| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 5 |
|---|
| Ligand | BDBM50509860 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1839521 (CHEMBL4339736) |
|---|
| Kd | 24±n/a nM |
|---|
| Citation |  Yang, MG; Xiao, Z; Cherney, RJ; Tebben, AJ; Batt, DG; Brown, GD; Chen, J; Cvijic, ME; Dabros, M; Duncia, JV; Galella, M; Gardner, DS; Khandelwal, P; Ko, SS; Malley, MF; Mo, R; Pang, J; Rose, AV; Santella, JB; Shi, H; Srivastava, A; Traeger, SC; Wang, B; Xu, S; Zhao, R; Barrish, JC; Mandlekar, S; Zhao, Q; Carter, PH Use of a Conformational-Switching Mechanism to Modulate Exposed Polarity: Discovery of CCR2 Antagonist BMS-741672. ACS Med Chem Lett10:300-305 (2019) [PubMed] Article Yang, MG; Xiao, Z; Cherney, RJ; Tebben, AJ; Batt, DG; Brown, GD; Chen, J; Cvijic, ME; Dabros, M; Duncia, JV; Galella, M; Gardner, DS; Khandelwal, P; Ko, SS; Malley, MF; Mo, R; Pang, J; Rose, AV; Santella, JB; Shi, H; Srivastava, A; Traeger, SC; Wang, B; Xu, S; Zhao, R; Barrish, JC; Mandlekar, S; Zhao, Q; Carter, PH Use of a Conformational-Switching Mechanism to Modulate Exposed Polarity: Discovery of CCR2 Antagonist BMS-741672. ACS Med Chem Lett10:300-305 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 5 |
|---|
| Name: | C-C chemokine receptor type 5 |
|---|
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40540.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51681 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKR
LKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFII
LLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSS
HFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTI
MIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFV
GEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
|
|
|
|---|
| BDBM50509860 |
|---|
| n/a |
|---|
| Name | BDBM50509860 |
|---|
| Synonyms: | CHEMBL4442783 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H33F3N6O2 |
|---|
| Mol. Mass. | 506.5637 |
|---|
| SMILES | CC(C)N(C)[C@@H]1CC[C@@H]([C@@H](C1)NC(C)=O)N1CC[C@H](Nc2ncnc3ccc(cc23)C(F)(F)F)C1=O |r| |
|---|
| Structure | 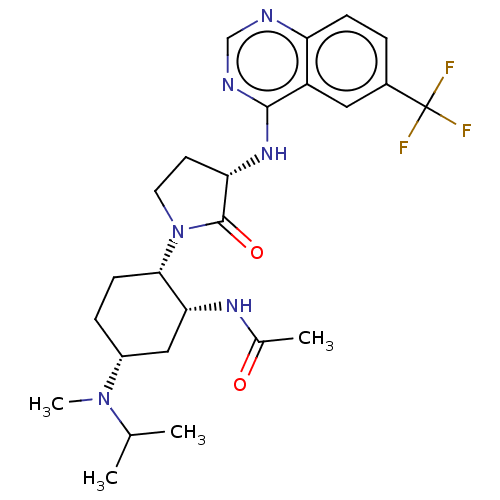
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yang, MG; Xiao, Z; Cherney, RJ; Tebben, AJ; Batt, DG; Brown, GD; Chen, J; Cvijic, ME; Dabros, M; Duncia, JV; Galella, M; Gardner, DS; Khandelwal, P; Ko, SS; Malley, MF; Mo, R; Pang, J; Rose, AV; Santella, JB; Shi, H; Srivastava, A; Traeger, SC; Wang, B; Xu, S; Zhao, R; Barrish, JC; Mandlekar, S; Zhao, Q; Carter, PH Use of a Conformational-Switching Mechanism to Modulate Exposed Polarity: Discovery of CCR2 Antagonist BMS-741672. ACS Med Chem Lett10:300-305 (2019) [PubMed] Article
Yang, MG; Xiao, Z; Cherney, RJ; Tebben, AJ; Batt, DG; Brown, GD; Chen, J; Cvijic, ME; Dabros, M; Duncia, JV; Galella, M; Gardner, DS; Khandelwal, P; Ko, SS; Malley, MF; Mo, R; Pang, J; Rose, AV; Santella, JB; Shi, H; Srivastava, A; Traeger, SC; Wang, B; Xu, S; Zhao, R; Barrish, JC; Mandlekar, S; Zhao, Q; Carter, PH Use of a Conformational-Switching Mechanism to Modulate Exposed Polarity: Discovery of CCR2 Antagonist BMS-741672. ACS Med Chem Lett10:300-305 (2019) [PubMed] Article