

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50059414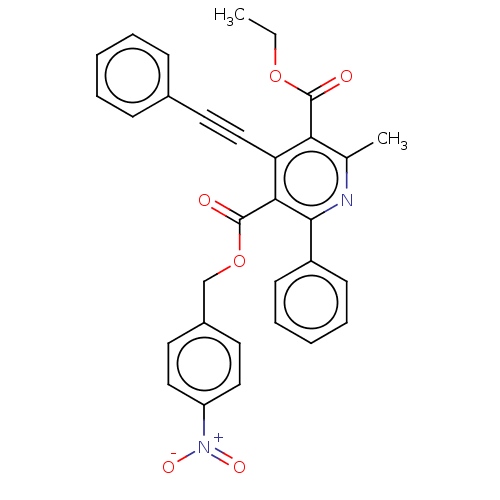 (2-Methyl-6-phenyl-4-phenylethynyl-1,4-dihydro-pyri...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Binding affinity at cloned human adenosine A3 receptor expressed in HEK-293 cells was determined using [125I]-AB-MECA as radioligand | J Med Chem 41: 2835-45 (1998) Article DOI: 10.1021/jm980094b BindingDB Entry DOI: 10.7270/Q25Q4V7D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM50214974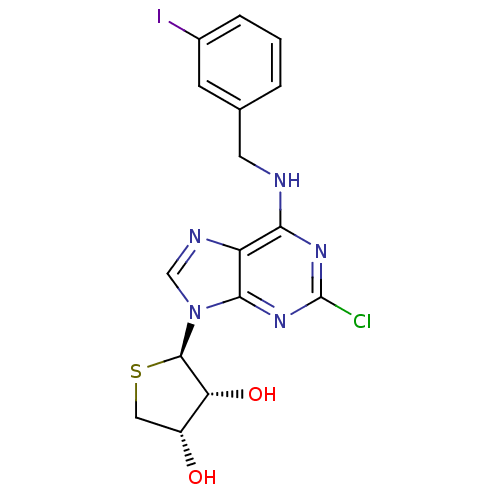 ((2R,3R,4S)-2-(2-chloro-6-(3-iodobenzylamino)-9H-pu...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.00483 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University Curated by ChEMBL | Assay Description Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay | J Med Chem 60: 7459-7475 (2017) Article DOI: 10.1021/acs.jmedchem.7b00805 BindingDB Entry DOI: 10.7270/Q2XK8HQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50117108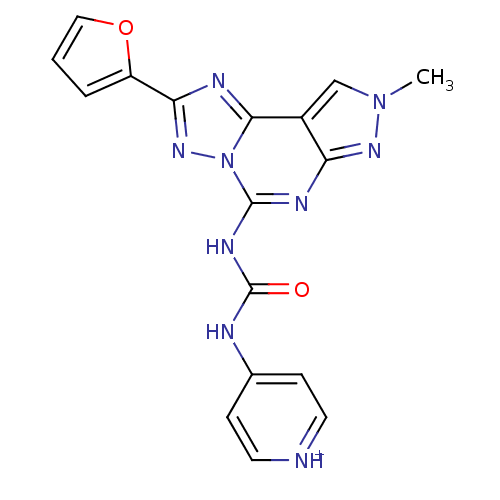 (4-[3-(2-Furan-2-yl-8-methyl-8H-pyrazolo[4,3-e][1,2...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Displacement of [3H]-MRE 3008-F20 from Human Adenosine A3 receptor expressed in HEK-293 cells | J Med Chem 45: 3579-82 (2002) BindingDB Entry DOI: 10.7270/Q2SN08B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM50214981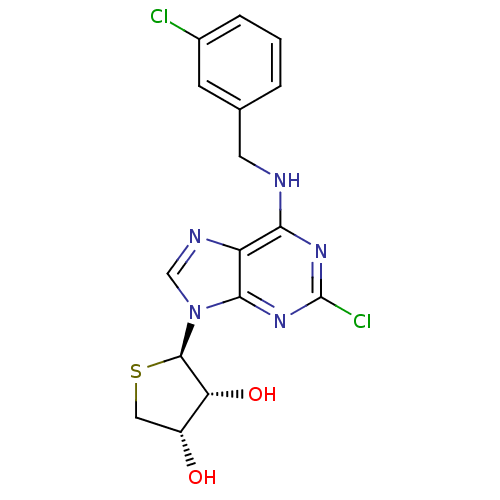 ((2R,3R,4S)-2-(2-chloro-6-(3-chlorobenzylamino)-9H-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0102 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University Curated by ChEMBL | Assay Description Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay | J Med Chem 60: 7459-7475 (2017) Article DOI: 10.1021/acs.jmedchem.7b00805 BindingDB Entry DOI: 10.7270/Q2XK8HQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM85776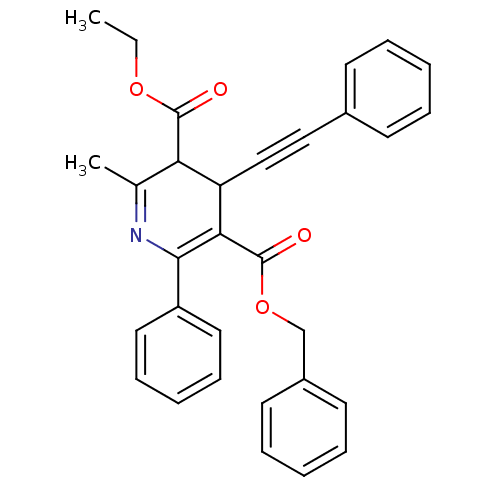 (CAS_393594 | CHEMBL89852 | MRS1191 | NSC_393594) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Binding affinity at cloned human adenosine A3 receptor expressed in HEK-293 cells was determined using [125I]-AB-MECA as radioligand | J Med Chem 41: 2835-45 (1998) Article DOI: 10.1021/jm980094b BindingDB Entry DOI: 10.7270/Q25Q4V7D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50117109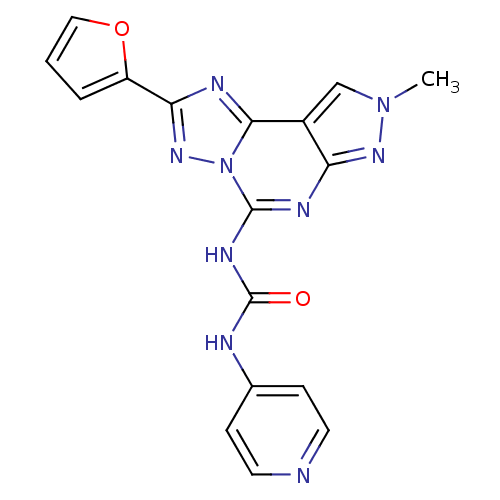 (1-(2-Furan-2-yl-8-methyl-8H-pyrazolo[4,3-e][1,2,4]...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Displacement of [3H]-MRE 3008-F20 from Human Adenosine A3 receptor expressed in HEK-293 cells | J Med Chem 45: 3579-82 (2002) BindingDB Entry DOI: 10.7270/Q2SN08B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM28661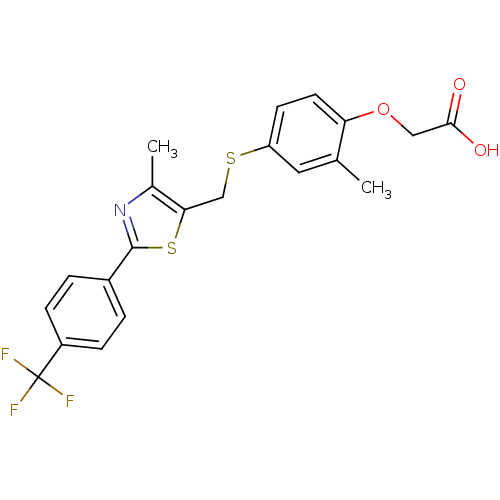 (2-{2-methyl-4-[({4-methyl-2-[4-(trifluoromethyl)ph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.0480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University Curated by ChEMBL | Assay Description Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay | J Med Chem 60: 7459-7475 (2017) Article DOI: 10.1021/acs.jmedchem.7b00805 BindingDB Entry DOI: 10.7270/Q2XK8HQH | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50085048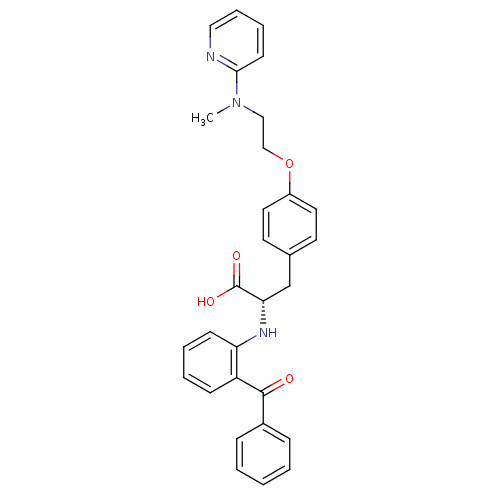 ((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(methyl-pyri...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.0497 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University Curated by ChEMBL | Assay Description Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARgamma LBD by TR-FRET assay | J Med Chem 60: 7459-7475 (2017) Article DOI: 10.1021/acs.jmedchem.7b00805 BindingDB Entry DOI: 10.7270/Q2XK8HQH | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50099491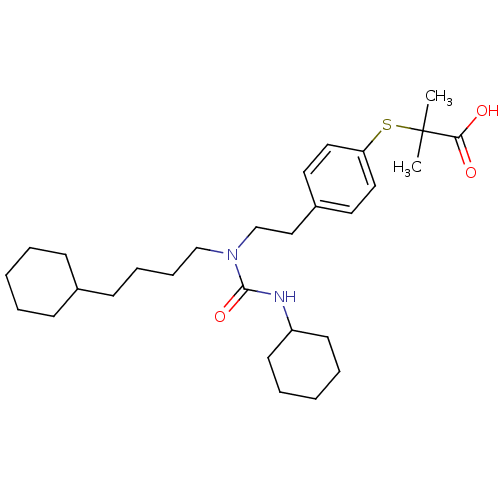 (2-(4-(2-(3-cyclohexyl-1-(4-cyclohexylbutyl)ureido)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.0541 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University Curated by ChEMBL | Assay Description Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARalpha LBD by TR-FRET assay | J Med Chem 60: 7459-7475 (2017) Article DOI: 10.1021/acs.jmedchem.7b00805 BindingDB Entry DOI: 10.7270/Q2XK8HQH | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| P2Y purinoceptor 14 (Homo sapiens (Human)) | BDBM50512416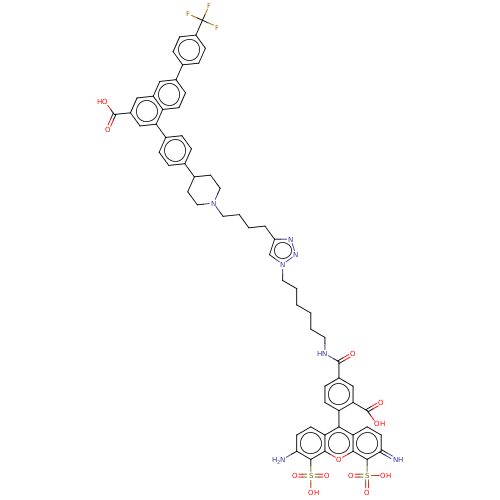 (CHEMBL4447162) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Antagonist activity at human P2Y14R expressed in CHO cells assessed as inhibition of UDPG-mediated reduction of forskolin-induced [3H]cAMP production... | J Med Chem 59: 6149-68 (2016) Article DOI: 10.1021/acs.jmedchem.6b00044 BindingDB Entry DOI: 10.7270/Q2DJ5K34 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2Y purinoceptor 14 (Homo sapiens (Human)) | BDBM50512416 (CHEMBL4447162) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Antagonist activity at human P2Y14R expressed in CHO cells assessed as inhibition of UDPG-mediated reduction of forskolin-induced [3H]cAMP production... | J Med Chem 59: 6149-68 (2016) Article DOI: 10.1021/acs.jmedchem.6b00044 BindingDB Entry DOI: 10.7270/Q2DJ5K34 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2Y purinoceptor 14 (Homo sapiens (Human)) | BDBM50512416 (CHEMBL4447162) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Inhibition of human P2Y14R | J Med Chem 63: 9563-9589 (2020) Article DOI: 10.1021/acs.jmedchem.0c00745 BindingDB Entry DOI: 10.7270/Q20R9SZP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Mus musculus) | BDBM50118812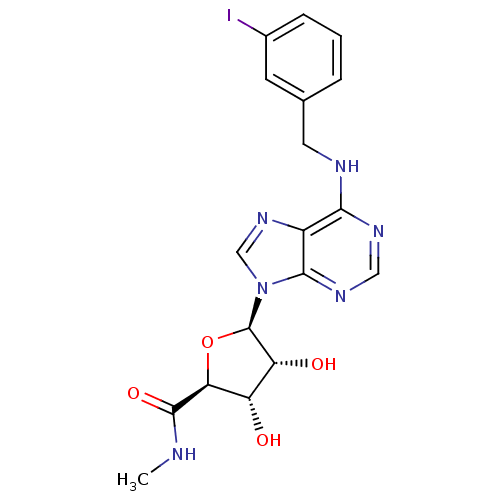 ((2S,3S,4R,5R)-3,4-Dihydroxy-5-[6-(3-iodo-benzylami...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.0870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of [125I]N6-(-amino-3-iodobenzyl)adenosine-5'-N-methyl-uronamide from mouse recombinant adenosine A3 receptor expressed in HEK293 cells | Bioorg Med Chem Lett 18: 2813-9 (2008) Article DOI: 10.1016/j.bmcl.2008.04.001 BindingDB Entry DOI: 10.7270/Q2XK8GFH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (BOVINE) | BDBM50008408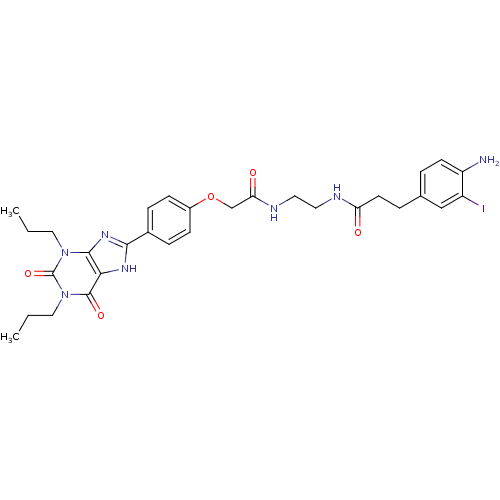 (3-(4-Amino-3-iodo-phenyl)-N-(2-{2-[4-(2,6-dioxo-1,...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Binding affinity for adenosine A1 receptor using [3H]- CHA or [3H]- PIA in bovine brain cortical membranes | J Med Chem 35: 407-22 (1992) BindingDB Entry DOI: 10.7270/Q2Z89D1W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50053927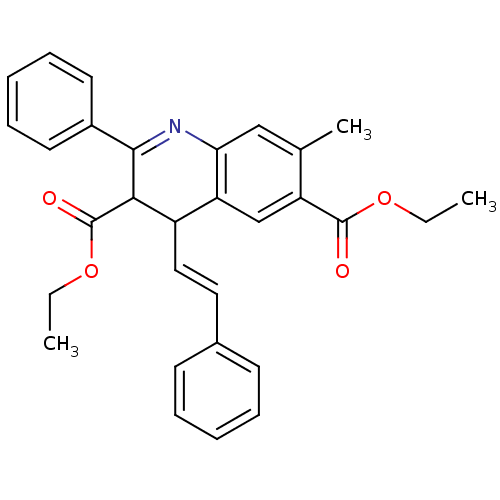 (7-Methyl-2-phenyl-4-((E)-styryl)-1,4-dihydro-quino...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description In vitro binding affinity at human Adenosine A3 receptor from HEK-293 cells by [125I]-AB-MECA displacement. | J Med Chem 39: 4142-8 (1996) Article DOI: 10.1021/jm960482i BindingDB Entry DOI: 10.7270/Q2FQ9VQ2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50004566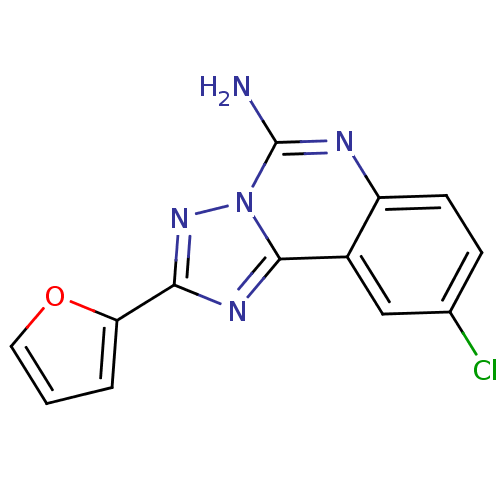 (9-Chloro-2-furan-2-yl-[1,2,4]triazolo[1,5-c]quinaz...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Binding affinity for HA-tagged wild type human Adenosine A2A receptor (WT) using [3H]CGS-21680 as radioligand expressed in COS-7 cells | J Med Chem 40: 2588-95 (1997) Article DOI: 10.1021/jm970084v BindingDB Entry DOI: 10.7270/Q27M08MC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Mus musculus) | BDBM50517301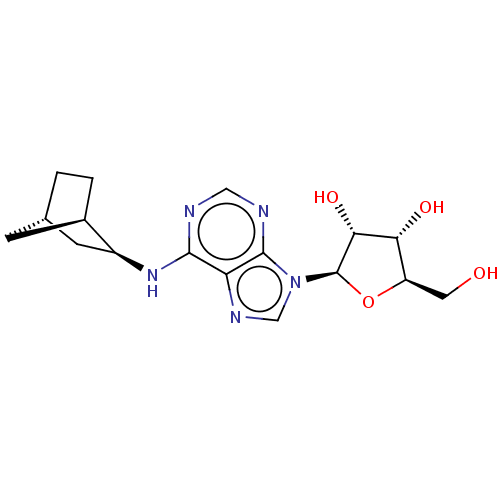 (CHEMBL1877326) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Wisconsin Curated by ChEMBL | Assay Description Displacement of [3H]N6-R-phenylisopropyladenosine from mouse A1A adenosine receptor expressed in CHO cell membranes after 60 mins by scintillation pr... | J Med Chem 62: 1502-1522 (2019) Article DOI: 10.1021/acs.jmedchem.8b01662 BindingDB Entry DOI: 10.7270/Q2TM7FGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50109461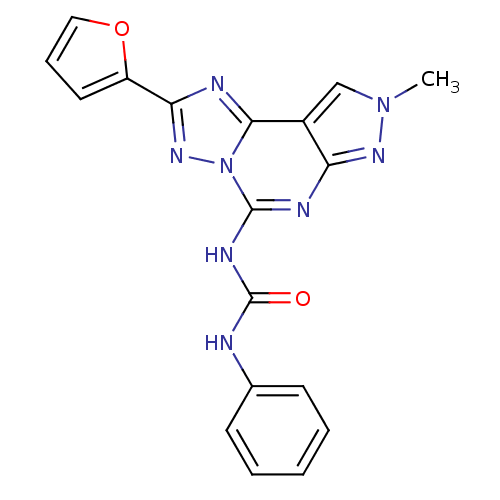 (1-(2-(furan-2-yl)-8-methyl-8H-pyrazolo[4,3-e][1,2,...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Displacement of [3H]-MRE 3008-F20 from Human Adenosine A3 receptor expressed in HEK-293 cells | J Med Chem 45: 3579-82 (2002) BindingDB Entry DOI: 10.7270/Q2SN08B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM50180197 ((2S,3S,4R,5R)-5-(2-chloro-6-(3-iodobenzylamino)-9H...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University Curated by ChEMBL | Assay Description Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay | J Med Chem 60: 7459-7475 (2017) Article DOI: 10.1021/acs.jmedchem.7b00805 BindingDB Entry DOI: 10.7270/Q2XK8HQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50304633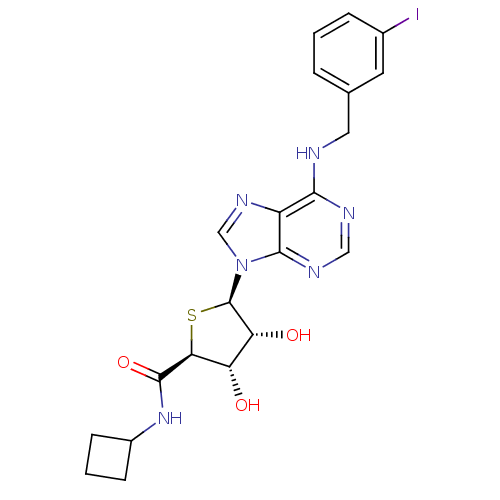 (9-(5'-Cyclobutylaminocarbonyl-4'-thio-beta-D-ribof...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University Curated by ChEMBL | Assay Description Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARgamma LBD by TR-FRET assay | J Med Chem 60: 7459-7475 (2017) Article DOI: 10.1021/acs.jmedchem.7b00805 BindingDB Entry DOI: 10.7270/Q2XK8HQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Mus musculus) | BDBM21221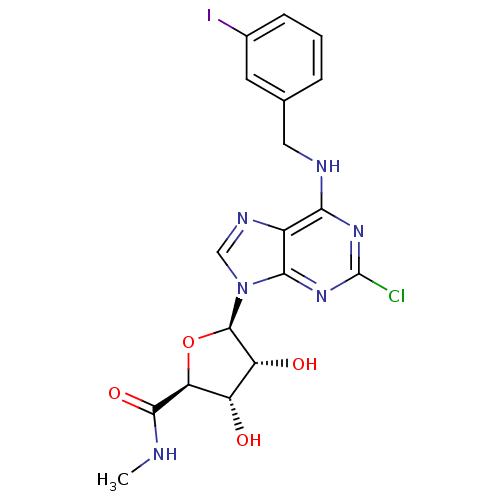 ((2S,3S,4R,5R)-5-(2-chloro-6-{[(3-iodophenyl)methyl...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of [125I]N6-(-amino-3-iodobenzyl)adenosine-5'-N-methyl-uronamide from mouse recombinant adenosine A3 receptor expressed in HEK293 cells | Bioorg Med Chem Lett 18: 2813-9 (2008) Article DOI: 10.1016/j.bmcl.2008.04.001 BindingDB Entry DOI: 10.7270/Q2XK8GFH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50106541 (4-Amino-2-(6-cyclopentylamino-purin-9-yl)-5-hydrox...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity at Mutant (H272E) human adenosine A3 receptor expressed in COS-7 cells | J Med Chem 44: 4125-36 (2001) BindingDB Entry DOI: 10.7270/Q2Z60PSZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a/A2b (Homo sapiens (Human)) | BDBM50008416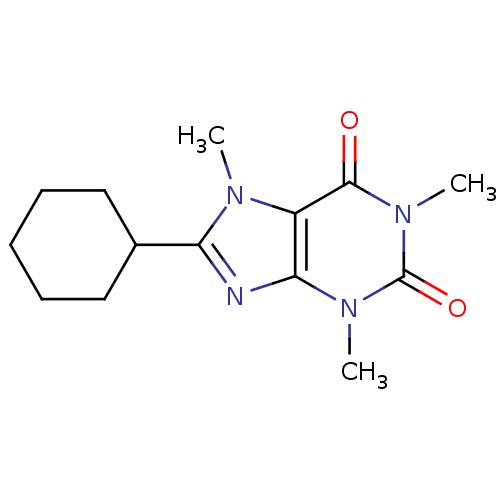 (8-Cyclohexyl-1,3,7-trimethyl-3,7-dihydro-purine-2,...) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Binding affinity for adenosine A2 receptor using [3H]- NECA antagonism of adenylate cyclase activation in human platelets | J Med Chem 35: 407-22 (1992) BindingDB Entry DOI: 10.7270/Q2Z89D1W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50094691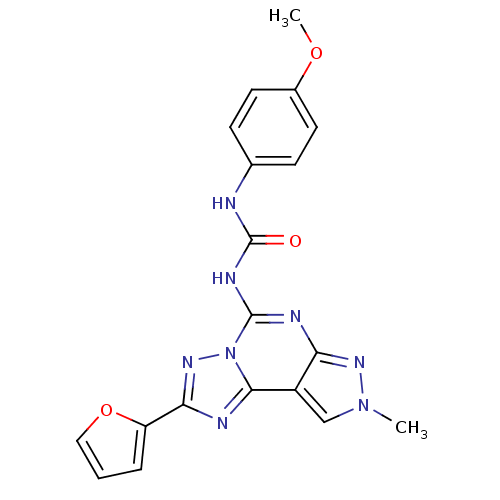 (1-(2-(furan-2-yl)-8-methyl-8H-pyrazolo[4,3-e][1,2,...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Università degli Studi di Ferrara Curated by ChEMBL | Assay Description Displacement of [3H]-MRE3008-F20 from human Adenosine A3 receptor expressed in CHO cells | J Med Chem 44: 2735-42 (2001) BindingDB Entry DOI: 10.7270/Q2WQ033H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Mus musculus) | BDBM50517302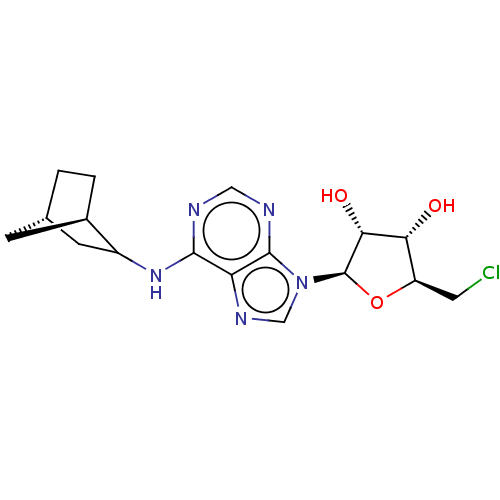 (CHEMBL4562258) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Wisconsin Curated by ChEMBL | Assay Description Displacement of [3H]N6-R-phenylisopropyladenosine from mouse A1A adenosine receptor expressed in CHO cell membranes after 60 mins by scintillation pr... | J Med Chem 62: 1502-1522 (2019) Article DOI: 10.1021/acs.jmedchem.8b01662 BindingDB Entry DOI: 10.7270/Q2TM7FGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Mus musculus) | BDBM50085658 ((2R,3R,4S,5R)-2-(2-Chloro-6-cyclopentylamino-purin...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Wisconsin Curated by ChEMBL | Assay Description Displacement of [3H]N6-R-phenylisopropyladenosine from mouse A1A adenosine receptor expressed in CHO cell membranes after 60 mins by scintillation pr... | J Med Chem 62: 1502-1522 (2019) Article DOI: 10.1021/acs.jmedchem.8b01662 BindingDB Entry DOI: 10.7270/Q2TM7FGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Mus musculus) | BDBM25400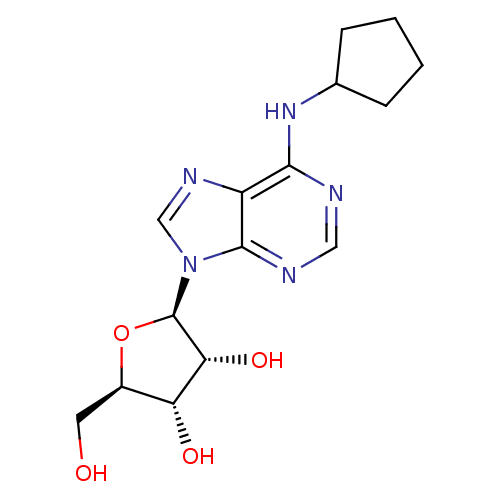 ((2R,3R,4S,5R)-2-[6-(cyclopentylamino)-9H-purin-9-y...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Wisconsin Curated by ChEMBL | Assay Description Displacement of [3H]N6-R-phenylisopropyladenosine from mouse A1A adenosine receptor expressed in CHO cell membranes after 60 mins by scintillation pr... | J Med Chem 62: 1502-1522 (2019) Article DOI: 10.1021/acs.jmedchem.8b01662 BindingDB Entry DOI: 10.7270/Q2TM7FGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50108018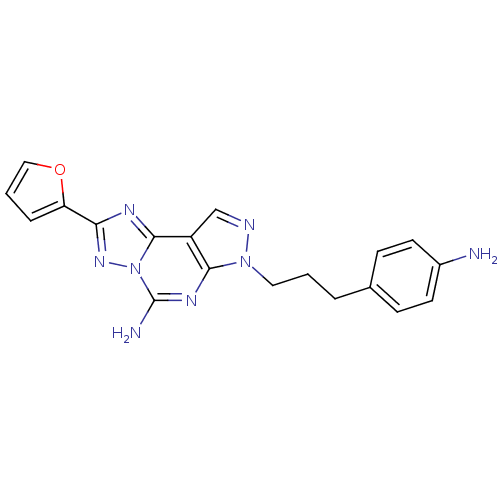 (7-(3-(4-aminophenyl)propyl)-2-(furan-2-yl)-7H-pyra...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Binding affinity against WT human adenosine A2A receptor expressed in CHO cells using [3H]- ZM-241385 | J Med Chem 46: 4847-59 (2003) Article DOI: 10.1021/jm0300431 BindingDB Entry DOI: 10.7270/Q24M9584 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50368538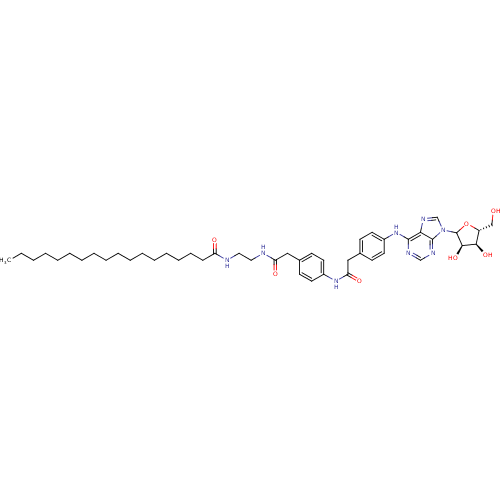 (CHEMBL610439) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Binding affinity for Adenosine A1 receptor using [3H]- CHA or [3H]- PIA | J Med Chem 35: 407-22 (1992) BindingDB Entry DOI: 10.7270/Q2Z89D1W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50008388 (3-[2-(4-Amino-3-iodo-phenyl)-ethyl]-8-cyclopentyl-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Binding affinity against adenosine A1 receptor using [3H]-CHA or [3H]PIA as radioligand | J Med Chem 35: 407-22 (1992) BindingDB Entry DOI: 10.7270/Q2Z89D1W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50252829 ((2S,3S,4R,5R)-5-(6-(3-iodobenzylamino)-9H-purin-9-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University Curated by ChEMBL | Assay Description Displacement of [125I]I-AB-MECA from human adenosine A3 receptor expressed in CHO cells after 60 mins by gamma counting analysis | J Med Chem 60: 3422-3437 (2017) Article DOI: 10.1021/acs.jmedchem.7b00241 BindingDB Entry DOI: 10.7270/Q2J105FT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50252829 ((2S,3S,4R,5R)-5-(6-(3-iodobenzylamino)-9H-purin-9-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ewha Womans University Curated by ChEMBL | Assay Description Displacement of [125I]AB-MECA from human adenosine A3 receptor expressed in CHO cells | Bioorg Med Chem 17: 8003-11 (2009) Article DOI: 10.1016/j.bmc.2009.10.011 BindingDB Entry DOI: 10.7270/Q29K4B9Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50252829 ((2S,3S,4R,5R)-5-(6-(3-iodobenzylamino)-9H-purin-9-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ewha Womans University Curated by ChEMBL | Assay Description Displacement of [125I]I-AB-MECA from human adenosine A3 receptor expressed in CHO cells | J Med Chem 51: 6609-13 (2008) Article DOI: 10.1021/jm8008647 BindingDB Entry DOI: 10.7270/Q2XG9QZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50561600 (CHEMBL4788488) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [125I]N6-(4-amino-3-iodobenzyl)adenosine-5'-N-methyluronamide from human adrenergic A3 receptor expressed in CHO cell membranes asses... | Citation and Details Article DOI: 10.1021/acsmedchemlett.9b00637 BindingDB Entry DOI: 10.7270/Q20C50H1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50180206 ((2S,3S,4R,5R)-5-(2-chloro-6-(methylamino)-9H-purin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ewha Womans University Curated by ChEMBL | Assay Description Displacement of [125I]AB-MECA from human adenosine A3 receptor transfected in CHO cells | J Med Chem 49: 273-81 (2006) Article DOI: 10.1021/jm050595e BindingDB Entry DOI: 10.7270/Q2736QG2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50180206 ((2S,3S,4R,5R)-5-(2-chloro-6-(methylamino)-9H-purin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ewha Womans University Curated by ChEMBL | Assay Description Binding affinity to human adenosine A3 receptor | Bioorg Med Chem 17: 8003-11 (2009) Article DOI: 10.1016/j.bmc.2009.10.011 BindingDB Entry DOI: 10.7270/Q29K4B9Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50180206 ((2S,3S,4R,5R)-5-(2-chloro-6-(methylamino)-9H-purin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ewha Womans University Curated by ChEMBL | Assay Description Displacement of [125I]AB-MECA from human adenosine A3 receptor expressed in CHO cells | Bioorg Med Chem 17: 8003-11 (2009) Article DOI: 10.1016/j.bmc.2009.10.011 BindingDB Entry DOI: 10.7270/Q29K4B9Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50163020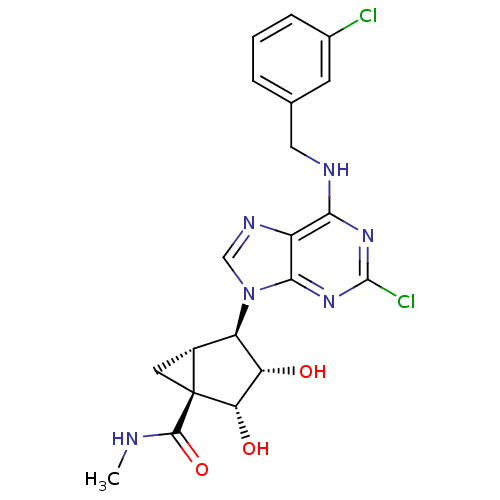 ((1S,2R,3S,4R,5S)-4-(6-(3-chlorobenzylamino)-2-chlo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of [3H]I-AB-MECA from human recombinant adenosine A3 receptor expressed in CHO cells after 60 mins by gamma counting | J Med Chem 55: 4847-60 (2012) Article DOI: 10.1021/jm300396n BindingDB Entry DOI: 10.7270/Q2765GCG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50163020 ((1S,2R,3S,4R,5S)-4-(6-(3-chlorobenzylamino)-2-chlo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of [3H]CGS21680 form human adenosine A3 receptor expressed in CHO cells | Bioorg Med Chem 16: 8546-56 (2008) Article DOI: 10.1016/j.bmc.2008.08.007 BindingDB Entry DOI: 10.7270/Q20R9P71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50163020 ((1S,2R,3S,4R,5S)-4-(6-(3-chlorobenzylamino)-2-chlo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Inhibition of [125I]-AB-MECA binding to human Adenosine A3 receptor expressed in CHO cells | J Med Chem 48: 1745-58 (2005) Article DOI: 10.1021/jm049580r BindingDB Entry DOI: 10.7270/Q23F4P52 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50163020 ((1S,2R,3S,4R,5S)-4-(6-(3-chlorobenzylamino)-2-chlo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of radioligand from human adenosine A3 receptor expressed in CHO cells | Bioorg Med Chem Lett 18: 2813-9 (2008) Article DOI: 10.1016/j.bmcl.2008.04.001 BindingDB Entry DOI: 10.7270/Q2XK8GFH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50300289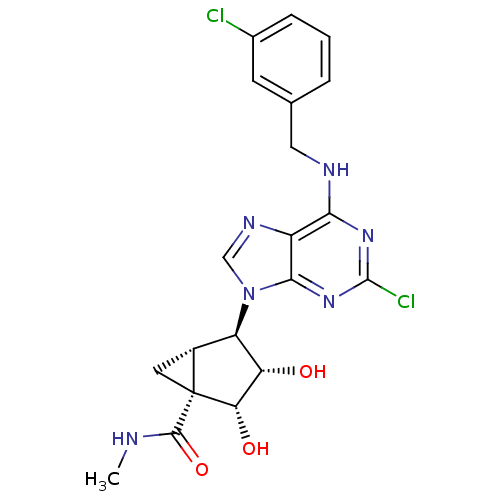 ((1R,2R,3S,4R,5S)-4-(2-chloro-6-(3-chlorobenzylamin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of [125I]AB-MECA from human recombinant adenosine A3 receptor expressed in CHO cells after 60 mins by liquid scintillation counting | J Med Chem 52: 7580-92 (2009) Article DOI: 10.1021/jm900426g BindingDB Entry DOI: 10.7270/Q2ZC82WM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50252829 ((2S,3S,4R,5R)-5-(6-(3-iodobenzylamino)-9H-purin-9-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University Curated by ChEMBL | Assay Description Displacement of [125I]I-AB-MECA from human A3 adenosine receptor expressed in CHO cell membranes after 60 mins by gamma counting method | J Med Chem 60: 7459-7475 (2017) Article DOI: 10.1021/acs.jmedchem.7b00805 BindingDB Entry DOI: 10.7270/Q2XK8HQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50452575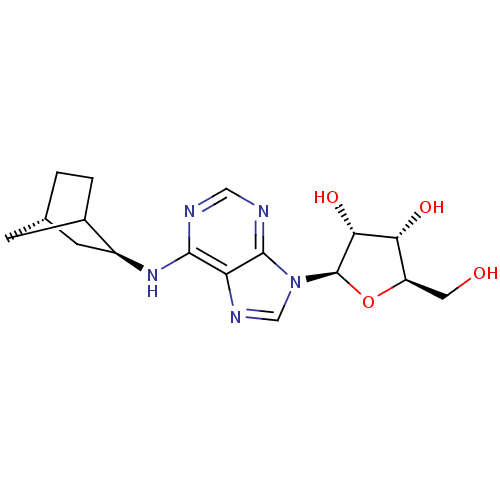 (CHEMBL2112182) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Binding affinity against adenosine A1 receptor using [3H]-CHA or [3H]PIA as radioligand | J Med Chem 35: 407-22 (1992) BindingDB Entry DOI: 10.7270/Q2Z89D1W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (BOVINE) | BDBM50207816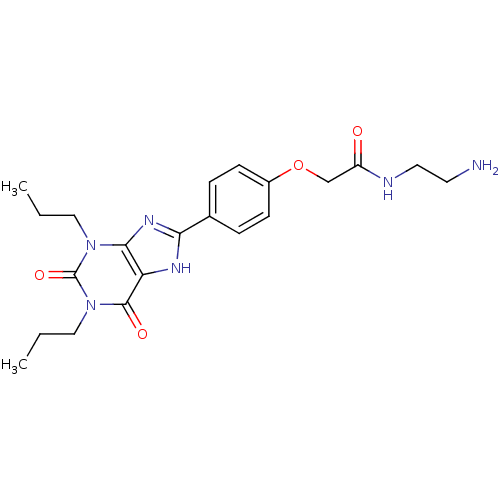 (CHEMBL273094 | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Affinity for Adenosine A1 receptor using [N6-[(3-[125I]-iodo-4-aminophenyl)ethyl]adenosine in calf brain | J Med Chem 32: 1043-51 (1989) BindingDB Entry DOI: 10.7270/Q2WQ04CH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50266666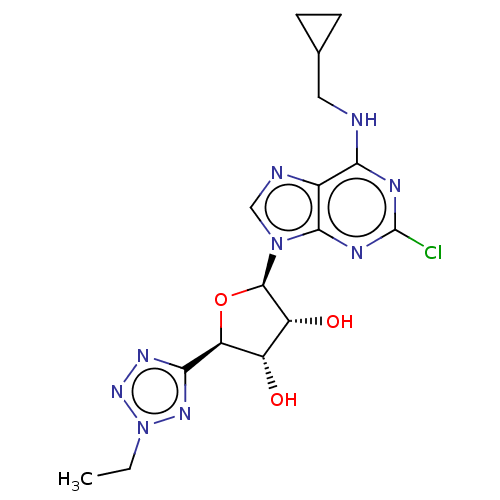 (CHEMBL4105164) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 0.313 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Camerino Curated by ChEMBL | Assay Description Displacement of [3H]HEMADO from recombinant human adenosine A3A receptor expressed in CHO cell membranes after 3 hrs by microbeta scintillation count... | J Med Chem 60: 4327-4341 (2017) Article DOI: 10.1021/acs.jmedchem.7b00291 BindingDB Entry DOI: 10.7270/Q2CJ8GZX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50094687 (1-(8-Butyl-2-furan-2-yl-8H-pyrazolo[4,3-e][1,2,4]t...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Università degli Studi di Ferrara Curated by ChEMBL | Assay Description Displacement of [3H]-MRE3008-F20 from human Adenosine A3 receptor expressed in CHO cells | J Med Chem 44: 2735-42 (2001) BindingDB Entry DOI: 10.7270/Q2WQ033H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Rattus norvegicus) | BDBM21221 ((2S,3S,4R,5R)-5-(2-chloro-6-{[(3-iodophenyl)methyl...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of radioligand from rat A3 adenosine receptor | Bioorg Med Chem Lett 18: 2813-9 (2008) Article DOI: 10.1016/j.bmcl.2008.04.001 BindingDB Entry DOI: 10.7270/Q2XK8GFH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Rattus norvegicus) | BDBM21221 ((2S,3S,4R,5R)-5-(2-chloro-6-{[(3-iodophenyl)methyl...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ewha Womans University Curated by ChEMBL | Assay Description Displacement of [125I]I-AB-MECA from rat adenosine A3 receptor expressed in CHO cells | J Med Chem 51: 6609-13 (2008) Article DOI: 10.1021/jm8008647 BindingDB Entry DOI: 10.7270/Q2XG9QZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Rattus norvegicus) | BDBM50369386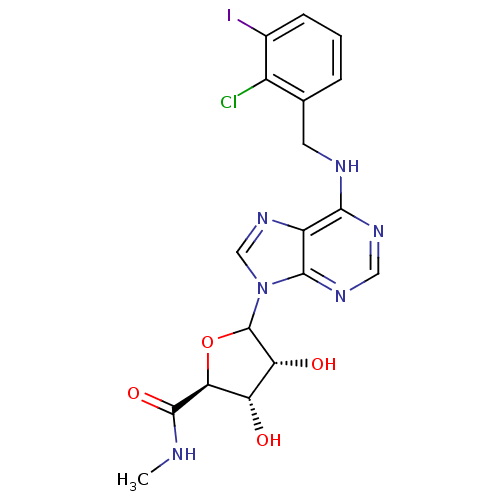 (CHEMBL608327) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Ferrara Curated by ChEMBL | Assay Description Binding affinity against rat adenosine A3 receptor expressed in CHO cells using [125I]-. | J Med Chem 41: 3174-85 (1998) Article DOI: 10.1021/jm980147p BindingDB Entry DOI: 10.7270/Q2765G10 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 9219 total ) | Next | Last >> |