

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenosine deaminase (Homo sapiens (Human)) | BDBM22925 ((8R)-3-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)oxol...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | 0.0330 | -59.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Fujisawa Pharmaceutical Co., Ltd. | Assay Description The reaction velocity was measured by change in absorbance at 265nm (A265) resulting from the deamination of adenosine. The reaction was started by a... | Bioorg Med Chem Lett 13: 1115-8 (2003) Article DOI: 10.1016/S0960-894X(03)00026-X BindingDB Entry DOI: 10.7270/Q29Z936D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50060964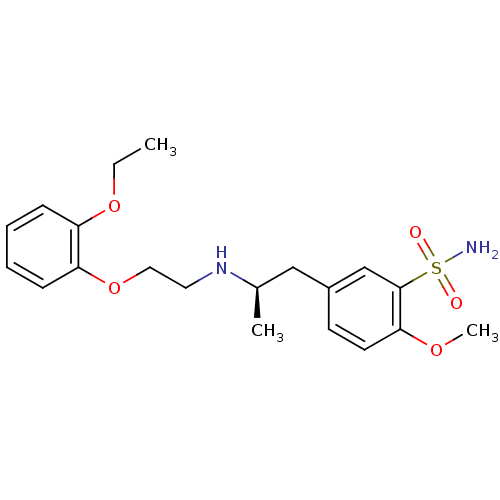 ((R)-5-(2-((2-(2-ethoxyphenoxy)ethyl)amino)propyl)-...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of [125I] HEAT from human alpha1D adrenergic receptor expressed in Chlorocebus aethiops COS1 cell membranes incubated for 60 mins by liq... | J Med Chem 59: 2989-3002 (2016) Article DOI: 10.1021/acs.jmedchem.5b01528 BindingDB Entry DOI: 10.7270/Q20G3N2B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM86231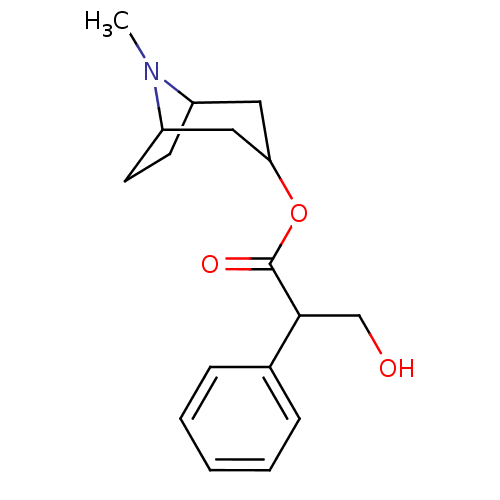 (ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50456840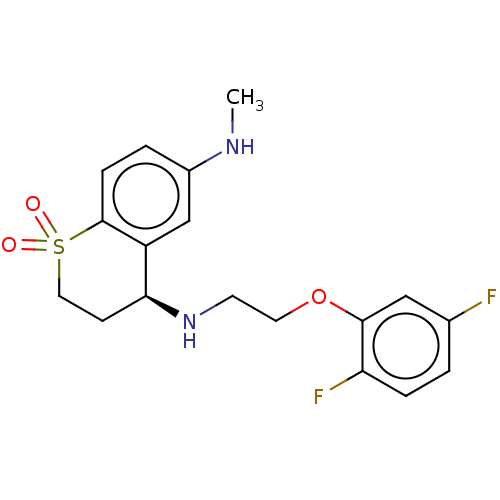 (CHEMBL4207824) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of 7-methoxy-[3H]-prazosin from human alpha1D-adrenoreceptor expressed in CHOK1 cell membranes after 60 mins by TopCount liquid scintill... | Eur J Med Chem 139: 114-127 (2017) Article DOI: 10.1016/j.ejmech.2017.07.071 BindingDB Entry DOI: 10.7270/Q2765HZH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M4 (Homo sapiens (Human)) | BDBM86231 (ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM82372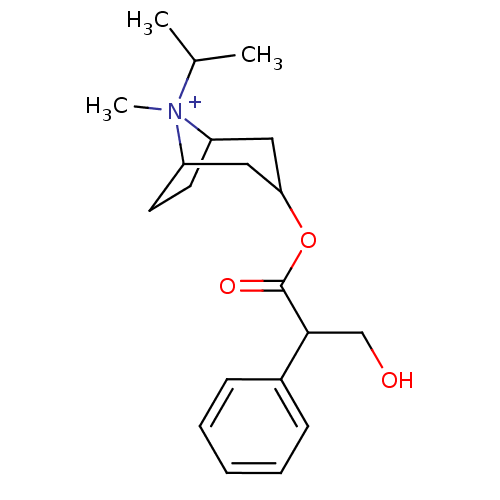 (CAS_22254-24-6 | Ipratropium | NSC_3746) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM86231 (ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM82372 (CAS_22254-24-6 | Ipratropium | NSC_3746) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M5 (Homo sapiens (Human)) | BDBM86231 (ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.540 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50164686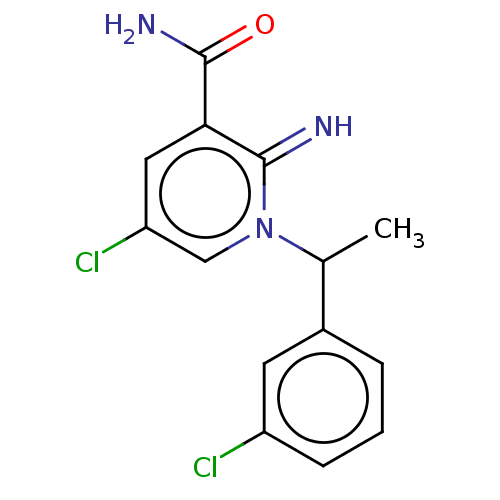 (CHEMBL3798124) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of 7-methoxy-[3H]-prazosin from human alpha1D adrenergic receptor expressed in CHO-K1 cell membranes incubated for 60 mins by liquid sci... | J Med Chem 59: 2989-3002 (2016) Article DOI: 10.1021/acs.jmedchem.5b01528 BindingDB Entry DOI: 10.7270/Q20G3N2B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M4 (Homo sapiens (Human)) | BDBM82372 (CAS_22254-24-6 | Ipratropium | NSC_3746) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50109647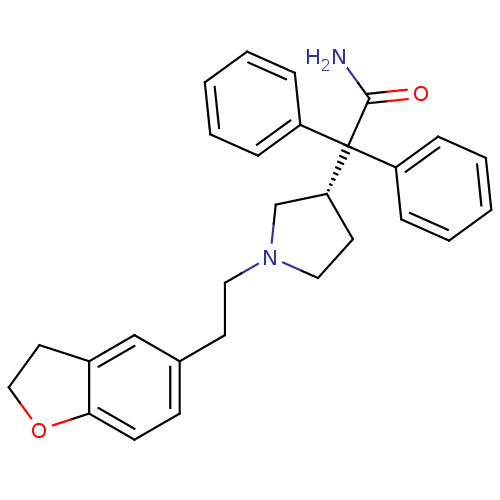 (2-{1-[2-(2,3-Dihydro-benzofuran-5-yl)-ethyl]-pyrro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | DrugBank PubMed | 0.840 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50456841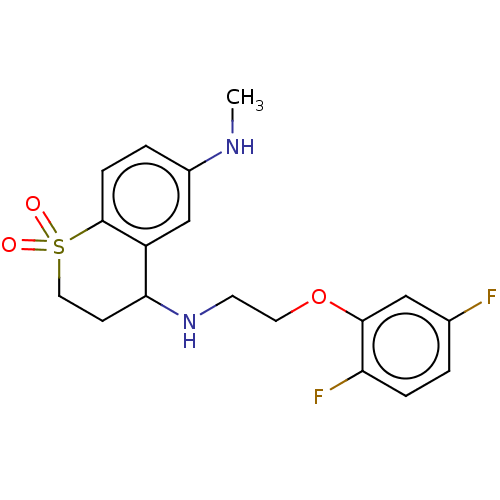 (CHEMBL4218307) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of 7-methoxy-[3H]-prazosin from human alpha1D-adrenoreceptor expressed in CHOK1 cell membranes after 60 mins by TopCount liquid scintill... | Eur J Med Chem 139: 114-127 (2017) Article DOI: 10.1016/j.ejmech.2017.07.071 BindingDB Entry DOI: 10.7270/Q2765HZH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50164745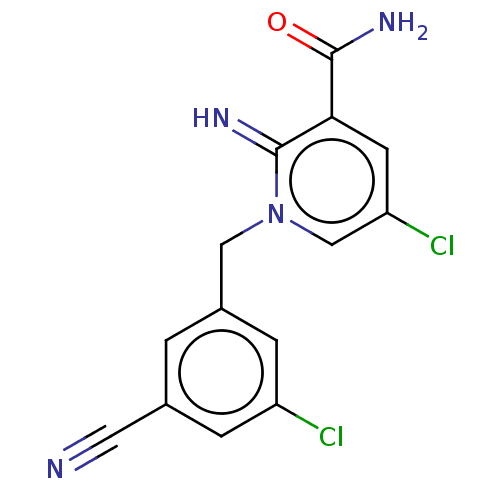 (CHEMBL3800606) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.860 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of 7-methoxy-[3H]-prazosin from human alpha1D adrenergic receptor expressed in CHO-K1 cell membranes incubated for 60 mins by liquid sci... | J Med Chem 59: 2989-3002 (2016) Article DOI: 10.1021/acs.jmedchem.5b01528 BindingDB Entry DOI: 10.7270/Q20G3N2B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095656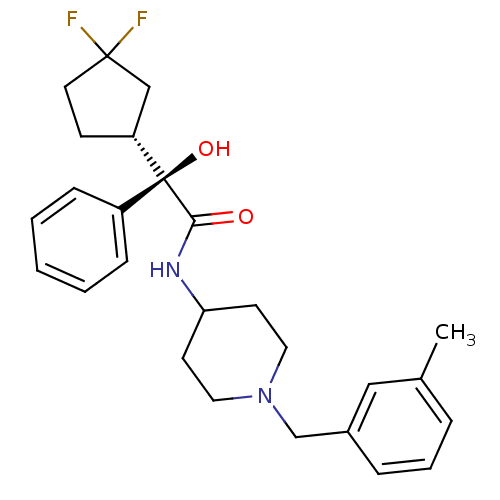 (2-(3,3-Difluoro-cyclopentyl)-2-hydroxy-N-[1-(3-met...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.880 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095656 (2-(3,3-Difluoro-cyclopentyl)-2-hydroxy-N-[1-(3-met...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.880 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50456848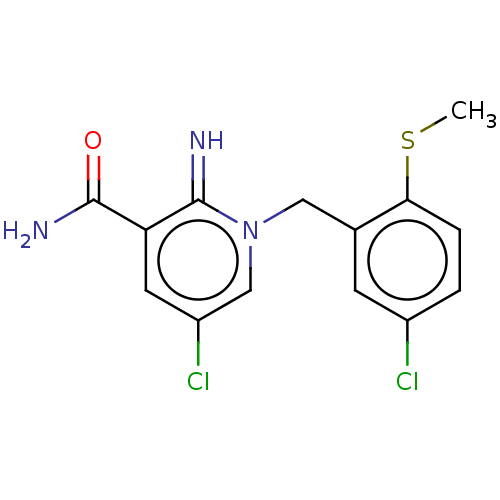 (CHEMBL4208816) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of 7-methoxy-[3H]-prazosin from human alpha1D-adrenoreceptor expressed in CHOK1 cell membranes after 60 mins by TopCount liquid scintill... | Eur J Med Chem 139: 114-127 (2017) Article DOI: 10.1016/j.ejmech.2017.07.071 BindingDB Entry DOI: 10.7270/Q2765HZH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50164769 (CHEMBL3799553) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of 7-methoxy-[3H]-prazosin from human alpha1D adrenergic receptor expressed in CHO-K1 cell membranes incubated for 60 mins by liquid sci... | J Med Chem 59: 2989-3002 (2016) Article DOI: 10.1021/acs.jmedchem.5b01528 BindingDB Entry DOI: 10.7270/Q20G3N2B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095679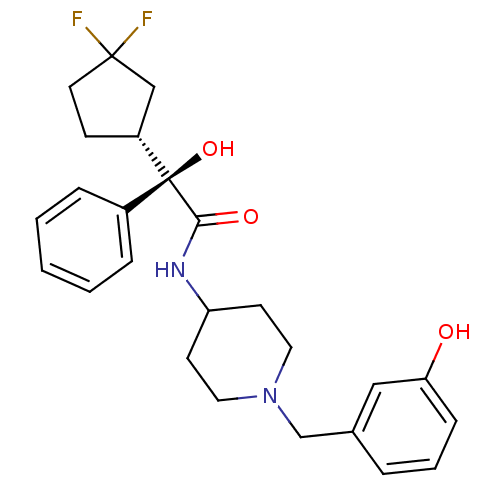 (2-(3,3-Difluoro-cyclopentyl)-2-hydroxy-N-[1-(3-hyd...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095679 (2-(3,3-Difluoro-cyclopentyl)-2-hydroxy-N-[1-(3-hyd...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095662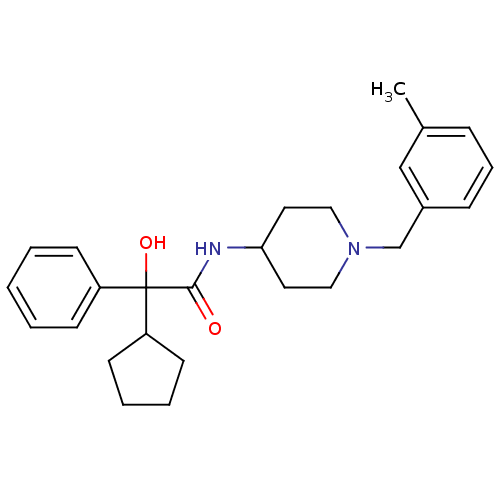 (2-Cyclopentyl-2-hydroxy-N-[1-(3-methyl-benzyl)-pip...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095662 (2-Cyclopentyl-2-hydroxy-N-[1-(3-methyl-benzyl)-pip...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50456851 (CHEMBL4213029) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of 7-methoxy-[3H]-prazosin from human alpha1D-adrenoreceptor expressed in CHOK1 cell membranes after 60 mins by TopCount liquid scintill... | Eur J Med Chem 139: 114-127 (2017) Article DOI: 10.1016/j.ejmech.2017.07.071 BindingDB Entry DOI: 10.7270/Q2765HZH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095657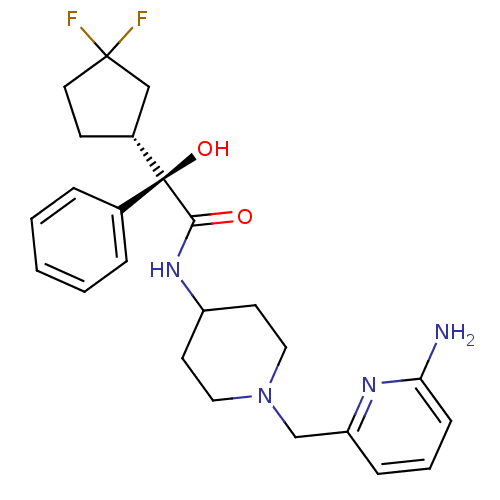 ((R)-N-[1-(6-Amino-pyridin-2-ylmethyl)-piperidin-4-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM82372 (CAS_22254-24-6 | Ipratropium | NSC_3746) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Patents Similars | PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM86231 (ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095657 ((R)-N-[1-(6-Amino-pyridin-2-ylmethyl)-piperidin-4-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50164687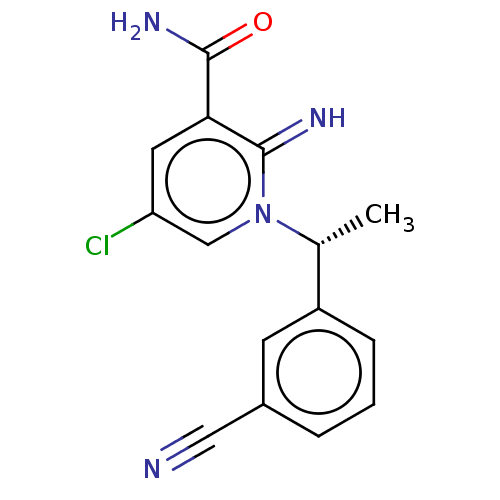 (CHEMBL3799292) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of 7-methoxy-[3H]-prazosin from human alpha1D adrenergic receptor expressed in CHO-K1 cell membranes incubated for 60 mins by liquid sci... | J Med Chem 59: 2989-3002 (2016) Article DOI: 10.1021/acs.jmedchem.5b01528 BindingDB Entry DOI: 10.7270/Q20G3N2B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M5 (Homo sapiens (Human)) | BDBM82372 (CAS_22254-24-6 | Ipratropium | NSC_3746) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Patents Similars | PubMed | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (RAT) | BDBM85787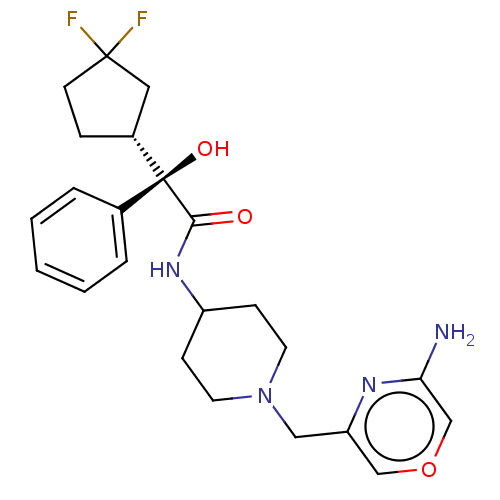 (Compound A) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | PubMed | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50095656 (2-(3,3-Difluoro-cyclopentyl)-2-hydroxy-N-[1-(3-met...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Ability to inhibit binding of [3H]-NMS was determined by receptor binding assay using membranes from chinese hamster ovary (CHO) cells expressing clo... | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50095656 (2-(3,3-Difluoro-cyclopentyl)-2-hydroxy-N-[1-(3-met...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Ability to inhibit binding of [3H]-NMS was determined by receptor binding assay using membranes from chinese hamster ovary (CHO) cells expressing clo... | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095680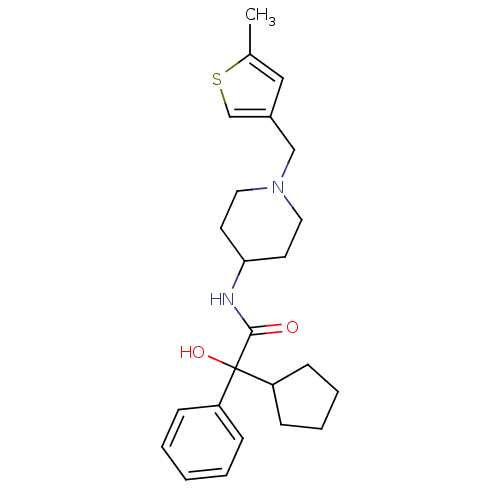 (2-Cyclopentyl-2-hydroxy-N-[1-(5-methyl-thiophen-3-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095680 (2-Cyclopentyl-2-hydroxy-N-[1-(5-methyl-thiophen-3-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50095662 (2-Cyclopentyl-2-hydroxy-N-[1-(3-methyl-benzyl)-pip...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M3 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50095662 (2-Cyclopentyl-2-hydroxy-N-[1-(3-methyl-benzyl)-pip...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M3 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M5 (Homo sapiens (Human)) | BDBM50109647 (2-{1-[2-(2,3-Dihydro-benzofuran-5-yl)-ethyl]-pyrro...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | DrugBank PubMed | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095670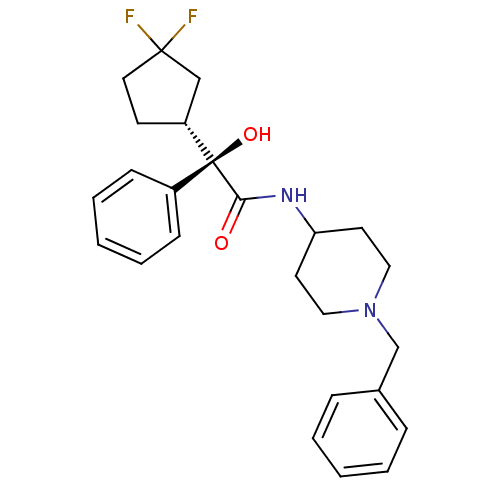 (CHEMBL147653 | N-(1-Benzyl-piperidin-4-yl)-2-(3,3-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095670 (CHEMBL147653 | N-(1-Benzyl-piperidin-4-yl)-2-(3,3-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095685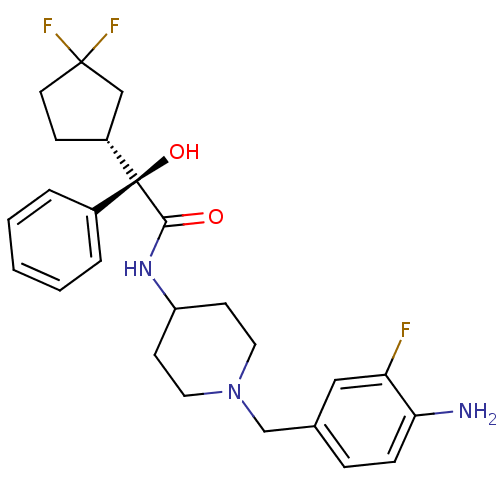 (CHEMBL356181 | N-[1-(4-Amino-3-fluoro-benzyl)-pipe...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50095661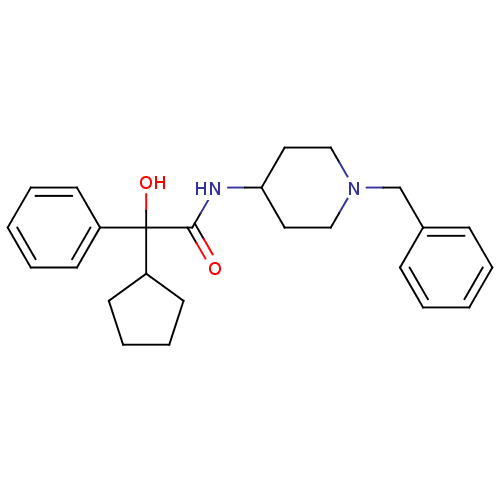 (CHEMBL146464 | N-(1-Benzyl-piperidin-4-yl)-2-cyclo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M3 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50095661 (CHEMBL146464 | N-(1-Benzyl-piperidin-4-yl)-2-cyclo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M3 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095653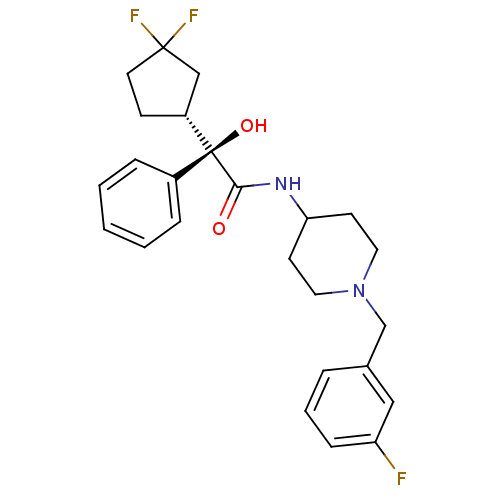 (2-(3,3-Difluoro-cyclopentyl)-N-[1-(3-fluoro-benzyl...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095653 (2-(3,3-Difluoro-cyclopentyl)-N-[1-(3-fluoro-benzyl...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095685 (CHEMBL356181 | N-[1-(4-Amino-3-fluoro-benzyl)-pipe...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095661 (CHEMBL146464 | N-(1-Benzyl-piperidin-4-yl)-2-cyclo...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1D adrenergic receptor (Homo sapiens (Human)) | BDBM50164739 (CHEMBL3800305) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Displacement of 7-methoxy-[3H]-prazosin from human alpha1D adrenergic receptor expressed in CHO-K1 cell membranes incubated for 60 mins by liquid sci... | J Med Chem 59: 2989-3002 (2016) Article DOI: 10.1021/acs.jmedchem.5b01528 BindingDB Entry DOI: 10.7270/Q20G3N2B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (RAT) | BDBM85787 (Compound A) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Pharmaceutical Co., Ltd. Curated by PDSP Ki Database | J Pharmacol Exp Ther 297: 790-7 (2001) BindingDB Entry DOI: 10.7270/Q24T6GX9 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50095661 (CHEMBL146464 | N-(1-Benzyl-piperidin-4-yl)-2-cyclo...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M1 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50095657 ((R)-N-[1-(6-Amino-pyridin-2-ylmethyl)-piperidin-4-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of [3H]-NMS binding to human muscarinic acetylcholine receptor M3 expressed in CHO cells | J Med Chem 43: 5017-29 (2001) BindingDB Entry DOI: 10.7270/Q2HM57P7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 1370 total ) | Next | Last >> |