 Found 4863 hits with Last Name = 'cohen' and Initial = 'g'
Found 4863 hits with Last Name = 'cohen' and Initial = 'g' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Choline O-acetyltransferase
(RAT) | BDBM50026220
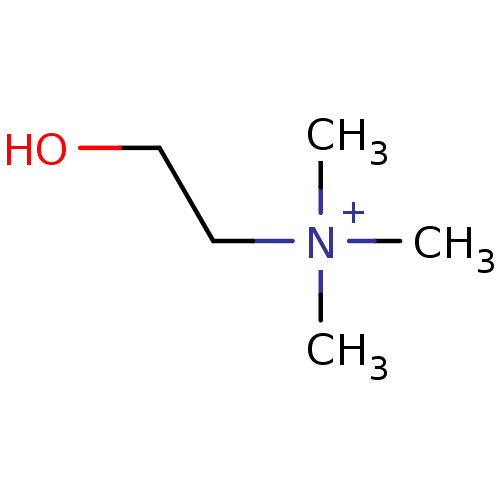
(2-hydroxy-N,N,N-trimethylethanaminium | CHEMBL2824...)Show InChI InChI=1S/C5H14NO/c1-6(2,3)4-5-7/h7H,4-5H2,1-3H3/q+1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 4.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(noncompetitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Choline O-acetyltransferase
(RAT) | BDBM50026470

((2-Hydroxy-ethyl)-dimethyl-sulfonium; iodide | CHE...)Show InChI InChI=1S/C4H11IOS/c1-7(2,5)4-3-6/h6H,3-4H2,1-2H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026480
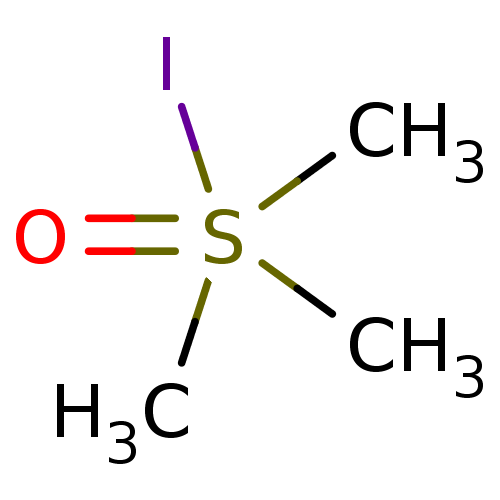
(CHEMBL283149 | Trimethyl-sulfoniumoxide; iodide) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.30E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026468
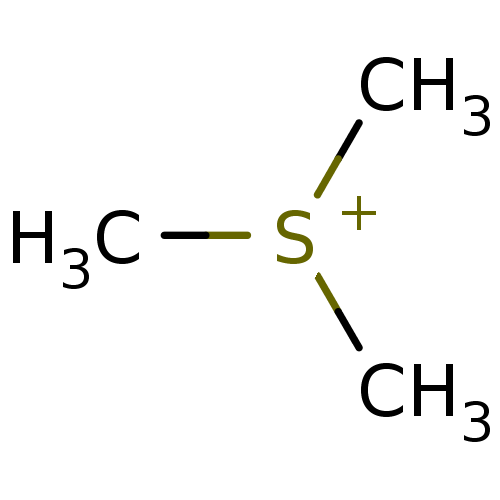
(CHEMBL25340 | Trimethyl-sulfonium; iodide) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
| PubMed
| 2.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026469

(2-Trimethylsilanyl-ethanol | CHEMBL25482)Show InChI InChI=1S/C5H14OSi/c1-7(2,3)5-4-6/h6H,4-5H2,1-3H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
| PubMed
| 3.30E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026475

(Acetic acid 2-methanesulfonyl-ethyl ester | CHEMBL...)Show InChI InChI=1S/C5H10O4S/c1-5(6)9-3-4-10(2,7)8/h3-4H2,1-2H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 6.40E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026475

(Acetic acid 2-methanesulfonyl-ethyl ester | CHEMBL...)Show InChI InChI=1S/C5H10O4S/c1-5(6)9-3-4-10(2,7)8/h3-4H2,1-2H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 6.40E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026468
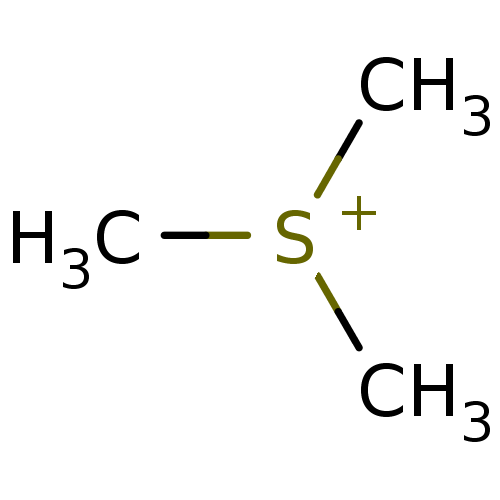
(CHEMBL25340 | Trimethyl-sulfonium; iodide) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
| PubMed
| 7.20E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026220

(2-hydroxy-N,N,N-trimethylethanaminium | CHEMBL2824...)Show InChI InChI=1S/C5H14NO/c1-6(2,3)4-5-7/h7H,4-5H2,1-3H3/q+1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 7.60E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(noncompetitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Choline O-acetyltransferase
(RAT) | BDBM50026477
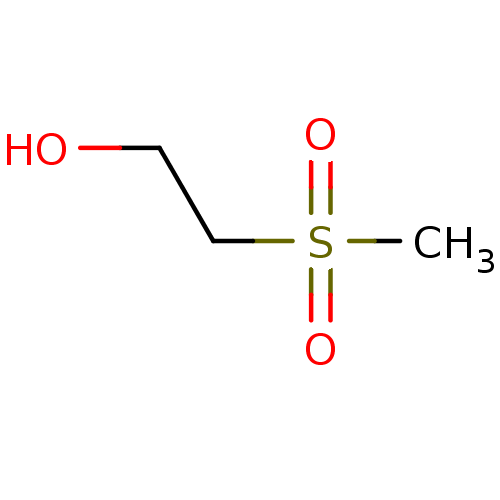
(2-Methanesulfonyl-ethanol | CHEMBL281616)Show InChI InChI=1S/C3H8O3S/c1-7(5,6)3-2-4/h4H,2-3H2,1H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| PubMed
| 8.70E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026470

((2-Hydroxy-ethyl)-dimethyl-sulfonium; iodide | CHE...)Show InChI InChI=1S/C4H11IOS/c1-7(2,5)4-3-6/h6H,3-4H2,1-2H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.30E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(noncompetitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026478
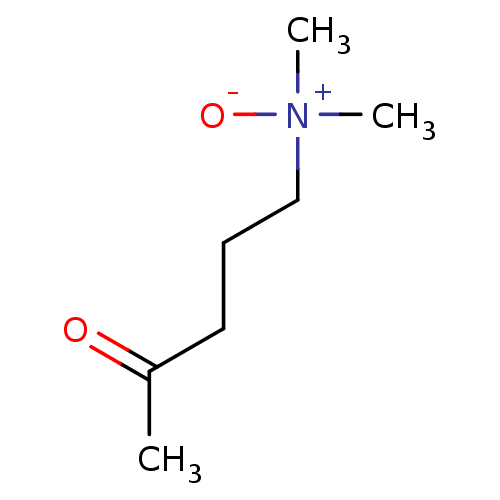
(5-dimethylamino-2-pentanone-N-oxide | CHEMBL25004)Show InChI InChI=1S/C7H15NO2/c1-7(9)5-4-6-8(2,3)10/h4-6H2,1-3H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 1.40E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis of acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026474

(3,3-Dimethyl-butan-1-ol | CHEMBL25029)Show InChI InChI=1S/C6H14O/c1-6(2,3)4-5-7/h7H,4-5H2,1-3H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.90E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(competitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026474

(3,3-Dimethyl-butan-1-ol | CHEMBL25029)Show InChI InChI=1S/C6H14O/c1-6(2,3)4-5-7/h7H,4-5H2,1-3H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.90E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(noncompetitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026472

((CH3)2SO | (methanesulfinyl)methanedimethyl sulfox...) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 2.50E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(com) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026473

(CHEMBL25028 | Sulfonylbismethane | dimethyl sulfon...) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.80E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(noncompetitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026479
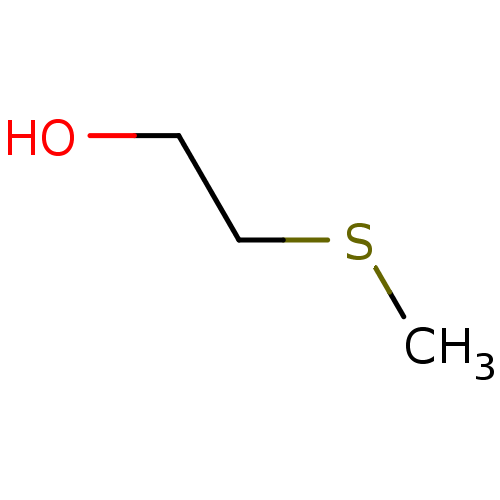
(2-Methylsulfanyl-ethanol | CHEMBL277871)Show InChI InChI=1S/C3H8OS/c1-5-3-2-4/h4H,2-3H2,1H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 4.00E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(com) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM36173
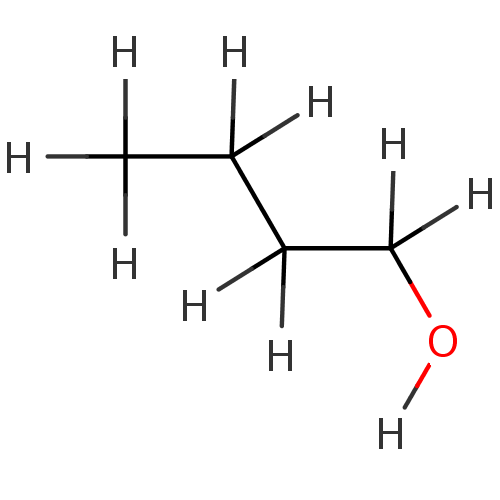
(1-butanol | 1-butanol-d10 | CHEMBL14245)Show InChI InChI=1S/C4H10O/c1-2-3-4-5/h5H,2-4H2,1H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 4.70E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(com) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026477

(2-Methanesulfonyl-ethanol | CHEMBL281616)Show InChI InChI=1S/C3H8O3S/c1-7(5,6)3-2-4/h4H,2-3H2,1H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| PubMed
| 1.00E+8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(com) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM50026473

(CHEMBL25028 | Sulfonylbismethane | dimethyl sulfon...) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.60E+8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for reversible inhibition of hydrolysis acetylcholine by acetylcholinesterase and represented as KI(noncompetitive) |
J Med Chem 28: 1309-13 (1985)
BindingDB Entry DOI: 10.7270/Q26W994C |
More data for this
Ligand-Target Pair | |
Dual specificity mitogen-activated protein kinase kinase 7
(Homo sapiens (Human)) | BDBM50553869

(CHEMBL4764965)Show SMILES C=CC(=O)Nc1cc(cc(c1)-c1n[nH]c2ccccc12)C(=O)NCC#C | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02206
BindingDB Entry DOI: 10.7270/Q2765KB8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM420298
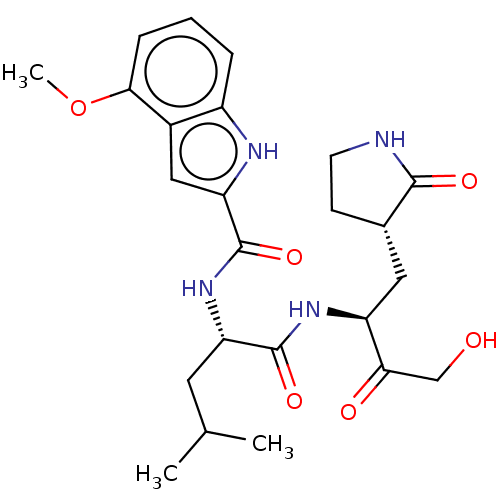
(CVD-0006356 | PF-00835231 | PF-0835231 | US1152494...)Show SMILES COc1cccc2[nH]c(cc12)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C[C@@H]1CCNC1=O)C(=O)CO Show InChI InChI=1S/C24H32N4O6/c1-13(2)9-18(23(32)27-17(20(30)12-29)10-14-7-8-25-22(14)31)28-24(33)19-11-15-16(26-19)5-4-6-21(15)34-3/h4-6,11,13-14,17-18,26,29H,7-10,12H2,1-3H3,(H,25,31)(H,27,32)(H,28,33)/t14-,17-,18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PDB
UniChem
| | n/a | n/a | 0.650 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM50097721
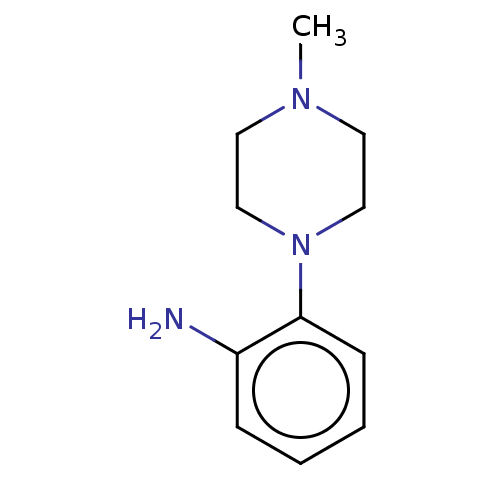
(CHEMBL1879790 | EN300-11843)Show InChI InChI=1S/C29H38FN5O3S/c1-4-6-7-8-9-10-15-33(3)26(36)20-35-19-24(16-23-17-31-28(38)34(5-2)18-23)27(37)32-29(35)39-21-22-11-13-25(30)14-12-22/h11-14,17-19H,4-10,15-16,20-21H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM496902
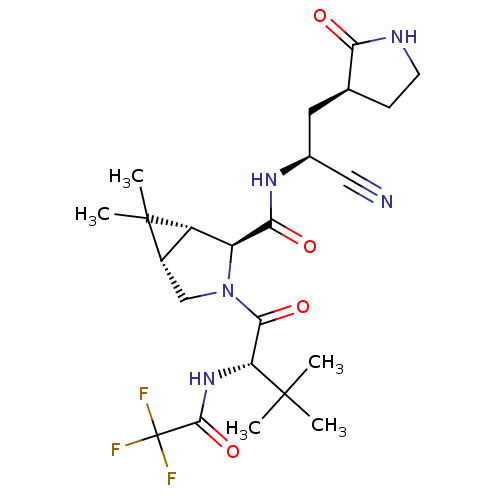
(CVD-0018409 | PF-07321332 | US11351149, Example 13...)Show SMILES CC(C)(C)[C@H](NC(=O)C(F)(F)F)C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)N[C@@H](C[C@@H]1CCNC1=O)C#N)C2(C)C Show InChI InChI=1S/C23H32F3N5O4/c1-21(2,3)16(30-20(35)23(24,25)26)19(34)31-10-13-14(22(13,4)5)15(31)18(33)29-12(9-27)8-11-6-7-28-17(11)32/h11-16H,6-8,10H2,1-5H3,(H,28,32)(H,29,33)(H,30,35)/t11-,12-,13-,14-,15-,16+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Dual specificity mitogen-activated protein kinase kinase 7
(Homo sapiens (Human)) | BDBM50594444

(CHEMBL5174214)Show SMILES Nc1ncnc2n(nc(-c3cn(CC(F)(F)c4ccc(Cl)cc4)nn3)c12)[C@@H]1CCCN(C1)C(=O)C=C |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02206
BindingDB Entry DOI: 10.7270/Q2765KB8 |
More data for this
Ligand-Target Pair | |
Dual specificity mitogen-activated protein kinase kinase 7
(Homo sapiens (Human)) | BDBM50594445

(CHEMBL5195838)Show SMILES Nc1ncnc2n(nc(-c3cn(CC(F)(F)c4ccccc4)nn3)c12)[C@@H]1CCCN(C1)C(=O)C=C |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02206
BindingDB Entry DOI: 10.7270/Q2765KB8 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM496478

(LOR-NOR-30067bb9-11)Show InChI InChI=1S/C16H9BrN2O2/c17-13-6-2-5-12-14(13)19(16(21)15(12)20)9-11-4-1-3-10(7-11)8-18/h1-7H,9H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Israel Institution of Biological Research
| Assay Description
The assay was performed according to the published procedure. Briefly, compounds were seeded into assay-ready plates (Greiner 384PP, cat# 781280) usi... |
bioRxiv 2021: (2021)
BindingDB Entry DOI: 10.7270/Q2MS3WV7 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM627791
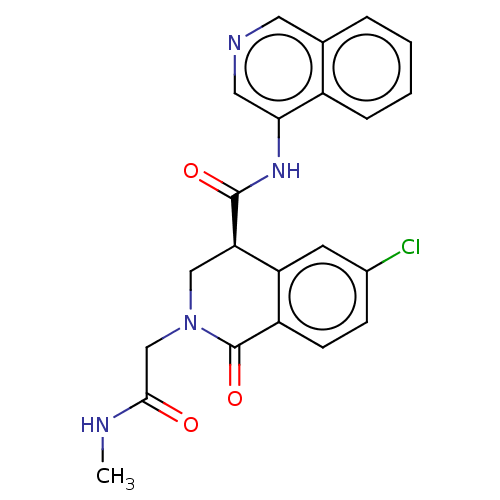
(CVD-0019230)Show SMILES CNC(=O)CN1C[C@@H](C(=O)Nc2cncc3ccccc23)c2cc(Cl)ccc2C1=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495599

(CVD-0016335 | STE-KUL-d79e3d6a-3)Show SMILES CC(C)C[C@@H](NC(=O)[C@@H](NC(=O)CCCC#C)C(C)(C)C)C(=O)NN(CCC(N)=O)C(=O)CCl Show InChI InChI=1S/C23H38ClN5O5/c1-7-8-9-10-18(31)27-20(23(4,5)6)22(34)26-16(13-15(2)3)21(33)28-29(19(32)14-24)12-11-17(25)30/h1,15-16,20H,8-14H2,2-6H3,(H2,25,30)(H,26,34)(H,27,31)(H,28,33)/t16-,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495599

(CVD-0016335 | STE-KUL-d79e3d6a-3)Show SMILES CC(C)C[C@@H](NC(=O)[C@@H](NC(=O)CCCC#C)C(C)(C)C)C(=O)NN(CCC(N)=O)C(=O)CCl Show InChI InChI=1S/C23H38ClN5O5/c1-7-8-9-10-18(31)27-20(23(4,5)6)22(34)26-16(13-15(2)3)21(33)28-29(19(32)14-24)12-11-17(25)30/h1,15-16,20H,8-14H2,2-6H3,(H2,25,30)(H,26,34)(H,27,31)(H,28,33)/t16-,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Israel Institution of Biological Research
| Assay Description
Compounds were seeded into assay-ready plates (Greiner 384 low volume, cat 784900) using an Echo 555 acoustic dispenser, and DMSO was back-filled for... |
bioRxiv 2021: (2021)
BindingDB Entry DOI: 10.7270/Q2MS3WV7 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM496258
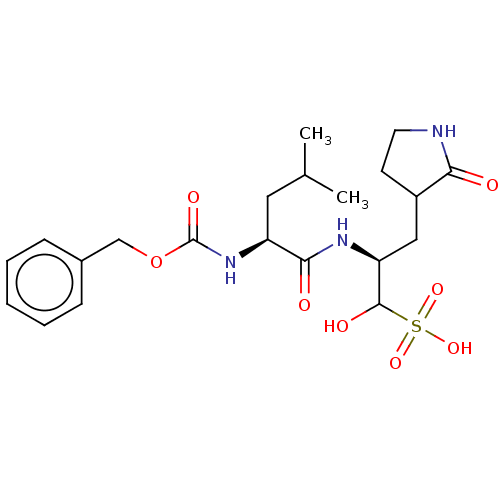
(CVD-0013146 | JOH-MSK-46727e7b-1)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC1CCNC1=O)C(O)S(O)(=O)=O Show InChI InChI=1S/C21H31N3O8S/c1-13(2)10-16(24-21(28)32-12-14-6-4-3-5-7-14)19(26)23-17(20(27)33(29,30)31)11-15-8-9-22-18(15)25/h3-7,13,15-17,20,27H,8-12H2,1-2H3,(H,22,25)(H,23,26)(H,24,28)(H,29,30,31)/t15?,16-,17-,20?/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Israel Institution of Biological Research
| Assay Description
The assay was performed according to the published procedure. Briefly, compounds were seeded into assay-ready plates (Greiner 384PP, cat# 781280) usi... |
bioRxiv 2021: (2021)
BindingDB Entry DOI: 10.7270/Q2MS3WV7 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM627915
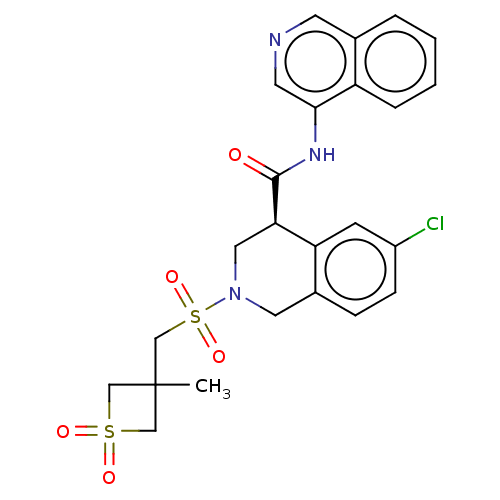
(CVD-0019273)Show SMILES CC1(CS(=O)(=O)N2C[C@@H](C(=O)Nc3cncc4ccccc34)c3cc(Cl)ccc3C2)CS(=O)(=O)C1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM625991
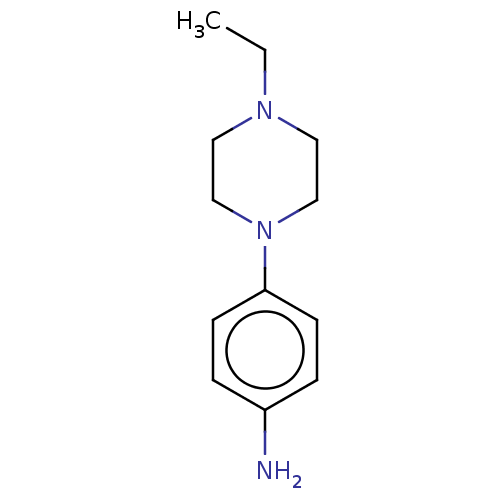
(EN300-11760) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Dual specificity mitogen-activated protein kinase kinase 7
(Homo sapiens (Human)) | BDBM50258966

(CHEMBL4071151)Show SMILES Nc1ncnc2n(nc(C#C)c12)[C@@H]1CCCN(C1)C(=O)C=C |r| Show InChI InChI=1S/C15H16N6O/c1-3-11-13-14(16)17-9-18-15(13)21(19-11)10-6-5-7-20(8-10)12(22)4-2/h1,4,9-10H,2,5-8H2,(H2,16,17,18)/t10-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02206
BindingDB Entry DOI: 10.7270/Q2765KB8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM496258

(CVD-0013146 | JOH-MSK-46727e7b-1)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC1CCNC1=O)C(O)S(O)(=O)=O Show InChI InChI=1S/C21H31N3O8S/c1-13(2)10-16(24-21(28)32-12-14-6-4-3-5-7-14)19(26)23-17(20(27)33(29,30)31)11-15-8-9-22-18(15)25/h3-7,13,15-17,20,27H,8-12H2,1-2H3,(H,22,25)(H,23,26)(H,24,28)(H,29,30,31)/t15?,16-,17-,20?/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Israel Institution of Biological Research
| Assay Description
Compounds were seeded into assay-ready plates (Greiner 384 low volume, cat 784900) using an Echo 555 acoustic dispenser, and DMSO was back-filled for... |
bioRxiv 2021: (2021)
BindingDB Entry DOI: 10.7270/Q2MS3WV7 |
More data for this
Ligand-Target Pair | |
Dual specificity mitogen-activated protein kinase kinase 7
(Homo sapiens (Human)) | BDBM50594446

(CHEMBL5169325)Show SMILES CO[C@@H]1CCCC[C@H]1n1cc(CNC(=O)c2cc(NC(=O)C=C)cc(c2)-c2n[nH]c3ccccc23)nn1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02206
BindingDB Entry DOI: 10.7270/Q2765KB8 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM626053
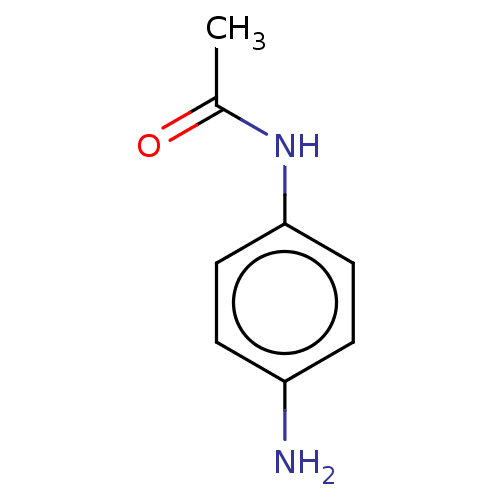
(EN300-17406) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495290
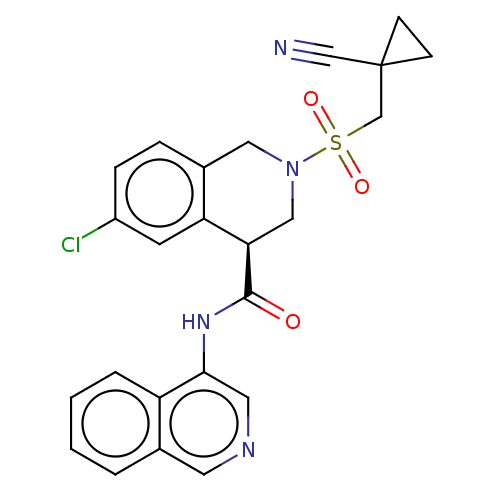
(CVD-0018692 | MAT-POS-e194df51-1)Show SMILES Clc1ccc2CN(C[C@@H](C(=O)Nc3cncc4ccccc34)c2c1)S(=O)(=O)CC1(CC1)C#N Show InChI InChI=1S/C24H21ClN4O3S/c25-18-6-5-17-12-29(33(31,32)15-24(14-26)7-8-24)13-21(20(17)9-18)23(30)28-22-11-27-10-16-3-1-2-4-19(16)22/h1-6,9-11,21H,7-8,12-13,15H2,(H,28,30)/t21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| MCE
PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495290

(CVD-0018692 | MAT-POS-e194df51-1)Show SMILES Clc1ccc2CN(C[C@@H](C(=O)Nc3cncc4ccccc34)c2c1)S(=O)(=O)CC1(CC1)C#N Show InChI InChI=1S/C24H21ClN4O3S/c25-18-6-5-17-12-29(33(31,32)15-24(14-26)7-8-24)13-21(20(17)9-18)23(30)28-22-11-27-10-16-3-1-2-4-19(16)22/h1-6,9-11,21H,7-8,12-13,15H2,(H,28,30)/t21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| MCE
PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Israel Institution of Biological Research
| Assay Description
Compounds were seeded into assay-ready plates (Greiner 384 low volume, cat 784900) using an Echo 555 acoustic dispenser, and DMSO was back-filled for... |
bioRxiv 2021: (2021)
BindingDB Entry DOI: 10.7270/Q2MS3WV7 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM625991

(EN300-11760) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM627860
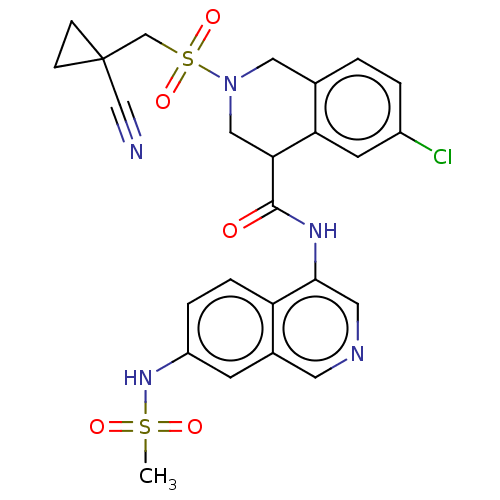
(CVD-0018385 | EDJ-MED-be9e6f63-3)Show SMILES CS(=O)(=O)Nc1ccc2c(NC(=O)C3CN(Cc4ccc(Cl)cc34)S(=O)(=O)CC3(CC3)C#N)cncc2c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM627860

(CVD-0018385 | EDJ-MED-be9e6f63-3)Show SMILES CS(=O)(=O)Nc1ccc2c(NC(=O)C3CN(Cc4ccc(Cl)cc34)S(=O)(=O)CC3(CC3)C#N)cncc2c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495803

(CVD-0015332 | MAT-POS-e69ad64a-2)Show SMILES Clc1ccc2OCC(C(=O)N(C(=O)C=C)c3cncc4ccccc34)c2c1 Show InChI InChI=1S/C21H15ClN2O3/c1-2-20(25)24(18-11-23-10-13-5-3-4-6-15(13)18)21(26)17-12-27-19-8-7-14(22)9-16(17)19/h2-11,17H,1,12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Israel Institution of Biological Research
| Assay Description
Compounds were seeded into assay-ready plates (Greiner 384 low volume, cat 784900) using an Echo 555 acoustic dispenser, and DMSO was back-filled for... |
bioRxiv 2021: (2021)
BindingDB Entry DOI: 10.7270/Q2MS3WV7 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495803

(CVD-0015332 | MAT-POS-e69ad64a-2)Show SMILES Clc1ccc2OCC(C(=O)N(C(=O)C=C)c3cncc4ccccc34)c2c1 Show InChI InChI=1S/C21H15ClN2O3/c1-2-20(25)24(18-11-23-10-13-5-3-4-6-15(13)18)21(26)17-12-27-19-8-7-14(22)9-16(17)19/h2-11,17H,1,12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 42 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM627972
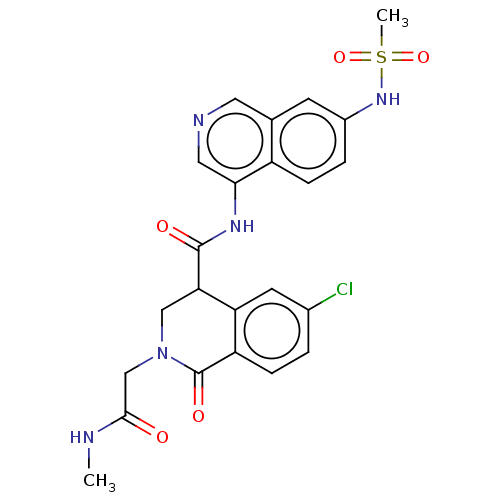
(CVD-0019478)Show SMILES CNC(=O)CN1CC(C(=O)Nc2cncc3cc(NS(C)(=O)=O)ccc23)c2cc(Cl)ccc2C1=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495598
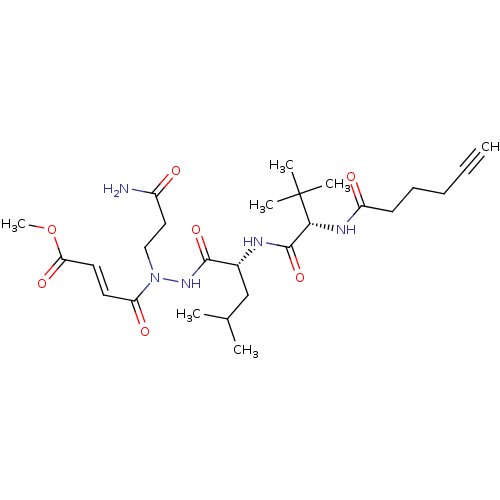
(CVD-0016334 | STE-KUL-d79e3d6a-2)Show SMILES COC(=O)\C=C\C(=O)N(CCC(N)=O)NC(=O)[C@@H](CC(C)C)NC(=O)[C@@H](NC(=O)CCCC#C)C(C)(C)C Show InChI InChI=1S/C26H41N5O7/c1-8-9-10-11-20(33)29-23(26(4,5)6)25(37)28-18(16-17(2)3)24(36)30-31(15-14-19(27)32)21(34)12-13-22(35)38-7/h1,12-13,17-18,23H,9-11,14-16H2,2-7H3,(H2,27,32)(H,28,37)(H,29,33)(H,30,36)/b13-12+/t18-,23-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
Israel Institution of Biological Research
| Assay Description
Compounds were seeded into assay-ready plates (Greiner 384 low volume, cat 784900) using an Echo 555 acoustic dispenser, and DMSO was back-filled for... |
bioRxiv 2021: (2021)
BindingDB Entry DOI: 10.7270/Q2MS3WV7 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495598

(CVD-0016334 | STE-KUL-d79e3d6a-2)Show SMILES COC(=O)\C=C\C(=O)N(CCC(N)=O)NC(=O)[C@@H](CC(C)C)NC(=O)[C@@H](NC(=O)CCCC#C)C(C)(C)C Show InChI InChI=1S/C26H41N5O7/c1-8-9-10-11-20(33)29-23(26(4,5)6)25(37)28-18(16-17(2)3)24(36)30-31(15-14-19(27)32)21(34)12-13-22(35)38-7/h1,12-13,17-18,23H,9-11,14-16H2,2-7H3,(H2,27,32)(H,28,37)(H,29,33)(H,30,36)/b13-12+/t18-,23-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM626189

(EN300-60157) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| | n/a | n/a | 45 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495783

(MAT-POS-f9802937-8)Show SMILES Clc1cc2NCC[C@@H](C(=O)Nc3cncc4ccccc34)c2cc1Cl Show InChI InChI=1S/C19H15Cl2N3O/c20-15-7-14-13(5-6-23-17(14)8-16(15)21)19(25)24-18-10-22-9-11-3-1-2-4-12(11)18/h1-4,7-10,13,23H,5-6H2,(H,24,25)/t13-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| | n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM495783

(MAT-POS-f9802937-8)Show SMILES Clc1cc2NCC[C@@H](C(=O)Nc3cncc4ccccc34)c2cc1Cl Show InChI InChI=1S/C19H15Cl2N3O/c20-15-7-14-13(5-6-23-17(14)8-16(15)21)19(25)24-18-10-22-9-11-3-1-2-4-12(11)18/h1-4,7-10,13,23H,5-6H2,(H,24,25)/t13-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| | n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data