 Found 197 hits with Last Name = 'warrack' and Initial = 'bm'
Found 197 hits with Last Name = 'warrack' and Initial = 'bm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM23165
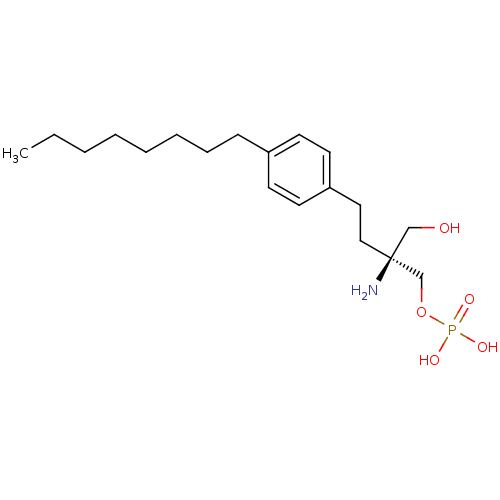
(CHEMBL366208 | FTY720-phosphate, (S)-2 | [(2S)-2-a...)Show SMILES CCCCCCCCc1ccc(CC[C@](N)(CO)COP(O)(O)=O)cc1 |r| Show InChI InChI=1S/C19H34NO5P/c1-2-3-4-5-6-7-8-17-9-11-18(12-10-17)13-14-19(20,15-21)16-25-26(22,23)24/h9-12,21H,2-8,13-16,20H2,1H3,(H2,22,23,24)/t19-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [33P]-SIP from human recombinant S1P1 expressed in CHO cell membranes measured after 45 mins by radioligand competitive binding analy... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01695
BindingDB Entry DOI: 10.7270/Q2TQ659P |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM23163
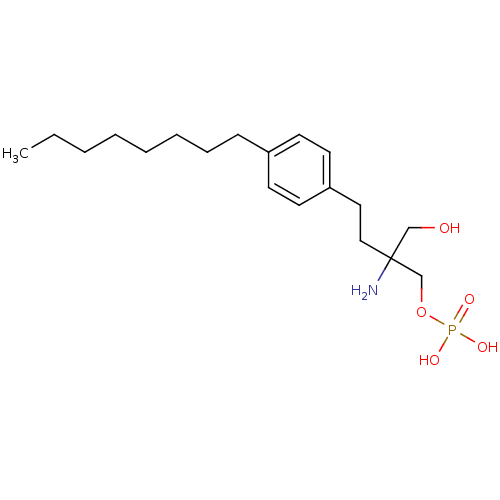
(CHEMBL114606 | FTY720-phosphate, rac-2 | {2-amino-...)Show InChI InChI=1S/C19H34NO5P/c1-2-3-4-5-6-7-8-17-9-11-18(12-10-17)13-14-19(20,15-21)16-25-26(22,23)24/h9-12,21H,2-8,13-16,20H2,1H3,(H2,22,23,24) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [33P]-SIP from human recombinant S1P1 expressed in CHO cell membranes measured after 45 mins by TopCount scintillation counting metho... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01109
BindingDB Entry DOI: 10.7270/Q28919KX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50169441

(CHEMBL3806205)Show SMILES CCCCCC[C@@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(COP(O)(O)=O)C1 |r| Show InChI InChI=1S/C22H36NO4P/c1-2-3-4-5-6-17-7-8-19-14-20(10-9-18(19)13-17)21-11-12-22(23,15-21)16-27-28(24,25)26/h9-10,14,17,21H,2-8,11-13,15-16,23H2,1H3,(H2,24,25,26)/t17-,21+,22-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [33P]-SIP from human recombinant S1P1 expressed in CHO cell membranes measured after 45 mins by TopCount scintillation counting metho... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01109
BindingDB Entry DOI: 10.7270/Q28919KX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50169441

(CHEMBL3806205)Show SMILES CCCCCC[C@@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(COP(O)(O)=O)C1 |r| Show InChI InChI=1S/C22H36NO4P/c1-2-3-4-5-6-17-7-8-19-14-20(10-9-18(19)13-17)21-11-12-22(23,15-21)16-27-28(24,25)26/h9-10,14,17,21H,2-8,11-13,15-16,23H2,1H3,(H2,24,25,26)/t17-,21+,22-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [33P]-SIP from human recombinant S1P1 expressed in CHO cell membranes measured after 45 mins by radioligand competitive binding analy... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01695
BindingDB Entry DOI: 10.7270/Q2TQ659P |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM258470
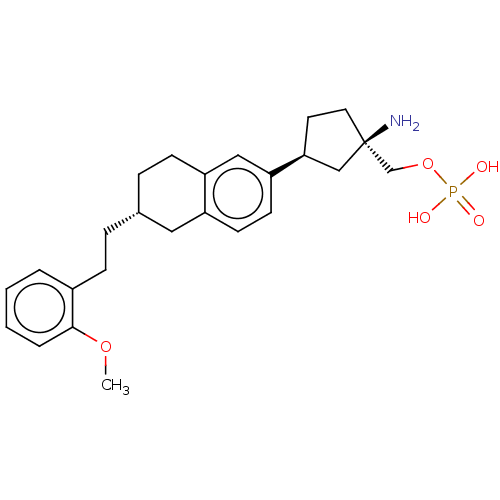
(US9522888, 697)Show SMILES COc1ccccc1CC[C@@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(COP(O)(O)=O)C1 |r| Show InChI InChI=1S/C25H34NO5P/c1-30-24-5-3-2-4-19(24)8-6-18-7-9-21-15-22(11-10-20(21)14-18)23-12-13-25(26,16-23)17-31-32(27,28)29/h2-5,10-11,15,18,23H,6-9,12-14,16-17,26H2,1H3,(H2,27,28,29)/t18-,23+,25-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [33P]-SIP from human recombinant S1P1 expressed in CHO cell membranes measured after 45 mins by radioligand competitive binding analy... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01695
BindingDB Entry DOI: 10.7270/Q2TQ659P |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM258466
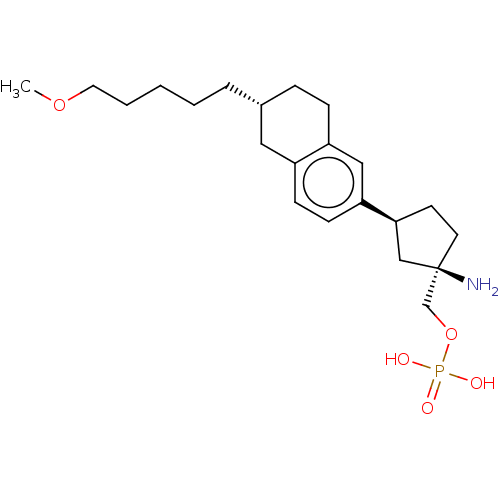
(US9522888, 689)Show SMILES COCCCCC[C@@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(COP(O)(O)=O)C1 |r| Show InChI InChI=1S/C22H36NO5P/c1-27-12-4-2-3-5-17-6-7-19-14-20(9-8-18(19)13-17)21-10-11-22(23,15-21)16-28-29(24,25)26/h8-9,14,17,21H,2-7,10-13,15-16,23H2,1H3,(H2,24,25,26)/t17-,21+,22-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [33P]-SIP from human recombinant S1P1 expressed in CHO cell membranes measured after 45 mins by TopCount scintillation counting metho... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01109
BindingDB Entry DOI: 10.7270/Q28919KX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50562873
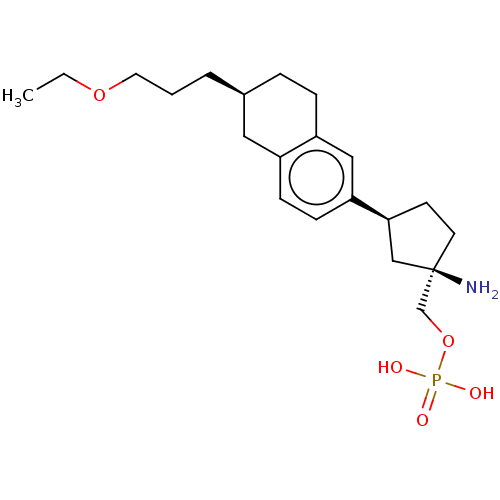
(CHEMBL4786296)Show SMILES CCOCCC[C@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(COP(O)(O)=O)C1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [33P]-SIP from human recombinant S1P1 expressed in CHO cell membranes measured after 45 mins by TopCount scintillation counting metho... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01109
BindingDB Entry DOI: 10.7270/Q28919KX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM197654

(US9216972, 7)Show SMILES OC(=O)C1CN(Cc2ccc3-c4noc(c4CCc3c2)-c2onc(c2C(F)(F)F)-c2ccccc2)C1 Show InChI InChI=1S/C26H20F3N3O4/c27-26(28,29)20-21(15-4-2-1-3-5-15)30-36-24(20)23-19-9-7-16-10-14(6-8-18(16)22(19)31-35-23)11-32-12-17(13-32)25(33)34/h1-6,8,10,17H,7,9,11-13H2,(H,33,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 0.380 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Binding affinity to human S1P1 |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24X5CD0 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156232
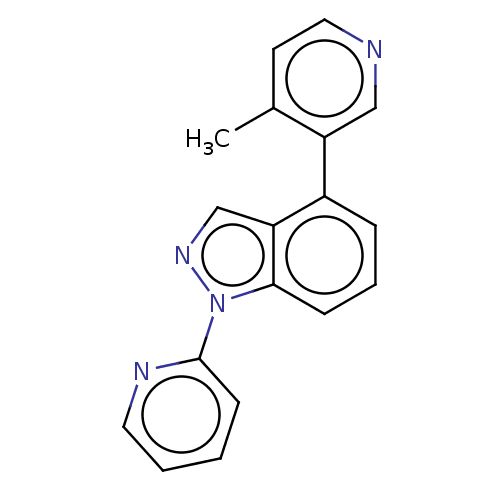
(CHEMBL3782020)Show InChI InChI=1S/C18H14N4/c1-13-8-10-19-11-15(13)14-5-4-6-17-16(14)12-21-22(17)18-7-2-3-9-20-18/h2-12H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Macaca fascicularis) | BDBM50156282
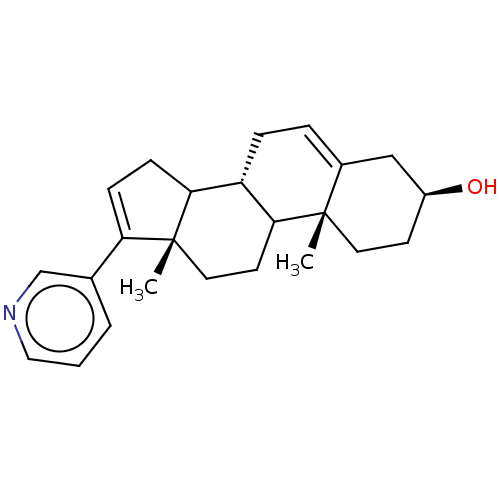
(CHEMBL3780847)Show SMILES [H][C@@]12CC=C3C[C@@H](O)CC[C@]3(C)C1CC[C@@]1(C)C2CC=C1c1cccnc1 |r,c:22,t:3| Show InChI InChI=1S/C24H31NO/c1-23-11-9-18(26)14-17(23)5-6-19-21-8-7-20(16-4-3-13-25-15-16)24(21,2)12-10-22(19)23/h3-5,7,13,15,18-19,21-22,26H,6,8-12,14H2,1-2H3/t18-,19-,21?,22?,23-,24+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP17A1 in cynomolgus monkey using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity assay in presence of... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Macaca fascicularis) | BDBM50391846

(CHEMBL2147041 | US9133160, 31)Show InChI InChI=1S/C15H12F3N3/c1-10-5-6-19-7-12(10)11-3-2-4-13-14(11)20-9-21(13)8-15(16,17)18/h2-7,9H,8H2,1H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP17A1 in cynomolgus monkey adrenal microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity a... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Macaca fascicularis) | BDBM50156282

(CHEMBL3780847)Show SMILES [H][C@@]12CC=C3C[C@@H](O)CC[C@]3(C)C1CC[C@@]1(C)C2CC=C1c1cccnc1 |r,c:22,t:3| Show InChI InChI=1S/C24H31NO/c1-23-11-9-18(26)14-17(23)5-6-19-21-8-7-20(16-4-3-13-25-15-16)24(21,2)12-10-22(19)23/h3-5,7,13,15,18-19,21-22,26H,6,8-12,14H2,1-2H3/t18-,19-,21?,22?,23-,24+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP17A1 in cynomolgus monkey adrenal microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity a... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156233
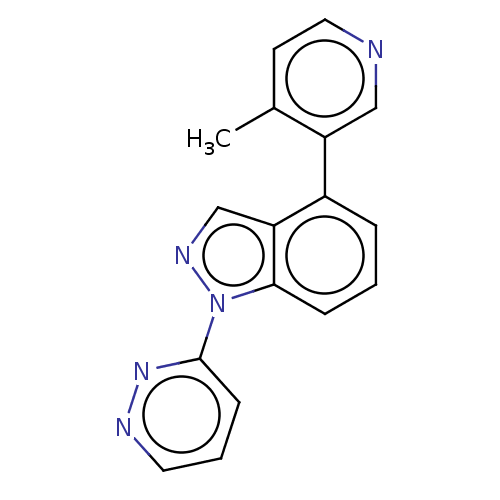
(CHEMBL3780658)Show InChI InChI=1S/C17H13N5/c1-12-7-9-18-10-14(12)13-4-2-5-16-15(13)11-20-22(16)17-6-3-8-19-21-17/h2-11H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156235
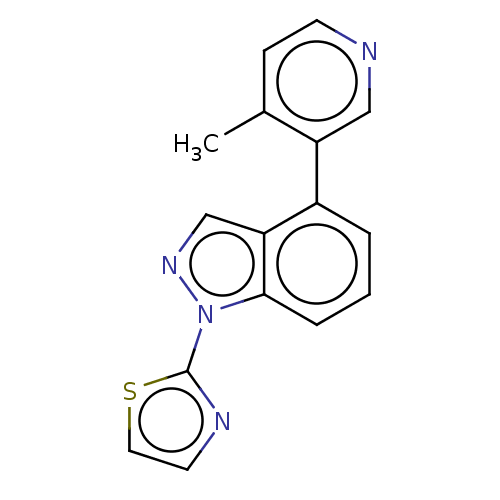
(CHEMBL3780266)Show InChI InChI=1S/C16H12N4S/c1-11-5-6-17-9-13(11)12-3-2-4-15-14(12)10-19-20(15)16-18-7-8-21-16/h2-10H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156281
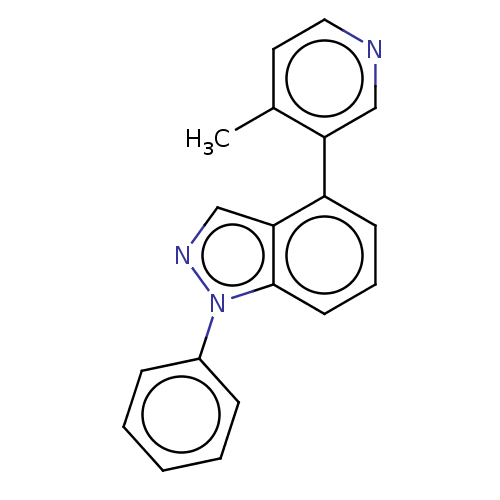
(CHEMBL3780743)Show InChI InChI=1S/C19H15N3/c1-14-10-11-20-12-17(14)16-8-5-9-19-18(16)13-21-22(19)15-6-3-2-4-7-15/h2-13H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Macaca fascicularis) | BDBM50391846

(CHEMBL2147041 | US9133160, 31)Show InChI InChI=1S/C15H12F3N3/c1-10-5-6-19-7-12(10)11-3-2-4-13-14(11)20-9-21(13)8-15(16,17)18/h2-7,9H,8H2,1H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP17A1 in cynomolgus monkey using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity assay in presence of... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156231

(CHEMBL3781112)Show InChI InChI=1S/C19H14FN3/c1-13-9-10-21-11-17(13)16-3-2-4-19-18(16)12-22-23(19)15-7-5-14(20)6-8-15/h2-12H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156234
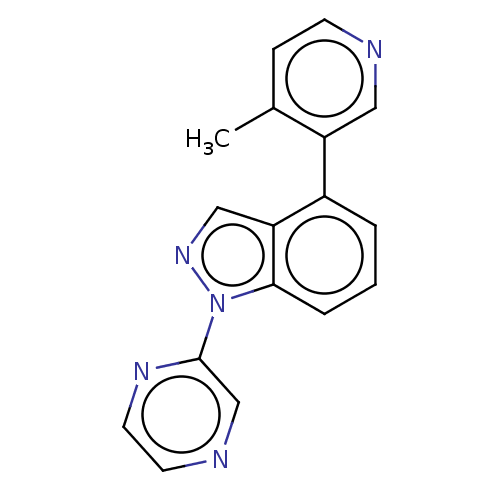
(CHEMBL3780226)Show InChI InChI=1S/C17H13N5/c1-12-5-6-18-9-14(12)13-3-2-4-16-15(13)10-21-22(16)17-11-19-7-8-20-17/h2-11H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156236
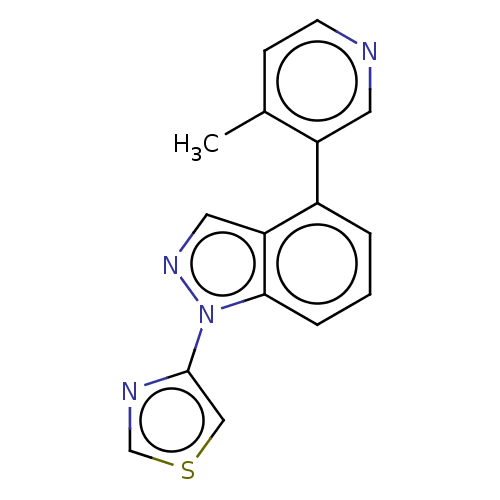
(CHEMBL3780048)Show InChI InChI=1S/C16H12N4S/c1-11-5-6-17-7-13(11)12-3-2-4-15-14(12)8-19-20(15)16-9-21-10-18-16/h2-10H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50391839
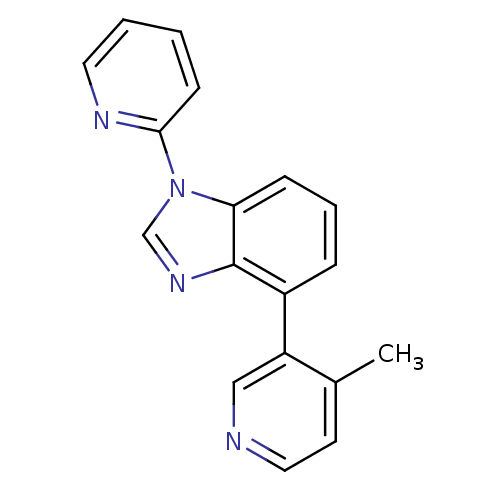
(CHEMBL2147034 | US9133160, 2)Show InChI InChI=1S/C18H14N4/c1-13-8-10-19-11-15(13)14-5-4-6-16-18(14)21-12-22(16)17-7-2-3-9-20-17/h2-12H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156238
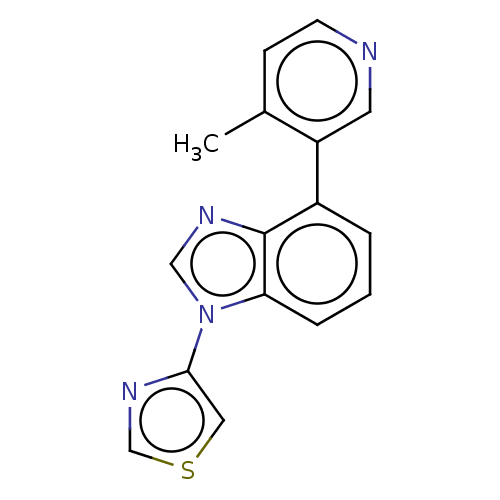
(CHEMBL3781910)Show InChI InChI=1S/C16H12N4S/c1-11-5-6-17-7-13(11)12-3-2-4-14-16(12)18-9-20(14)15-8-21-10-19-15/h2-10H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156237
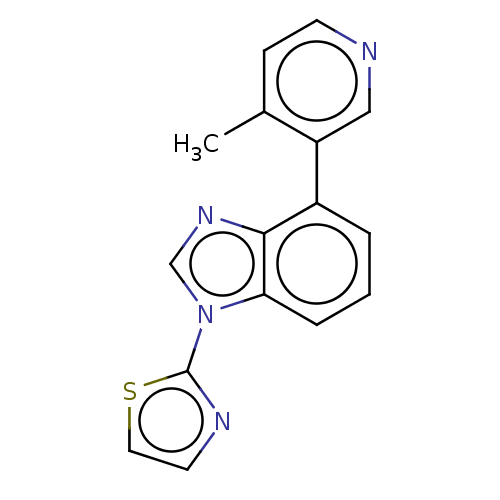
(CHEMBL3781487)Show InChI InChI=1S/C16H12N4S/c1-11-5-6-17-9-13(11)12-3-2-4-14-15(12)19-10-20(14)16-18-7-8-21-16/h2-10H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50391846

(CHEMBL2147041 | US9133160, 31)Show InChI InChI=1S/C15H12F3N3/c1-10-5-6-19-7-12(10)11-3-2-4-13-14(11)20-9-21(13)8-15(16,17)18/h2-7,9H,8H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50391846

(CHEMBL2147041 | US9133160, 31)Show InChI InChI=1S/C15H12F3N3/c1-10-5-6-19-7-12(10)11-3-2-4-13-14(11)20-9-21(13)8-15(16,17)18/h2-7,9H,8H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP17A1 in human adrenal microsomes using [3H]-pregnenolone as substrate incubated for 90 mins by scintillation proximity assay in pres... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50156282

(CHEMBL3780847)Show SMILES [H][C@@]12CC=C3C[C@@H](O)CC[C@]3(C)C1CC[C@@]1(C)C2CC=C1c1cccnc1 |r,c:22,t:3| Show InChI InChI=1S/C24H31NO/c1-23-11-9-18(26)14-17(23)5-6-19-21-8-7-20(16-4-3-13-25-15-16)24(21,2)12-10-22(19)23/h3-5,7,13,15,18-19,21-22,26H,6,8-12,14H2,1-2H3/t18-,19-,21?,22?,23-,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP17A1 in human adrenal microsomes using [3H]-pregnenolone as substrate incubated for 90 mins by scintillation proximity assay in pres... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50391840
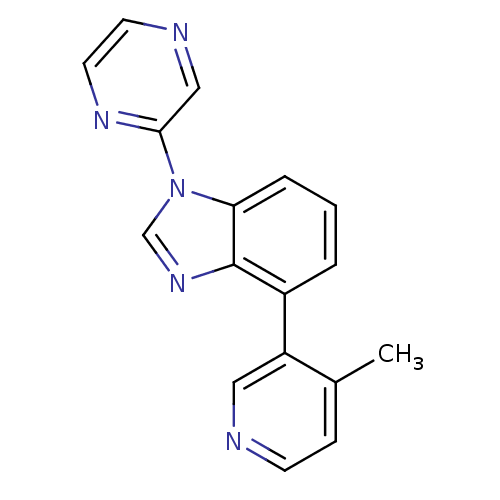
(CHEMBL2147035 | US9133160, 4)Show InChI InChI=1S/C17H13N5/c1-12-5-6-18-9-14(12)13-3-2-4-15-17(13)21-11-22(15)16-10-19-7-8-20-16/h2-11H,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human CYP17A1 expressed in HEK293 cell microsomes using [3H]-pregnenolone as substrate incubated for 45 mins by scintillation proximity... |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50156236

(CHEMBL3780048)Show InChI InChI=1S/C16H12N4S/c1-11-5-6-17-7-13(11)12-3-2-4-15-14(12)8-19-20(15)16-9-21-10-18-16/h2-10H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50156237

(CHEMBL3781487)Show InChI InChI=1S/C16H12N4S/c1-11-5-6-17-9-13(11)12-3-2-4-14-15(12)19-10-20(14)16-18-7-8-21-16/h2-10H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 75 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50156235

(CHEMBL3780266)Show InChI InChI=1S/C16H12N4S/c1-11-5-6-17-9-13(11)12-3-2-4-15-14(12)10-19-20(15)16-18-7-8-21-16/h2-10H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50391839

(CHEMBL2147034 | US9133160, 2)Show InChI InChI=1S/C18H14N4/c1-13-8-10-19-11-15(13)14-5-4-6-16-18(14)21-12-22(16)17-7-2-3-9-20-17/h2-12H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 94 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50156281

(CHEMBL3780743)Show InChI InChI=1S/C19H15N3/c1-14-10-11-20-12-17(14)16-8-5-9-19-18(16)13-21-22(19)15-6-3-2-4-7-15/h2-13H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 119 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50156232

(CHEMBL3782020)Show InChI InChI=1S/C18H14N4/c1-13-8-10-19-11-15(13)14-5-4-6-17-16(14)12-21-22(17)18-7-2-3-9-20-18/h2-12H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50156238

(CHEMBL3781910)Show InChI InChI=1S/C16H12N4S/c1-11-5-6-17-7-13(11)12-3-2-4-14-16(12)18-9-20(14)15-8-21-10-19-15/h2-10H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50391840

(CHEMBL2147035 | US9133160, 4)Show InChI InChI=1S/C17H13N5/c1-12-5-6-18-9-14(12)13-3-2-4-15-17(13)21-11-22(15)16-10-19-7-8-20-16/h2-11H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50156231

(CHEMBL3781112)Show InChI InChI=1S/C19H14FN3/c1-13-9-10-21-11-17(13)16-3-2-4-19-18(16)12-22-23(19)15-7-5-14(20)6-8-15/h2-12H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50156233

(CHEMBL3780658)Show InChI InChI=1S/C17H13N5/c1-12-7-9-18-10-14(12)13-4-2-5-16-15(13)11-20-22(16)17-6-3-8-19-21-17/h2-11H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 11B1, mitochondrial
(Homo sapiens (Human)) | BDBM50156282

(CHEMBL3780847)Show SMILES [H][C@@]12CC=C3C[C@@H](O)CC[C@]3(C)C1CC[C@@]1(C)C2CC=C1c1cccnc1 |r,c:22,t:3| Show InChI InChI=1S/C24H31NO/c1-23-11-9-18(26)14-17(23)5-6-19-21-8-7-20(16-4-3-13-25-15-16)24(21,2)12-10-22(19)23/h3-5,7,13,15,18-19,21-22,26H,6,8-12,14H2,1-2H3/t18-,19-,21?,22?,23-,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 872 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP11B1 in human H295R cells using deoxycorticosterone as substrate incubated for 48 hrs by LC-MS method |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50169441

(CHEMBL3806205)Show SMILES CCCCCC[C@@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(COP(O)(O)=O)C1 |r| Show InChI InChI=1S/C22H36NO4P/c1-2-3-4-5-6-17-7-8-19-14-20(10-9-18(19)13-17)21-11-12-22(23,15-21)16-27-28(24,25)26/h9-10,14,17,21H,2-8,11-13,15-16,23H2,1H3,(H2,24,25,26)/t17-,21+,22-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Antagonist activity at human S1P3 expressed in EDG3-Ga15-bla HEK293T cells incubated for 45 mins by scintillation counting based [35S]GTP-gamma-S bin... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.6b01433
BindingDB Entry DOI: 10.7270/Q29S1VRX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50558763
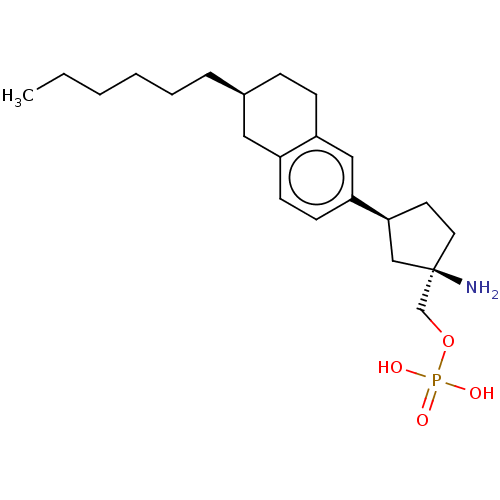
(CHEMBL4762613)Show SMILES CCCCCC[C@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(COP(O)(O)=O)C1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Antagonist activity at human S1P3 expressed in EDG3-Ga15-bla HEK293T cells incubated for 45 mins by scintillation counting based [35S]GTP-gamma-S bin... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.6b01433
BindingDB Entry DOI: 10.7270/Q29S1VRX |
More data for this
Ligand-Target Pair | |
Steroid 21-hydroxylase
(Homo sapiens (Human)) | BDBM50156282

(CHEMBL3780847)Show SMILES [H][C@@]12CC=C3C[C@@H](O)CC[C@]3(C)C1CC[C@@]1(C)C2CC=C1c1cccnc1 |r,c:22,t:3| Show InChI InChI=1S/C24H31NO/c1-23-11-9-18(26)14-17(23)5-6-19-21-8-7-20(16-4-3-13-25-15-16)24(21,2)12-10-22(19)23/h3-5,7,13,15,18-19,21-22,26H,6,8-12,14H2,1-2H3/t18-,19-,21?,22?,23-,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.05E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP21A2 in human H295R cells using hydroxy progesterone as substrate incubated for 120 mins by LC-MS method |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM197654

(US9216972, 7)Show SMILES OC(=O)C1CN(Cc2ccc3-c4noc(c4CCc3c2)-c2onc(c2C(F)(F)F)-c2ccccc2)C1 Show InChI InChI=1S/C26H20F3N3O4/c27-26(28,29)20-21(15-4-2-1-3-5-15)30-36-24(20)23-19-9-7-16-10-14(6-8-18(16)22(19)31-35-23)11-32-12-17(13-32)25(33)34/h1-6,8,10,17H,7,9,11-13H2,(H,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human ERG by patch clamp assay |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24X5CD0 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50558764
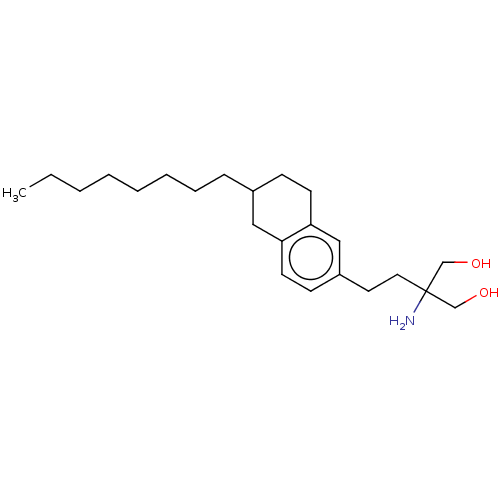
(CHEMBL4750944) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C8 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.6b01433
BindingDB Entry DOI: 10.7270/Q29S1VRX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50156234

(CHEMBL3780226)Show InChI InChI=1S/C17H13N5/c1-12-5-6-18-9-14(12)13-3-2-4-16-15(13)10-21-22(16)17-11-19-7-8-20-17/h2-11H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50558765
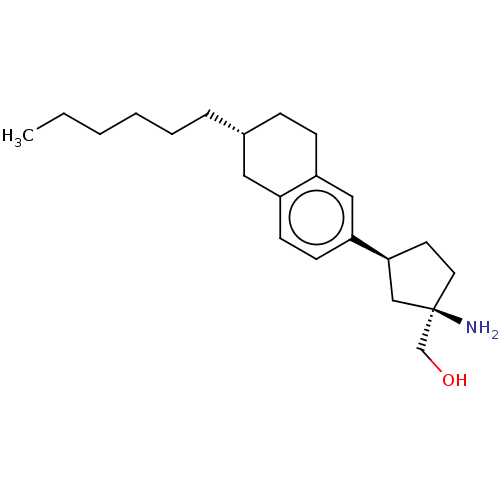
(Bms-986104)Show SMILES CCCCCC[C@@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(CO)C1 |r| | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C8 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.6b01433
BindingDB Entry DOI: 10.7270/Q29S1VRX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50563973

(Bms-986166)Show SMILES COc1ccccc1CC[C@@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(CO)C1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01695
BindingDB Entry DOI: 10.7270/Q2TQ659P |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50563973

(Bms-986166)Show SMILES COc1ccccc1CC[C@@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(CO)C1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01695
BindingDB Entry DOI: 10.7270/Q2TQ659P |
More data for this
Ligand-Target Pair | |
Steroid 21-hydroxylase
(Homo sapiens (Human)) | BDBM50391846

(CHEMBL2147041 | US9133160, 31)Show InChI InChI=1S/C15H12F3N3/c1-10-5-6-19-7-12(10)11-3-2-4-13-14(11)20-9-21(13)8-15(16,17)18/h2-7,9H,8H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP21A2 in human H295R cells using hydroxy progesterone as substrate incubated for 120 mins by LC-MS method |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 11B1, mitochondrial
(Homo sapiens (Human)) | BDBM50391846

(CHEMBL2147041 | US9133160, 31)Show InChI InChI=1S/C15H12F3N3/c1-10-5-6-19-7-12(10)11-3-2-4-13-14(11)20-9-21(13)8-15(16,17)18/h2-7,9H,8H2,1H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP11B1 in human H295R cells using deoxycorticosterone as substrate incubated for 48 hrs by LC-MS method |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50563973

(Bms-986166)Show SMILES COc1ccccc1CC[C@@H]1CCc2cc(ccc2C1)[C@H]1CC[C@](N)(CO)C1 |r| | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C8 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01695
BindingDB Entry DOI: 10.7270/Q2TQ659P |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50391846

(CHEMBL2147041 | US9133160, 31)Show InChI InChI=1S/C15H12F3N3/c1-10-5-6-19-7-12(10)11-3-2-4-13-14(11)20-9-21(13)8-15(16,17)18/h2-7,9H,8H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 7: 40-5 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00310
BindingDB Entry DOI: 10.7270/Q2TH8PK5 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data