

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50079267 Congo Red::Direct red 28::Kongorot::Sodium diphenyldiazo-bis-alpha-naphthylaminesulfonate::disodium 3,3'-((1,1'-biphenyl)-4,4'-diylbis(azo))bis(4-aminonaphthalene-1-sulphonate)::disodium 3,3'-(biphenyl-4,4'-diyldidiazene-2,1-diyl)bis(4-aminonaphthalene-1-sulfonate)
SMILES: Nc1c(cc(c2ccccc12)S([O-])(=O)=O)\N=N\c1ccc(cc1)-c1ccc(cc1)\N=N\c1cc(c2ccccc2c1N)S([O-])(=O)=O
InChI Key: InChIKey=HFHIDKQMGIGARX-ATXIYDNESA-L
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Choline acetylase (Homo sapiens (Human)) | BDBM50079267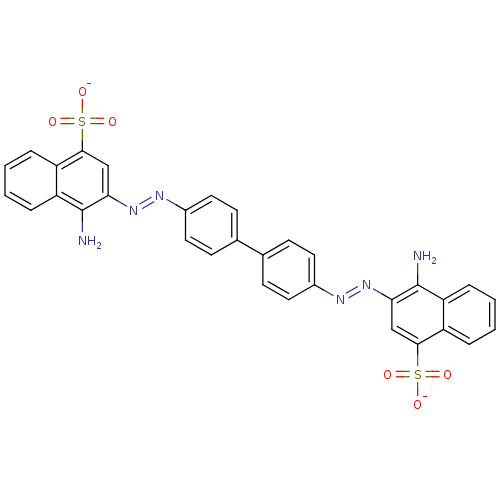 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | PubMed | 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of choline acetyltransferase isolated from squid head ganglia | J Med Chem 24: 1534-7 (1982) BindingDB Entry DOI: 10.7270/Q2T43TMK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta amyloid A4 protein (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagasaki University Curated by ChEMBL | Assay Description Displacement of [125I]6-iodo-4'-dimethyaminofl avone from beta amyloid (1-42) protein aggregates | J Med Chem 48: 7253-60 (2005) Article DOI: 10.1021/jm050635e BindingDB Entry DOI: 10.7270/Q2H70FC0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta amyloid A4 protein (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagasaki University Curated by ChEMBL | Assay Description Displacement of [125I]6-iodo-4'-dimethyaminofl avone from beta amyloid (1-40) protein aggregates | J Med Chem 48: 7253-60 (2005) Article DOI: 10.1021/jm050635e BindingDB Entry DOI: 10.7270/Q2H70FC0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta amyloid A4 protein (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Normal University Curated by ChEMBL | Assay Description Displacement of [125I-N-Methyl-4-(4-bromoanilino)phthalimide from beta-amyloid plaques isolated from Alzheimer's disease patient brain | Bioorg Med Chem 18: 1337-43 (2010) Article DOI: 10.1016/j.bmc.2009.12.023 BindingDB Entry DOI: 10.7270/Q2445Q9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ectonucleotide pyrophosphatase/phosphodiesterase family member 2 (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | 1.89E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Memphis Curated by ChEMBL | Assay Description Noncompetitive inhibition of human recombinant C-terminal FLAG-tagged autotaxin expressed in baculovirus-infected Sf9 cells assessed as FS-3 hydrolys... | J Med Chem 53: 3095-105 (2010) Article DOI: 10.1021/jm901718z BindingDB Entry DOI: 10.7270/Q2QZ2BXS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-galactosidase (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of Beta-galactosidase | J Med Chem 45: 1712-22 (2002) BindingDB Entry DOI: 10.7270/Q20C4WHD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase AmpC (Escherichia coli) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 3.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibitory activity against beta-lactamase in the presence of 500 mMKPi concentration of buffer | J Med Chem 45: 1712-22 (2002) BindingDB Entry DOI: 10.7270/Q20C4WHD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase AmpC (Escherichia coli) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibitory activity against Amp C beta-Lactamase | J Med Chem 46: 4265-72 (2003) Article DOI: 10.1021/jm030266r BindingDB Entry DOI: 10.7270/Q29C6Z59 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase AmpC (Escherichia coli) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibitory activity against Beta-lactamase from DMSO stock was determined | J Med Chem 46: 4265-72 (2003) Article DOI: 10.1021/jm030266r BindingDB Entry DOI: 10.7270/Q29C6Z59 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ectonucleotide pyrophosphatase/phosphodiesterase family member 2 (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.31E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Memphis Curated by ChEMBL | Assay Description Inhibition of human recombinant C-terminal FLAG-tagged autotaxin expressed in baculovirus-infected Sf9 cells assessed as FS-3 hydrolysis | J Med Chem 53: 3095-105 (2010) Article DOI: 10.1021/jm901718z BindingDB Entry DOI: 10.7270/Q2QZ2BXS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta amyloid A4 protein (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AC Immune SA Curated by ChEMBL | Assay Description Inhibition of HFIP-pretreated amyloid beta (1 to 42 residues) (unknown origin) aggregation after 24 hrs by ThT fluorescence assay | Bioorg Med Chem Lett 27: 1405-1411 (2017) Article DOI: 10.1016/j.bmcl.2017.02.001 BindingDB Entry DOI: 10.7270/Q2R49TSG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta amyloid A4 protein (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AC Immune SA Curated by ChEMBL | Assay Description Inhibition of Abeta42 (unknown origin) aggregation measured after 24 hrs by ThT fluorescence assay | Bioorg Med Chem Lett 26: 3330-3335 (2016) Article DOI: 10.1016/j.bmcl.2016.05.040 BindingDB Entry DOI: 10.7270/Q2XW4NT3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase AmpC (Escherichia coli) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibitory activity against beta-lactamase in the presence of 50 mM KPi concentration of buffer | J Med Chem 45: 1712-22 (2002) BindingDB Entry DOI: 10.7270/Q20C4WHD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Gallus gallus (Chicken)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibitory activity against cloned Dihydrofolate reductase (cDHFR) | J Med Chem 45: 1712-22 (2002) BindingDB Entry DOI: 10.7270/Q20C4WHD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Chymotrypsin (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibitory activity against chymotrypsinogen | J Med Chem 45: 1712-22 (2002) BindingDB Entry DOI: 10.7270/Q20C4WHD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase AmpC (Escherichia coli) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 650 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibitory activity against beta-lactamase in the presence of 5 mM KPi concentration of buffer | J Med Chem 45: 1712-22 (2002) BindingDB Entry DOI: 10.7270/Q20C4WHD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| PH domain leucine-rich repeat-containing protein phosphatase 2 (Homo sapiens (Human)) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California San Diego Curated by ChEMBL | Assay Description Inhibition of PHLPP2 phosphatase domain expressed in Escherichia coli | J Med Chem 53: 6899-911 (2010) Article DOI: 10.1021/jm100331d BindingDB Entry DOI: 10.7270/Q2154H8Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase AmpC (Escherichia coli) | BDBM50079267 (Congo Red | Direct red 28 | Kongorot | Sodium diph...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibitory activity against beta-lactamase | J Med Chem 45: 1712-22 (2002) BindingDB Entry DOI: 10.7270/Q20C4WHD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||