| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Amine oxidase [flavin-containing] B |
|---|
| Ligand | BDBM50122955 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_124426 (CHEMBL874045) |
|---|
| IC50 | 200±n/a nM |
|---|
| Citation |  Musso, DL; Cochran, FR; Kelley, JL; McLean, EW; Selph, JL; Rigdon, GC; Orr, GF; Davis, RG; Cooper, BR; Styles, VL; Thompson, JB; Hall, WR Indanylidenes. 1. Design and synthesis of (E)-2-(4,6-difluoro-1-indanylidene)acetamide, a potent, centrally acting muscle relaxant with antiinflammatory and analgesic activity. J Med Chem46:399-408 (2003) [PubMed] Article Musso, DL; Cochran, FR; Kelley, JL; McLean, EW; Selph, JL; Rigdon, GC; Orr, GF; Davis, RG; Cooper, BR; Styles, VL; Thompson, JB; Hall, WR Indanylidenes. 1. Design and synthesis of (E)-2-(4,6-difluoro-1-indanylidene)acetamide, a potent, centrally acting muscle relaxant with antiinflammatory and analgesic activity. J Med Chem46:399-408 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Amine oxidase [flavin-containing] B |
|---|
| Name: | Amine oxidase [flavin-containing] B |
|---|
| Synonyms: | AOFB_HUMAN | MAO-B | MAOB | Monoamine oxidase type B | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAOB) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 58768.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P27338 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MSNKCDVVVVGGGISGMAAAKLLHDSGLNVVVLEARDRVGGRTYTLRNQKVKYVDLGGSY
VGPTQNRILRLAKELGLETYKVNEVERLIHHVKGKSYPFRGPFPPVWNPITYLDHNNFWR
TMDDMGREIPSDAPWKAPLAEEWDNMTMKELLDKLCWTESAKQLATLFVNLCVTAETHEV
SALWFLWYVKQCGGTTRIISTTNGGQERKFVGGSGQVSERIMDLLGDRVKLERPVIYIDQ
TRENVLVETLNHEMYEAKYVISAIPPTLGMKIHFNPPLPMMRNQMITRVPLGSVIKCIVY
YKEPFWRKKDYCGTMIIDGEEAPVAYTLDDTKPEGNYAAIMGFILAHKARKLARLTKEER
LKKLCELYAKVLGSLEALEPVHYEEKNWCEEQYSGGCYTTYFPPGILTQYGRVLRQPVDR
IYFAGTETATHWSGYMEGAVEAGERAAREILHAMGKIPEDEIWQSEPESVDVPAQPITTT
FLERHLPSVPGLLRLIGLTTIFSATALGFLAHKRGLLVRV
|
|
|
|---|
| BDBM50122955 |
|---|
| n/a |
|---|
| Name | BDBM50122955 |
|---|
| Synonyms: | 2-(4,6-Difluoro-indan-1-ylidene)-acetamide | CHEMBL130217 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H9F2NO |
|---|
| Mol. Mass. | 209.1921 |
|---|
| SMILES | NC(=O)\C=C1/CCc2c1cc(F)cc2F |
|---|
| Structure | 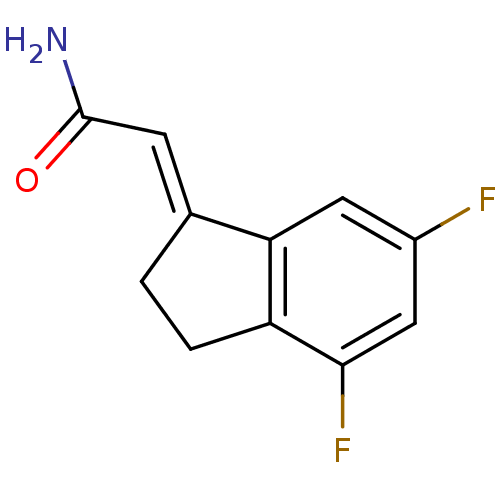
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Musso, DL; Cochran, FR; Kelley, JL; McLean, EW; Selph, JL; Rigdon, GC; Orr, GF; Davis, RG; Cooper, BR; Styles, VL; Thompson, JB; Hall, WR Indanylidenes. 1. Design and synthesis of (E)-2-(4,6-difluoro-1-indanylidene)acetamide, a potent, centrally acting muscle relaxant with antiinflammatory and analgesic activity. J Med Chem46:399-408 (2003) [PubMed] Article
Musso, DL; Cochran, FR; Kelley, JL; McLean, EW; Selph, JL; Rigdon, GC; Orr, GF; Davis, RG; Cooper, BR; Styles, VL; Thompson, JB; Hall, WR Indanylidenes. 1. Design and synthesis of (E)-2-(4,6-difluoro-1-indanylidene)acetamide, a potent, centrally acting muscle relaxant with antiinflammatory and analgesic activity. J Med Chem46:399-408 (2003) [PubMed] Article