| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C8 |
|---|
| Ligand | BDBM50553584 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2046890 (CHEMBL4701589) |
|---|
| IC50 | 1900±n/a nM |
|---|
| Citation |  Shi, Q; Xiao, Z; Yang, MG; Marcoux, D; Cherney, RJ; Yip, S; Li, P; Wu, DR; Weigelt, CA; Sack, J; Khan, J; Ruzanov, M; Wang, J; Yarde, M; Ellen Cvijic, M; Li, S; Shuster, DJ; Xie, J; Sherry, T; Obermeier, M; Fura, A; Stefanski, K; Cornelius, G; Chacko, S; Shu, YZ; Khandelwal, P; Hynes, J; Tino, JA; Salter-Cid, L; Denton, R; Zhao, Q; Dhar, TGM Tricyclic sulfones as potent, selective and efficacious ROR?t inverse agonists - Exploring C6 and C8 SAR using late-stage functionalization. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article Shi, Q; Xiao, Z; Yang, MG; Marcoux, D; Cherney, RJ; Yip, S; Li, P; Wu, DR; Weigelt, CA; Sack, J; Khan, J; Ruzanov, M; Wang, J; Yarde, M; Ellen Cvijic, M; Li, S; Shuster, DJ; Xie, J; Sherry, T; Obermeier, M; Fura, A; Stefanski, K; Cornelius, G; Chacko, S; Shu, YZ; Khandelwal, P; Hynes, J; Tino, JA; Salter-Cid, L; Denton, R; Zhao, Q; Dhar, TGM Tricyclic sulfones as potent, selective and efficacious ROR?t inverse agonists - Exploring C6 and C8 SAR using late-stage functionalization. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C8 |
|---|
| Name: | Cytochrome P450 2C8 |
|---|
| Synonyms: | CP2C8_HUMAN | CYP2C8 | CYPIIC8 | Cytochrome P450 2C8 (CYP2C8) | P450 IIC2 | P450 MP-12/MP-20 | P450 form 1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55839.23 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10632 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MEPFVVLVLCLSFMLLFSLWRQSCRRRKLPPGPTPLPIIGNMLQIDVKDICKSFTNFSKV
YGPVFTVYFGMNPIVVFHGYEAVKEALIDNGEEFSGRGNSPISQRITKGLGIISSNGKRW
KEIRRFSLTTLRNFGMGKRSIEDRVQEEAHCLVEELRKTKASPCDPTFILGCAPCNVICS
VVFQKRFDYKDQNFLTLMKRFNENFRILNSPWIQVCNNFPLLIDCFPGTHNKVLKNVALT
RSYIREKVKEHQASLDVNNPRDFIDCFLIKMEQEKDNQKSEFNIENLVGTVADLFVAGTE
TTSTTLRYGLLLLLKHPEVTAKVQEEIDHVIGRHRSPCMQDRSHMPYTDAVVHEIQRYSD
LVPTGVPHAVTTDTKFRNYLIPKGTTIMALLTSVLHDDKEFPNPNIFDPGHFLDKNGNFK
KSDYFMPFSAGKRICAGEGLARMELFLFLTTILQNFNLKSVDDLKNLNTTAVTKGIVSLP
PSYQICFIPV
|
|
|
|---|
| BDBM50553584 |
|---|
| n/a |
|---|
| Name | BDBM50553584 |
|---|
| Synonyms: | CHEMBL4763134 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H29F8NO5S |
|---|
| Mol. Mass. | 667.607 |
|---|
| SMILES | [H][C@@]12CCc3cc(c(F)cc3[C@@]1(CCN2C(=O)[C@@H]1CC[C@H](C[C@@H]1C)C(O)=O)S(=O)(=O)c1ccccc1)C(F)(C(F)(F)F)C(F)(F)F |r| |
|---|
| Structure | 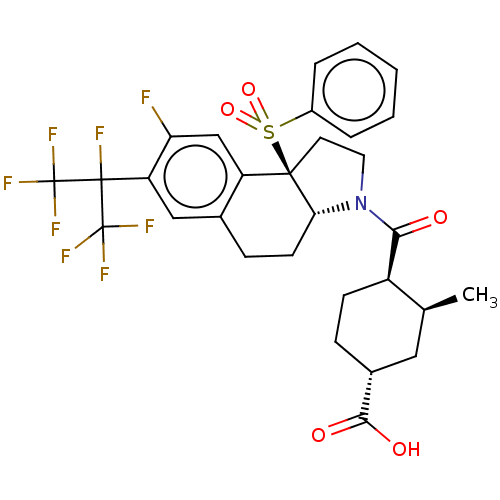
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shi, Q; Xiao, Z; Yang, MG; Marcoux, D; Cherney, RJ; Yip, S; Li, P; Wu, DR; Weigelt, CA; Sack, J; Khan, J; Ruzanov, M; Wang, J; Yarde, M; Ellen Cvijic, M; Li, S; Shuster, DJ; Xie, J; Sherry, T; Obermeier, M; Fura, A; Stefanski, K; Cornelius, G; Chacko, S; Shu, YZ; Khandelwal, P; Hynes, J; Tino, JA; Salter-Cid, L; Denton, R; Zhao, Q; Dhar, TGM Tricyclic sulfones as potent, selective and efficacious ROR?t inverse agonists - Exploring C6 and C8 SAR using late-stage functionalization. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article
Shi, Q; Xiao, Z; Yang, MG; Marcoux, D; Cherney, RJ; Yip, S; Li, P; Wu, DR; Weigelt, CA; Sack, J; Khan, J; Ruzanov, M; Wang, J; Yarde, M; Ellen Cvijic, M; Li, S; Shuster, DJ; Xie, J; Sherry, T; Obermeier, M; Fura, A; Stefanski, K; Cornelius, G; Chacko, S; Shu, YZ; Khandelwal, P; Hynes, J; Tino, JA; Salter-Cid, L; Denton, R; Zhao, Q; Dhar, TGM Tricyclic sulfones as potent, selective and efficacious ROR?t inverse agonists - Exploring C6 and C8 SAR using late-stage functionalization. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article