| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 6 |
|---|
| Ligand | BDBM50516525 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1865787 (CHEMBL4366762) |
|---|
| Ki | 87±n/a nM |
|---|
| Citation |  Ali, W; Wi?cek, M; ?a?ewska, D; Kurczab, R; Jastrz?bska-Wi?sek, M; Sata?a, G; Kucwaj-Brysz, K; Lubelska, A; G?uch-Lutwin, M; Mordyl, B; Siwek, A; Nasim, MJ; Partyka, A; Sudo?, S; Latacz, G; Weso?owska, A; Kie?-Kononowicz, K; Handzlik, J Synthesis and computer-aided SAR studies for derivatives of phenoxyalkyl-1,3,5-triazine as the new potent ligands for serotonin receptors 5-HT Eur J Med Chem178:740-751 (2019) [PubMed] Article Ali, W; Wi?cek, M; ?a?ewska, D; Kurczab, R; Jastrz?bska-Wi?sek, M; Sata?a, G; Kucwaj-Brysz, K; Lubelska, A; G?uch-Lutwin, M; Mordyl, B; Siwek, A; Nasim, MJ; Partyka, A; Sudo?, S; Latacz, G; Weso?owska, A; Kie?-Kononowicz, K; Handzlik, J Synthesis and computer-aided SAR studies for derivatives of phenoxyalkyl-1,3,5-triazine as the new potent ligands for serotonin receptors 5-HT Eur J Med Chem178:740-751 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 6 |
|---|
| Name: | 5-hydroxytryptamine receptor 6 |
|---|
| Synonyms: | 5-HT-6 | 5-HT6 | 5-hydroxytryptamine receptor 6 (5-HT-6) | 5-hydroxytryptamine receptor 6 (5-HT6R) | 5-hydroxytryptamine receptor 6 (5HT6) | 5HT6R_HUMAN | HTR6 | Serotonin (5-HT3) receptor | Serotonin 6 (5-HT6) receptor | Serotonin Receptor 6 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46968.43 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P50406 |
|---|
| Residue: | 440 |
|---|
| Sequence: | MVPEPGPTANSTPAWGAGPPSAPGGSGWVAAALCVVIALTAAANSLLIALICTQPALRNT
SNFFLVSLFTSDLMVGLVVMPPAMLNALYGRWVLARGLCLLWTAFDVMCCSASILNLCLI
SLDRYLLILSPLRYKLRMTPLRALALVLGAWSLAALASFLPLLLGWHELGHARPPVPGQC
RLLASLPFVLVASGLTFFLPSGAICFTYCRILLAARKQAVQVASLTTGMASQASETLQVP
RTPRPGVESADSRRLATKHSRKALKASLTLGILLGMFFVTWLPFFVANIVQAVCDCISPG
LFDVLTWLGYCNSTMNPIIYPLFMRDFKRALGRFLPCPRCPRERQASLASPSLRTSHSGP
RPGLSLQQVLPLPLPPDSDSDSDAGSGGSSGLRLTAQLLLPGEATQDPPLPTRAAAAVNF
FNIDPAEPELRPHPLGIPTN
|
|
|
|---|
| BDBM50516525 |
|---|
| n/a |
|---|
| Name | BDBM50516525 |
|---|
| Synonyms: | CHEMBL4570584 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H22N6O |
|---|
| Mol. Mass. | 314.3855 |
|---|
| SMILES | CN1CCN(CC1)c1nc(N)nc(COc2ccccc2C)n1 |
|---|
| Structure | 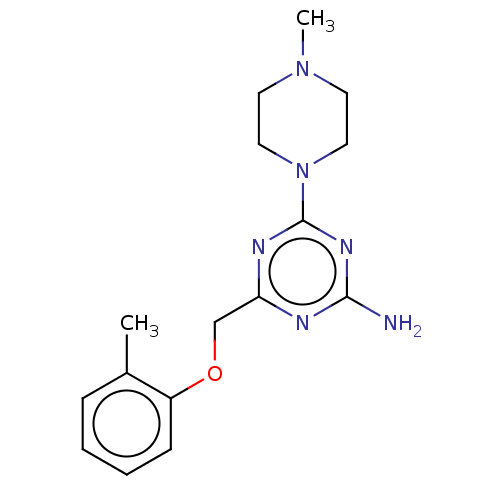
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ali, W; Wi?cek, M; ?a?ewska, D; Kurczab, R; Jastrz?bska-Wi?sek, M; Sata?a, G; Kucwaj-Brysz, K; Lubelska, A; G?uch-Lutwin, M; Mordyl, B; Siwek, A; Nasim, MJ; Partyka, A; Sudo?, S; Latacz, G; Weso?owska, A; Kie?-Kononowicz, K; Handzlik, J Synthesis and computer-aided SAR studies for derivatives of phenoxyalkyl-1,3,5-triazine as the new potent ligands for serotonin receptors 5-HT Eur J Med Chem178:740-751 (2019) [PubMed] Article
Ali, W; Wi?cek, M; ?a?ewska, D; Kurczab, R; Jastrz?bska-Wi?sek, M; Sata?a, G; Kucwaj-Brysz, K; Lubelska, A; G?uch-Lutwin, M; Mordyl, B; Siwek, A; Nasim, MJ; Partyka, A; Sudo?, S; Latacz, G; Weso?owska, A; Kie?-Kononowicz, K; Handzlik, J Synthesis and computer-aided SAR studies for derivatives of phenoxyalkyl-1,3,5-triazine as the new potent ligands for serotonin receptors 5-HT Eur J Med Chem178:740-751 (2019) [PubMed] Article