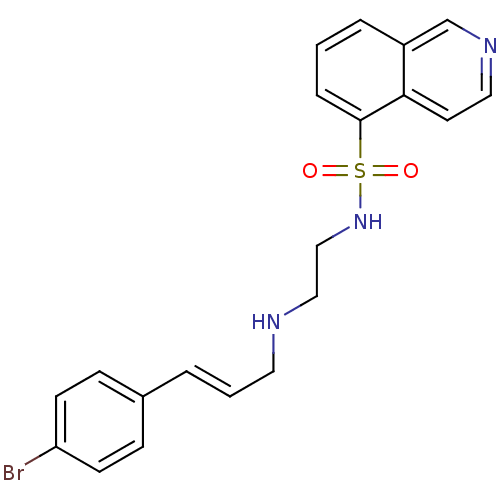
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lavogina, D; Lust, M; Viil, I; König, N; Raidaru, G; Rogozina, J; Enkvist, E; Uri, A; Bossemeyer, D Structural analysis of ARC-type inhibitor (ARC-1034) binding to protein kinase A catalytic subunit and rational design of bisubstrate analogue inhibitors of basophilic protein kinases. J Med Chem52:308-21 (2009) [PubMed] Article
Lavogina, D; Lust, M; Viil, I; König, N; Raidaru, G; Rogozina, J; Enkvist, E; Uri, A; Bossemeyer, D Structural analysis of ARC-type inhibitor (ARC-1034) binding to protein kinase A catalytic subunit and rational design of bisubstrate analogue inhibitors of basophilic protein kinases. J Med Chem52:308-21 (2009) [PubMed] Article