| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sphingosine 1-phosphate receptor 2 |
|---|
| Ligand | BDBM50158337 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_430294 (CHEMBL913920) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Hale, JJ; Lynch, CL; Neway, W; Mills, SG; Hajdu, R; Keohane, CA; Rosenbach, MJ; Milligan, JA; Shei, GJ; Parent, SA; Chrebet, G; Bergstrom, J; Card, D; Ferrer, M; Hodder, P; Strulovici, B; Rosen, H; Mandala, S A rational utilization of high-throughput screening affords selective, orally bioavailable 1-benzyl-3-carboxyazetidine sphingosine-1-phosphate-1 receptor agonists. J Med Chem47:6662-5 (2004) [PubMed] Article Hale, JJ; Lynch, CL; Neway, W; Mills, SG; Hajdu, R; Keohane, CA; Rosenbach, MJ; Milligan, JA; Shei, GJ; Parent, SA; Chrebet, G; Bergstrom, J; Card, D; Ferrer, M; Hodder, P; Strulovici, B; Rosen, H; Mandala, S A rational utilization of high-throughput screening affords selective, orally bioavailable 1-benzyl-3-carboxyazetidine sphingosine-1-phosphate-1 receptor agonists. J Med Chem47:6662-5 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sphingosine 1-phosphate receptor 2 |
|---|
| Name: | Sphingosine 1-phosphate receptor 2 |
|---|
| Synonyms: | EDG5 | S1P2 | S1PR2 | S1PR2_HUMAN | Sphingosine 1-phosphate receptor | Sphingosine 1-phosphate receptor Edg-5 | Sphingosine-1-phosphate receptor 2 | ndothelial differentiation G-protein coupled receptor 5 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 38883.16 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Membranes isolated from S1P2-transfected CHO cells were used in ligand binding assay. |
|---|
| Residue: | 353 |
|---|
| Sequence: | MGSLYSEYLNPNKVQEHYNYTKETLETQETTSRQVASAFIVILCCAIVVENLLVLIAVAR
NSKFHSAMYLFLGNLAASDLLAGVAFVANTLLSGSVTLRLTPVQWFAREGSAFITLSASV
FSLLAIAIERHVAIAKVKLYGSDKSCRMLLLIGASWLISLVLGGLPILGWNCLGHLEACS
TVLPLYAKHYVLCVVTIFSIILLAIVALYVRIYCVVRSSHADMAAPQTLALLKTVTIVLG
VFIVCWLPAFSILLLDYACPVHSCPILYKAHYFFAVSTLNSLLNPVIYTWRSRDLRREVL
RPLQCWRPGVGVQGRRRGGTPGHHLLPLRSSSSLERGMHMPTSPTFLEGNTVV
|
|
|
|---|
| BDBM50158337 |
|---|
| n/a |
|---|
| Name | BDBM50158337 |
|---|
| Synonyms: | 3-(2,4-dichlorophenyl)-5-(4-phenyl-5-(trifluoromethyl)thiophen-2-yl)-1,2,4-oxadiazole | CHEMBL374614 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H9Cl2F3N2OS |
|---|
| Mol. Mass. | 441.254 |
|---|
| SMILES | FC(F)(F)c1sc(cc1-c1ccccc1)-c1nc(no1)-c1ccc(Cl)cc1Cl |
|---|
| Structure | 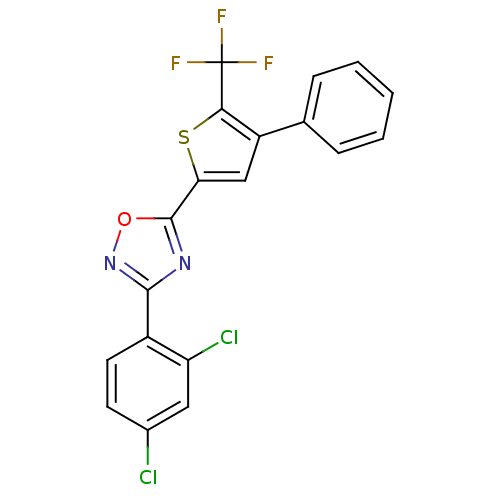
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hale, JJ; Lynch, CL; Neway, W; Mills, SG; Hajdu, R; Keohane, CA; Rosenbach, MJ; Milligan, JA; Shei, GJ; Parent, SA; Chrebet, G; Bergstrom, J; Card, D; Ferrer, M; Hodder, P; Strulovici, B; Rosen, H; Mandala, S A rational utilization of high-throughput screening affords selective, orally bioavailable 1-benzyl-3-carboxyazetidine sphingosine-1-phosphate-1 receptor agonists. J Med Chem47:6662-5 (2004) [PubMed] Article
Hale, JJ; Lynch, CL; Neway, W; Mills, SG; Hajdu, R; Keohane, CA; Rosenbach, MJ; Milligan, JA; Shei, GJ; Parent, SA; Chrebet, G; Bergstrom, J; Card, D; Ferrer, M; Hodder, P; Strulovici, B; Rosen, H; Mandala, S A rational utilization of high-throughput screening affords selective, orally bioavailable 1-benzyl-3-carboxyazetidine sphingosine-1-phosphate-1 receptor agonists. J Med Chem47:6662-5 (2004) [PubMed] Article