| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50341023 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_740159 (CHEMBL1763219) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Riether, D; Wu, L; Cirillo, PF; Berry, A; Walker, ER; Ermann, M; Noya-Marino, B; Jenkins, JE; Albaugh, D; Albrecht, C; Fisher, M; Gemkow, MJ; Grbic, H; Löbbe, S; Möller, C; O'Shea, K; Sauer, A; Shih, DT; Thomson, DS 1,4-Diazepane compounds as potent and selective CB2 agonists: optimization of metabolic stability. Bioorg Med Chem Lett21:2011-6 (2011) [PubMed] Article Riether, D; Wu, L; Cirillo, PF; Berry, A; Walker, ER; Ermann, M; Noya-Marino, B; Jenkins, JE; Albaugh, D; Albrecht, C; Fisher, M; Gemkow, MJ; Grbic, H; Löbbe, S; Möller, C; O'Shea, K; Sauer, A; Shih, DT; Thomson, DS 1,4-Diazepane compounds as potent and selective CB2 agonists: optimization of metabolic stability. Bioorg Med Chem Lett21:2011-6 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50341023 |
|---|
| n/a |
|---|
| Name | BDBM50341023 |
|---|
| Synonyms: | CHEMBL1762293 | N-(5-tert-butylisoxazol-3-yl)-4-(tetrahydro-2H-pyran-4-carbonyl)-1,4-diazepane-1-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H30N4O4 |
|---|
| Mol. Mass. | 378.4659 |
|---|
| SMILES | CC(C)(C)c1cc(NC(=O)N2CCCN(CC2)C(=O)C2CCOCC2)no1 |
|---|
| Structure | 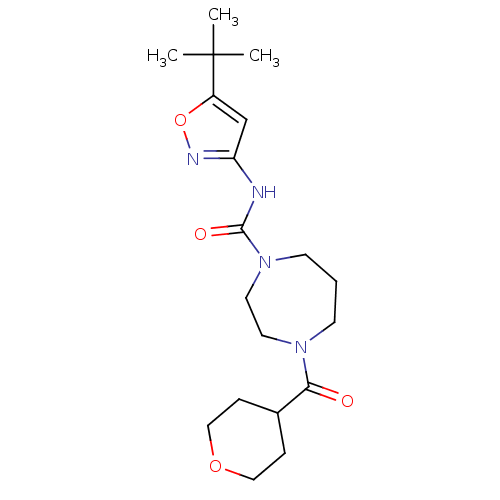
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Riether, D; Wu, L; Cirillo, PF; Berry, A; Walker, ER; Ermann, M; Noya-Marino, B; Jenkins, JE; Albaugh, D; Albrecht, C; Fisher, M; Gemkow, MJ; Grbic, H; Löbbe, S; Möller, C; O'Shea, K; Sauer, A; Shih, DT; Thomson, DS 1,4-Diazepane compounds as potent and selective CB2 agonists: optimization of metabolic stability. Bioorg Med Chem Lett21:2011-6 (2011) [PubMed] Article
Riether, D; Wu, L; Cirillo, PF; Berry, A; Walker, ER; Ermann, M; Noya-Marino, B; Jenkins, JE; Albaugh, D; Albrecht, C; Fisher, M; Gemkow, MJ; Grbic, H; Löbbe, S; Möller, C; O'Shea, K; Sauer, A; Shih, DT; Thomson, DS 1,4-Diazepane compounds as potent and selective CB2 agonists: optimization of metabolic stability. Bioorg Med Chem Lett21:2011-6 (2011) [PubMed] Article