| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bile acid receptor |
|---|
| Ligand | BDBM21674 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1728694 (CHEMBL4143972) |
|---|
| EC50 | 8660±n/a nM |
|---|
| Citation |  Xiao, H; Li, P; Li, X; He, H; Wang, J; Guo, F; Zhang, J; Wei, L; Zhang, H; Shi, Y; Hou, L; Shen, L; Chen, Z; Du, C; Fu, S; Zhang, P; Hao, F; Wang, P; Xu, D; Liang, W; Tian, X; Zhang, A; Cheng, X; Yang, L; Wang, X; Zhang, X; Li, J; Chen, S Synthesis and Biological Evaluation of a Series of Bile Acid Derivatives as FXR Agonists for Treatment of NASH. ACS Med Chem Lett8:1246-1251 (2017) [PubMed] Article Xiao, H; Li, P; Li, X; He, H; Wang, J; Guo, F; Zhang, J; Wei, L; Zhang, H; Shi, Y; Hou, L; Shen, L; Chen, Z; Du, C; Fu, S; Zhang, P; Hao, F; Wang, P; Xu, D; Liang, W; Tian, X; Zhang, A; Cheng, X; Yang, L; Wang, X; Zhang, X; Li, J; Chen, S Synthesis and Biological Evaluation of a Series of Bile Acid Derivatives as FXR Agonists for Treatment of NASH. ACS Med Chem Lett8:1246-1251 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bile acid receptor |
|---|
| Name: | Bile acid receptor |
|---|
| Synonyms: | BAR | Bile acid receptor FXR | FXR | Farnesol receptor HRR-1 | HRR1 | NR1H4 | NR1H4_HUMAN | Nuclear receptor subfamily 1 group H member 4 | RIP14 | RXR-interacting protein 14 | Retinoid X receptor-interacting protein 14 | farnesoid x receptor |
|---|
| Type: | Nuclear Receptor |
|---|
| Mol. Mass.: | 55916.24 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q96RI1 |
|---|
| Residue: | 486 |
|---|
| Sequence: | MVMQFQGLENPIQISPHCSCTPSGFFMEMMSMKPAKGVLTEQVAGPLGQNLEVEPYSQYS
NVQFPQVQPQISSSSYYSNLGFYPQQPEEWYSPGIYELRRMPAETLYQGETEVAEMPVTK
KPRMGASAGRIKGDELCVVCGDRASGYHYNALTCEGCKGFFRRSITKNAVYKCKNGGNCV
MDMYMRRKCQECRLRKCKEMGMLAECMYTGLLTEIQCKSKRLRKNVKQHADQTVNEDSEG
RDLRQVTSTTKSCREKTELTPDQQTLLHFIMDSYNKQRMPQEITNKILKEEFSAEENFLI
LTEMATNHVQVLVEFTKKLPGFQTLDHEDQIALLKGSAVEAMFLRSAEIFNKKLPSGHSD
LLEERIRNSGISDEYITPMFSFYKSIGELKMTQEEYALLTAIVILSPDRQYIKDREAVEK
LQEPLLDVLQKLCKIHQPENPQHFACLLGRLTELRTFNHHHAEMLMSWRVNDHKFTPLLC
EIWDVQ
|
|
|
|---|
| BDBM21674 |
|---|
| n/a |
|---|
| Name | BDBM21674 |
|---|
| Synonyms: | (4R)-4-[(1S,2S,5R,7S,9R,10R,11S,14R,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan-14-yl]pentanoic acid | CDCA | CHENODIOL | Chenodeoxycholic Acid | US10208081, CDCA |
|---|
| Type | Steroid |
|---|
| Emp. Form. | C24H40O4 |
|---|
| Mol. Mass. | 392.572 |
|---|
| SMILES | [H][C@@]1(CC[C@@]2([H])[C@]3([H])[C@H](O)C[C@]4([H])C[C@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)CCC(O)=O |
|---|
| Structure | 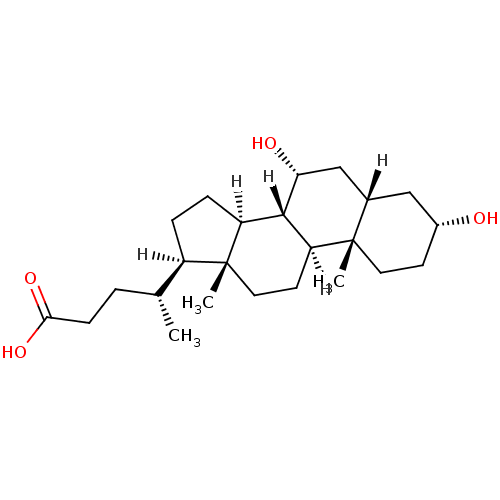
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Xiao, H; Li, P; Li, X; He, H; Wang, J; Guo, F; Zhang, J; Wei, L; Zhang, H; Shi, Y; Hou, L; Shen, L; Chen, Z; Du, C; Fu, S; Zhang, P; Hao, F; Wang, P; Xu, D; Liang, W; Tian, X; Zhang, A; Cheng, X; Yang, L; Wang, X; Zhang, X; Li, J; Chen, S Synthesis and Biological Evaluation of a Series of Bile Acid Derivatives as FXR Agonists for Treatment of NASH. ACS Med Chem Lett8:1246-1251 (2017) [PubMed] Article
Xiao, H; Li, P; Li, X; He, H; Wang, J; Guo, F; Zhang, J; Wei, L; Zhang, H; Shi, Y; Hou, L; Shen, L; Chen, Z; Du, C; Fu, S; Zhang, P; Hao, F; Wang, P; Xu, D; Liang, W; Tian, X; Zhang, A; Cheng, X; Yang, L; Wang, X; Zhang, X; Li, J; Chen, S Synthesis and Biological Evaluation of a Series of Bile Acid Derivatives as FXR Agonists for Treatment of NASH. ACS Med Chem Lett8:1246-1251 (2017) [PubMed] Article