

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Glutamate receptor ionotropic, NMDA 2B | ||
| Ligand | BDBM50220611 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_457343 (CHEMBL940934) | ||
| Ki | 5.6±n/a nM | ||
| Citation |  Kawai, M; Sakurada, I; Morita, A; Iwamuro, Y; Ando, K; Omura, H; Sakakibara, S; Masuda, T; Koike, H; Honma, T; Hattori, K; Takashima, T; Mizuno, K; Mizutani, M; Kawamura, M Structure-activity relationship study of novel NR2B-selective antagonists with arylamides to avoid reactive metabolites formation. Bioorg Med Chem Lett17:5537-42 (2007) [PubMed] Article Kawai, M; Sakurada, I; Morita, A; Iwamuro, Y; Ando, K; Omura, H; Sakakibara, S; Masuda, T; Koike, H; Honma, T; Hattori, K; Takashima, T; Mizuno, K; Mizutani, M; Kawamura, M Structure-activity relationship study of novel NR2B-selective antagonists with arylamides to avoid reactive metabolites formation. Bioorg Med Chem Lett17:5537-42 (2007) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Glutamate receptor ionotropic, NMDA 2B | |||
| Name: | Glutamate receptor ionotropic, NMDA 2B | ||
| Synonyms: | GluN2B | Glutamate [NMDA] receptor subunit epsilon 2 | Grin2b | N-methyl D-aspartate receptor subtype 2B | NMDA receptor subunit N2B (GluN2B) | NMDAR2B | NMDE2_RAT | NR2B | ||
| Type: | Protein | ||
| Mol. Mass.: | 166077.66 | ||
| Organism: | Rattus norvegicus (Rat) | ||
| Description: | Q00960 | ||
| Residue: | 1482 | ||
| Sequence: |
| ||
| BDBM50220611 | |||
| n/a | |||
| Name | BDBM50220611 | ||
| Synonyms: | 6-hydroxy-N-(((1s,4s)-4-(2-phenoxyethyl)cyclohexyl)methyl)pyridazine-3-carboxamide | CHEMBL250114 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C20H25N3O3 | ||
| Mol. Mass. | 355.4308 | ||
| SMILES | O=C(NC[C@@H]1CC[C@H](CCOc2ccccc2)CC1)c1ccc(=O)[nH]n1 |wU:4.3,7.7,(20.2,-34.76,;20.2,-36.3,;21.54,-37.06,;22.87,-36.29,;24.2,-37.05,;24.2,-38.59,;25.53,-39.36,;26.86,-38.59,;28.2,-39.37,;29.53,-38.6,;30.86,-39.37,;32.2,-38.6,;33.53,-39.38,;34.86,-38.61,;34.87,-37.07,;33.53,-36.3,;32.2,-37.07,;26.86,-37.05,;25.53,-36.28,;18.87,-37.07,;17.54,-36.3,;16.2,-37.07,;16.21,-38.61,;14.88,-39.39,;17.55,-39.38,;18.88,-38.6,)| | ||
| Structure | 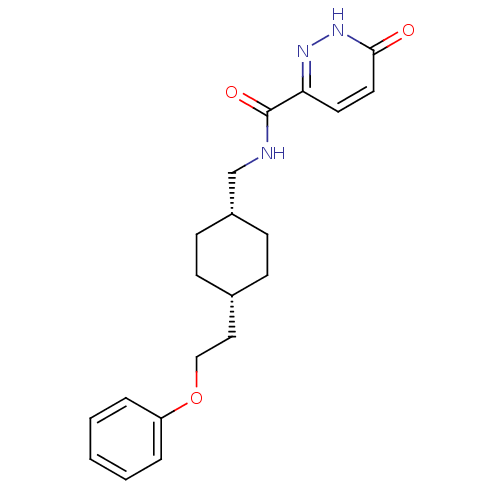
| ||