| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sphingosine 1-phosphate receptor 5 |
|---|
| Ligand | BDBM50063864 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1463807 (CHEMBL3404970) |
|---|
| EC50 | 5.8±n/a nM |
|---|
| Citation |  Buzard, DJ; Schrader, TO; Zhu, X; Lehmann, J; Johnson, B; Kasem, M; Kim, SH; Kawasaki, A; Lopez, L; Moody, J; Han, S; Gao, Y; Edwards, J; Barden, J; Thatte, J; Gatlin, J; Jones, RM Design and synthesis of new tricyclic indoles as potent modulators of the S1P1 receptor. Bioorg Med Chem Lett25:659-63 (2015) [PubMed] Article Buzard, DJ; Schrader, TO; Zhu, X; Lehmann, J; Johnson, B; Kasem, M; Kim, SH; Kawasaki, A; Lopez, L; Moody, J; Han, S; Gao, Y; Edwards, J; Barden, J; Thatte, J; Gatlin, J; Jones, RM Design and synthesis of new tricyclic indoles as potent modulators of the S1P1 receptor. Bioorg Med Chem Lett25:659-63 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sphingosine 1-phosphate receptor 5 |
|---|
| Name: | Sphingosine 1-phosphate receptor 5 |
|---|
| Synonyms: | EDG8 | Endothelial differentiation sphingolipid G-protein-coupled receptor 8 | S1P5 | S1PR5 | S1PR5_HUMAN | Sphingosine 1-phosphate receptor | Sphingosine 1-phosphate receptor Edg-8 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 41796.42 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 398 |
|---|
| Sequence: | MESGLLRPAPVSEVIVLHYNYTGKLRGARYQPGAGLRADAVVCLAVCAFIVLENLAVLLV
LGRHPRFHAPMFLLLGSLTLSDLLAGAAYAANILLSGPLTLKLSPALWFAREGGVFVALT
ASVLSLLAIALERSLTMARRGPAPVSSRGRTLAMAAAAWGVSLLLGLLPALGWNCLGRLD
ACSTVLPLYAKAYVLFCVLAFVGILAAICALYARIYCQVRANARRLPARPGTAGTTSTRA
RRKPRSLALLRTLSVVLLAFVACWGPLFLLLLLDVACPARTCPVLLQADPFLGLAMANSL
LNPIIYTLTNRDLRHALLRLVCCGRHSCGRDPSGSQQSASAAEASGGLRRCLPPGLDGSF
SGSERSSPQRDGLDTSGSTGSPGAPTAARTLVSEPAAD
|
|
|
|---|
| BDBM50063864 |
|---|
| n/a |
|---|
| Name | BDBM50063864 |
|---|
| Synonyms: | CHEMBL3403619 | US10676467, Compound TZ 50 13 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H27F3N2O3 |
|---|
| Mol. Mass. | 472.4994 |
|---|
| SMILES | OC(=O)CC1NCCc2c1[nH]c1ccc(OCc3ccc(C4CCCC4)c(c3)C(F)(F)F)cc21 |
|---|
| Structure | 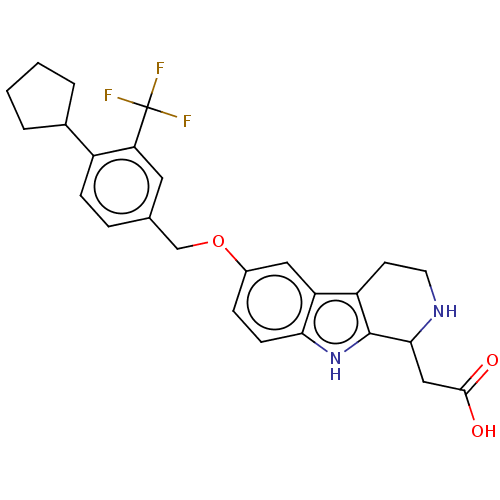
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Buzard, DJ; Schrader, TO; Zhu, X; Lehmann, J; Johnson, B; Kasem, M; Kim, SH; Kawasaki, A; Lopez, L; Moody, J; Han, S; Gao, Y; Edwards, J; Barden, J; Thatte, J; Gatlin, J; Jones, RM Design and synthesis of new tricyclic indoles as potent modulators of the S1P1 receptor. Bioorg Med Chem Lett25:659-63 (2015) [PubMed] Article
Buzard, DJ; Schrader, TO; Zhu, X; Lehmann, J; Johnson, B; Kasem, M; Kim, SH; Kawasaki, A; Lopez, L; Moody, J; Han, S; Gao, Y; Edwards, J; Barden, J; Thatte, J; Gatlin, J; Jones, RM Design and synthesis of new tricyclic indoles as potent modulators of the S1P1 receptor. Bioorg Med Chem Lett25:659-63 (2015) [PubMed] Article