| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 11 |
|---|
| Ligand | BDBM50236473 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_463058 (CHEMBL928968) |
|---|
| IC50 | 1.5±n/a nM |
|---|
| Citation |  Hynes, J; Wu, H; Pitt, S; Shen, DR; Zhang, R; Schieven, GL; Gillooly, KM; Shuster, DJ; Taylor, TL; Yang, X; McIntyre, KW; McKinnon, M; Zhang, H; Marathe, PH; Doweyko, AM; Kish, K; Kiefer, SE; Sack, JS; Newitt, JA; Barrish, JC; Dodd, J; Leftheris, K The discovery of (R)-2-(sec-butylamino)-N-(2-methyl-5-(methylcarbamoyl)phenyl) thiazole-5-carboxamide (BMS-640994)-A potent and efficacious p38alpha MAP kinase inhibitor. Bioorg Med Chem Lett18:1762-7 (2008) [PubMed] Article Hynes, J; Wu, H; Pitt, S; Shen, DR; Zhang, R; Schieven, GL; Gillooly, KM; Shuster, DJ; Taylor, TL; Yang, X; McIntyre, KW; McKinnon, M; Zhang, H; Marathe, PH; Doweyko, AM; Kish, K; Kiefer, SE; Sack, JS; Newitt, JA; Barrish, JC; Dodd, J; Leftheris, K The discovery of (R)-2-(sec-butylamino)-N-(2-methyl-5-(methylcarbamoyl)phenyl) thiazole-5-carboxamide (BMS-640994)-A potent and efficacious p38alpha MAP kinase inhibitor. Bioorg Med Chem Lett18:1762-7 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 11 |
|---|
| Name: | Mitogen-activated protein kinase 11 |
|---|
| Synonyms: | MAP kinase p38 | MAPK11 | MK11_HUMAN | Mitogen-activated protein kinase 11 | Mitogen-activated protein kinase p38 beta | PRKM11 | SAPK2 | SAPK2B | Stress-activated protein kinase 2 | p38 MAP kinase alpha/beta | p38-2 | p38-beta | p38b |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 41351.73 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_20128774 |
|---|
| Residue: | 364 |
|---|
| Sequence: | MSGPRAGFYRQELNKTVWEVPQRLQGLRPVGSGAYGSVCSAYDARLRQKVAVKKLSRPFQ
SLIHARRTYRELRLLKHLKHENVIGLLDVFTPATSIEDFSEVYLVTTLMGADLNNIVKCQ
ALSDEHVQFLVYQLLRGLKYIHSAGIIHRDLKPSNVAVNEDCELRILDFGLARQADEEMT
GYVATRWYRAPEIMLNWMHYNQTVDIWSVGCIMAELLQGKALFPGSDYIDQLKRIMEVVG
TPSPEVLAKISSEHARTYIQSLPPMPQKDLSSIFRGANPLAIDLLGRMLVLDSDQRVSAA
EALAHAYFSQYHDPEDEPEAEPYDESVEAKERTLEEWKELTYQEVLSFKPPEPPKPPGSL
EIEQ
|
|
|
|---|
| BDBM50236473 |
|---|
| n/a |
|---|
| Name | BDBM50236473 |
|---|
| Synonyms: | (R)-2-(sec-butylamino)-N-(2-methyl-5-(methylcarbamoyl)phenyl)thiazole-5-carboxamide | BMS-640994 | CHEMBL258202 | N-[2-methyl-5-(methylcarbamoyl)phenyl]-2-{[(1R)-1-methylpropyl]amino}-1,3-thiazole-5-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H22N4O2S |
|---|
| Mol. Mass. | 346.447 |
|---|
| SMILES | CC[C@@H](C)Nc1ncc(s1)C(=O)Nc1cc(ccc1C)C(=O)NC |
|---|
| Structure | 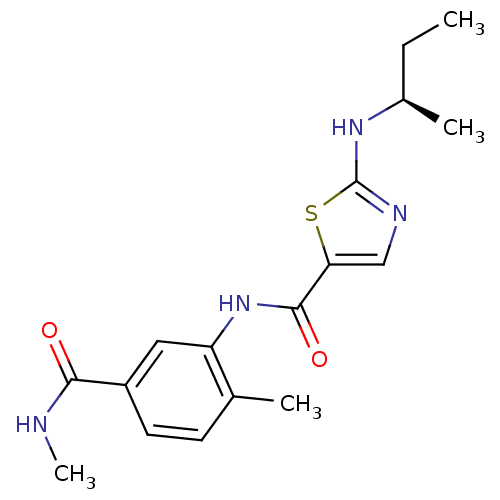
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hynes, J; Wu, H; Pitt, S; Shen, DR; Zhang, R; Schieven, GL; Gillooly, KM; Shuster, DJ; Taylor, TL; Yang, X; McIntyre, KW; McKinnon, M; Zhang, H; Marathe, PH; Doweyko, AM; Kish, K; Kiefer, SE; Sack, JS; Newitt, JA; Barrish, JC; Dodd, J; Leftheris, K The discovery of (R)-2-(sec-butylamino)-N-(2-methyl-5-(methylcarbamoyl)phenyl) thiazole-5-carboxamide (BMS-640994)-A potent and efficacious p38alpha MAP kinase inhibitor. Bioorg Med Chem Lett18:1762-7 (2008) [PubMed] Article
Hynes, J; Wu, H; Pitt, S; Shen, DR; Zhang, R; Schieven, GL; Gillooly, KM; Shuster, DJ; Taylor, TL; Yang, X; McIntyre, KW; McKinnon, M; Zhang, H; Marathe, PH; Doweyko, AM; Kish, K; Kiefer, SE; Sack, JS; Newitt, JA; Barrish, JC; Dodd, J; Leftheris, K The discovery of (R)-2-(sec-butylamino)-N-(2-methyl-5-(methylcarbamoyl)phenyl) thiazole-5-carboxamide (BMS-640994)-A potent and efficacious p38alpha MAP kinase inhibitor. Bioorg Med Chem Lett18:1762-7 (2008) [PubMed] Article