| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50293851 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_572402 (CHEMBL1036138) |
|---|
| IC50 | 40000±n/a nM |
|---|
| Citation |  Liu, W; Liu, K; Wood, HB; McCann, ME; Doebber, TW; Chang, CH; Akiyama, TE; Einstein, M; Berger, JP; Meinke, PT Discovery of a peroxisome proliferator activated receptor gamma (PPARgamma) modulator with balanced PPARalpha activity for the treatment of type 2 diabetes and dyslipidemia. J Med Chem52:4443-53 (2009) [PubMed] Article Liu, W; Liu, K; Wood, HB; McCann, ME; Doebber, TW; Chang, CH; Akiyama, TE; Einstein, M; Berger, JP; Meinke, PT Discovery of a peroxisome proliferator activated receptor gamma (PPARgamma) modulator with balanced PPARalpha activity for the treatment of type 2 diabetes and dyslipidemia. J Med Chem52:4443-53 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50293851 |
|---|
| n/a |
|---|
| Name | BDBM50293851 |
|---|
| Synonyms: | (2R)-2-{3-[[3-(4-Chlorobenzoyl)-2-methyl-6-(trifluoromethoxy)-1H-indol-1-yl]methyl]phenoxy}butanoic Acid | CHEMBL552240 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H23ClF3NO5 |
|---|
| Mol. Mass. | 545.934 |
|---|
| SMILES | CC[C@@H](Oc1cccc(Cn2c(C)c(C(=O)c3ccc(Cl)cc3)c3ccc(OC(F)(F)F)cc23)c1)C(O)=O |r| |
|---|
| Structure | 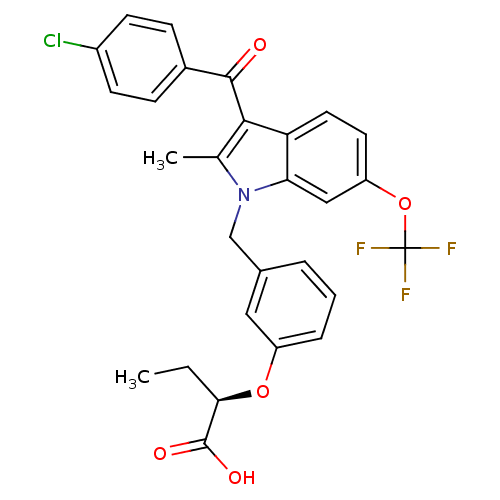
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, W; Liu, K; Wood, HB; McCann, ME; Doebber, TW; Chang, CH; Akiyama, TE; Einstein, M; Berger, JP; Meinke, PT Discovery of a peroxisome proliferator activated receptor gamma (PPARgamma) modulator with balanced PPARalpha activity for the treatment of type 2 diabetes and dyslipidemia. J Med Chem52:4443-53 (2009) [PubMed] Article
Liu, W; Liu, K; Wood, HB; McCann, ME; Doebber, TW; Chang, CH; Akiyama, TE; Einstein, M; Berger, JP; Meinke, PT Discovery of a peroxisome proliferator activated receptor gamma (PPARgamma) modulator with balanced PPARalpha activity for the treatment of type 2 diabetes and dyslipidemia. J Med Chem52:4443-53 (2009) [PubMed] Article