| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2B |
|---|
| Ligand | BDBM50130841 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1525542 (CHEMBL3637049) |
|---|
| IC50 | 6.7±n/a nM |
|---|
| Citation |  Lingam, VS; Dahale, DH; Rathi, VE; Shingote, YB; Thakur, RR; Mindhe, AS; Kummari, S; Khairatkar-Joshi, N; Bajpai, M; Shah, DM; Sapalya, RS; Gullapalli, S; Gupta, PK; Gudi, GS; Jadhav, SB; Pattem, R; Thomas, A Design, Synthesis, and Pharmacological Evaluation of 5,6-Disubstituted Pyridin-2(1H)-one Derivatives as Phosphodiesterase 10A (PDE10A) Antagonists. J Med Chem58:8292-308 (2015) [PubMed] Article Lingam, VS; Dahale, DH; Rathi, VE; Shingote, YB; Thakur, RR; Mindhe, AS; Kummari, S; Khairatkar-Joshi, N; Bajpai, M; Shah, DM; Sapalya, RS; Gullapalli, S; Gupta, PK; Gudi, GS; Jadhav, SB; Pattem, R; Thomas, A Design, Synthesis, and Pharmacological Evaluation of 5,6-Disubstituted Pyridin-2(1H)-one Derivatives as Phosphodiesterase 10A (PDE10A) Antagonists. J Med Chem58:8292-308 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2B |
|---|
| Name: | 5-hydroxytryptamine receptor 2B |
|---|
| Synonyms: | 5-HT-2B | 5-HT2B | 5-hydroxytryptamine (serotonin) receptor 2B [Homo sapiens] | 5-hydroxytryptamine receptor 2B (5-HT2B) | 5-hydroxytryptamine receptor 2C (5HT2C) | 5HT2B_HUMAN | HTR2B | Serotonin (5-HT3) receptor | Serotonin 2b (5-HT2b) receptor | Serotonin Receptor 2B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 54312.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells. |
|---|
| Residue: | 481 |
|---|
| Sequence: | MALSYRVSELQSTIPEHILQSTFVHVISSNWSGLQTESIPEEMKQIVEEQGNKLHWAALL
ILMVIIPTIGGNTLVILAVSLEKKLQYATNYFLMSLAVADLLVGLFVMPIALLTIMFEAM
WPLPLVLCPAWLFLDVLFSTASIMHLCAISVDRYIAIKKPIQANQYNSRATAFIKITVVW
LISIGIAIPVPIKGIETDVDNPNNITCVLTKERFGDFMLFGSLAAFFTPLAIMIVTYFLT
IHALQKKAYLVKNKPPQRLTWLTVSTVFQRDETPCSSPEKVAMLDGSRKDKALPNSGDET
LMRRTSTIGKKSVQTISNEQRASKVLGIVFFLFLLMWCPFFITNITLVLCDSCNQTTLQM
LLEIFVWIGYVSSGVNPLVYTLFNKTFRDAFGRYITCNYRATKSVKTLRKRSSKIYFRNP
MAENSKFFKKHGIRNGINPAMYQSPMRLRSSTIQSSSIILLDTLLLTENEGDKTEEQVSY
V
|
|
|
|---|
| BDBM50130841 |
|---|
| n/a |
|---|
| Name | BDBM50130841 |
|---|
| Synonyms: | CHEMBL3634745 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H19FN2O2 |
|---|
| Mol. Mass. | 422.4504 |
|---|
| SMILES | Fc1ccc(cc1)-c1[nH]c(=O)ccc1-c1ccc(OCc2ccc3ccccc3n2)cc1 |
|---|
| Structure | 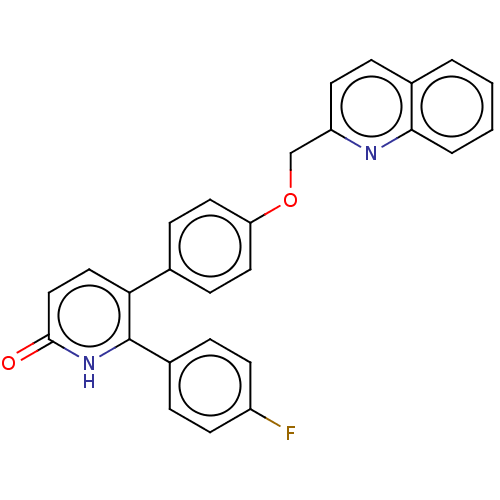
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lingam, VS; Dahale, DH; Rathi, VE; Shingote, YB; Thakur, RR; Mindhe, AS; Kummari, S; Khairatkar-Joshi, N; Bajpai, M; Shah, DM; Sapalya, RS; Gullapalli, S; Gupta, PK; Gudi, GS; Jadhav, SB; Pattem, R; Thomas, A Design, Synthesis, and Pharmacological Evaluation of 5,6-Disubstituted Pyridin-2(1H)-one Derivatives as Phosphodiesterase 10A (PDE10A) Antagonists. J Med Chem58:8292-308 (2015) [PubMed] Article
Lingam, VS; Dahale, DH; Rathi, VE; Shingote, YB; Thakur, RR; Mindhe, AS; Kummari, S; Khairatkar-Joshi, N; Bajpai, M; Shah, DM; Sapalya, RS; Gullapalli, S; Gupta, PK; Gudi, GS; Jadhav, SB; Pattem, R; Thomas, A Design, Synthesis, and Pharmacological Evaluation of 5,6-Disubstituted Pyridin-2(1H)-one Derivatives as Phosphodiesterase 10A (PDE10A) Antagonists. J Med Chem58:8292-308 (2015) [PubMed] Article