 Found 765 hits with Last Name = 'howard' and Initial = 'l'
Found 765 hits with Last Name = 'howard' and Initial = 'l' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50006774
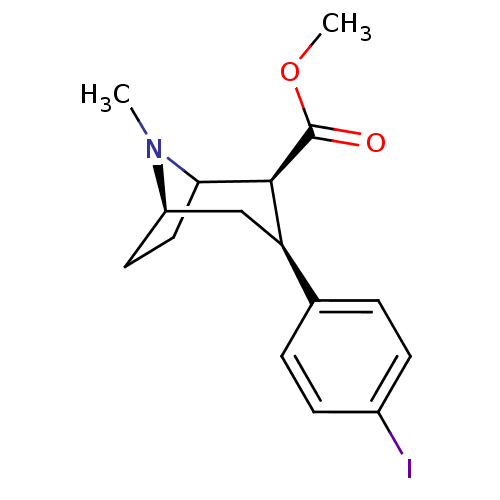
((2S,3S)-methyl 3-(4-iodophenyl)-8-methyl-8-aza-bic...)Show SMILES COC(=O)[C@@H]1C2CC[C@H](C[C@@H]1c1ccc(I)cc1)N2C |TLB:19:18:4.10.9:6.7,THB:2:4:18:6.7,11:10:18:6.7| Show InChI InChI=1S/C16H20INO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12-,13-,14?,15+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]paroxetine from 5HTT |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50156916
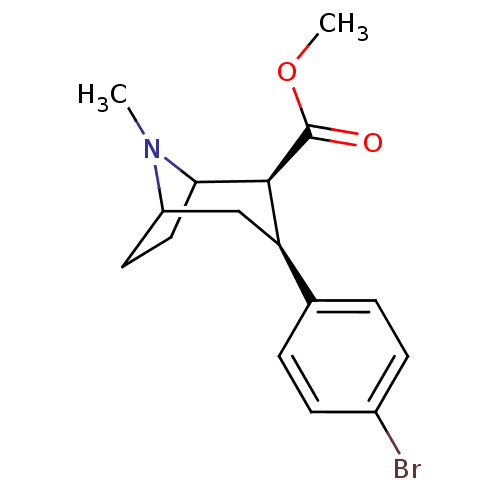
((1R)-3beta-(4-bromophenyl)tropane-2beta-carboxylic...)Show SMILES COC(=O)[C@@H]1C2CCC(C[C@@H]1c1ccc(Br)cc1)N2C |TLB:11:10:18:7.6,THB:2:4:18:7.6| Show InChI InChI=1S/C16H20BrNO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12?,13-,14?,15+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.960 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]paroxetine from 5HTT |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192752
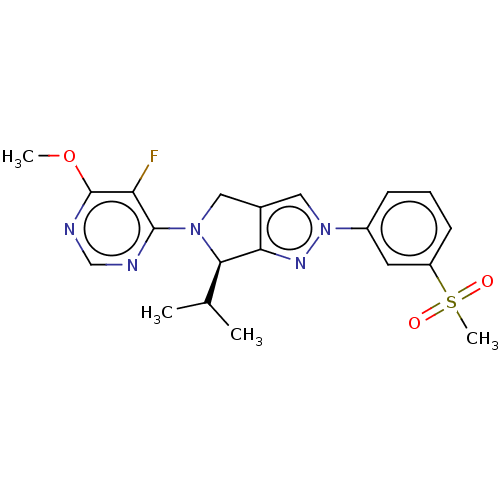
(CHEMBL3905741)Show SMILES COc1ncnc(N2Cc3cn(nc3[C@H]2C(C)C)-c2cccc(c2)S(C)(=O)=O)c1F |r| Show InChI InChI=1S/C20H22FN5O3S/c1-12(2)18-17-13(9-25(18)19-16(21)20(29-3)23-11-22-19)10-26(24-17)14-6-5-7-15(8-14)30(4,27)28/h5-8,10-12,18H,9H2,1-4H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177016
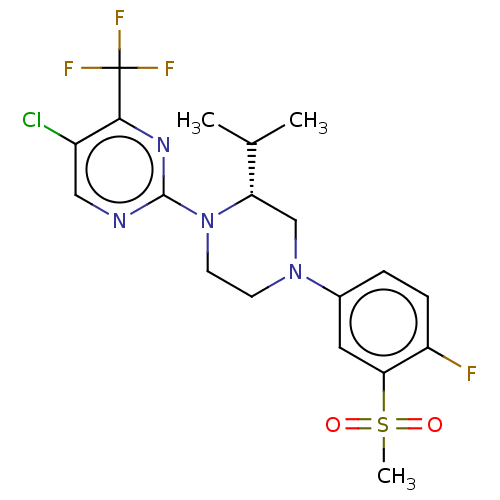
(CHEMBL3814501)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(Cl)c(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H21ClF4N4O2S/c1-11(2)15-10-27(12-4-5-14(21)16(8-12)31(3,29)30)6-7-28(15)18-25-9-13(20)17(26-18)19(22,23)24/h4-5,8-9,11,15H,6-7,10H2,1-3H3/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177012
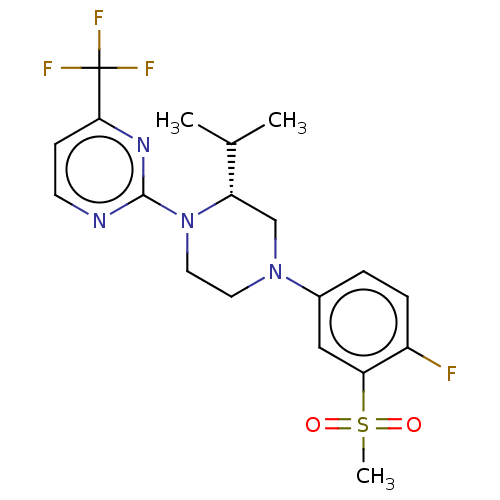
(CHEMBL3814153 | US10144715, Compound 7-32)Show SMILES CC(C)[C@@H]1CN(CCN1c1nccc(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H22F4N4O2S/c1-12(2)15-11-26(13-4-5-14(20)16(10-13)30(3,28)29)8-9-27(15)18-24-7-6-17(25-18)19(21,22)23/h4-7,10,12,15H,8-9,11H2,1-3H3/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177010
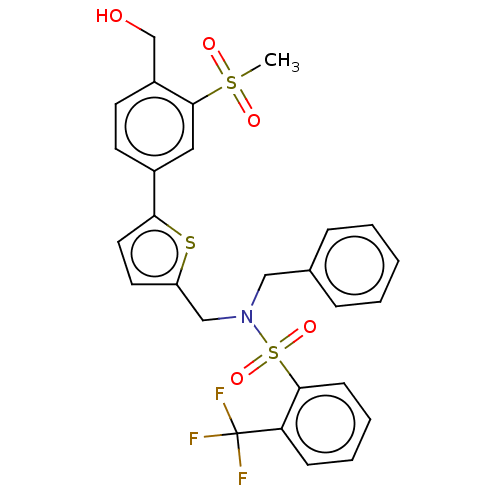
(CHEMBL3814006)Show SMILES CS(=O)(=O)c1cc(ccc1CO)-c1ccc(CN(Cc2ccccc2)S(=O)(=O)c2ccccc2C(F)(F)F)s1 Show InChI InChI=1S/C27H24F3NO5S3/c1-38(33,34)26-15-20(11-12-21(26)18-32)24-14-13-22(37-24)17-31(16-19-7-3-2-4-8-19)39(35,36)25-10-6-5-9-23(25)27(28,29)30/h2-15,32H,16-18H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177015
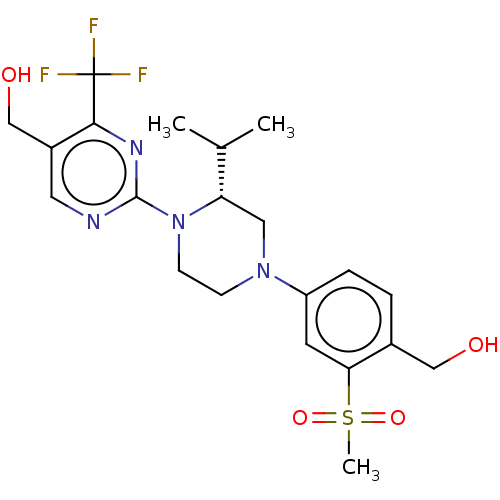
(CHEMBL3814206 | US10144715, Compound 19-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(CO)c(n1)C(F)(F)F)c1ccc(CO)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H27F3N4O4S/c1-13(2)17-10-27(16-5-4-14(11-29)18(8-16)33(3,31)32)6-7-28(17)20-25-9-15(12-30)19(26-20)21(22,23)24/h4-5,8-9,13,17,29-30H,6-7,10-12H2,1-3H3/t17-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177015

(CHEMBL3814206 | US10144715, Compound 19-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(CO)c(n1)C(F)(F)F)c1ccc(CO)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H27F3N4O4S/c1-13(2)17-10-27(16-5-4-14(11-29)18(8-16)33(3,31)32)6-7-28(17)20-25-9-15(12-30)19(26-20)21(22,23)24/h4-5,8-9,13,17,29-30H,6-7,10-12H2,1-3H3/t17-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177016

(CHEMBL3814501)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(Cl)c(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H21ClF4N4O2S/c1-11(2)15-10-27(12-4-5-14(21)16(8-12)31(3,29)30)6-7-28(15)18-25-9-13(20)17(26-18)19(22,23)24/h4-5,8-9,11,15H,6-7,10H2,1-3H3/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192753
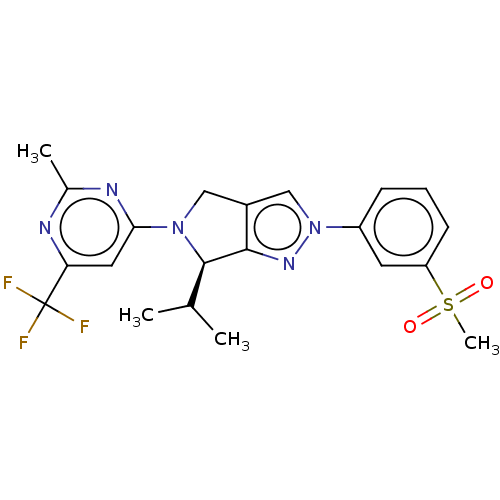
(CHEMBL3985591)Show SMILES CC(C)[C@H]1N(Cc2cn(nc12)-c1cccc(c1)S(C)(=O)=O)c1cc(nc(C)n1)C(F)(F)F |r| Show InChI InChI=1S/C21H22F3N5O2S/c1-12(2)20-19-14(10-28(20)18-9-17(21(22,23)24)25-13(3)26-18)11-29(27-19)15-6-5-7-16(8-15)32(4,30)31/h5-9,11-12,20H,10H2,1-4H3/t20-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50156915
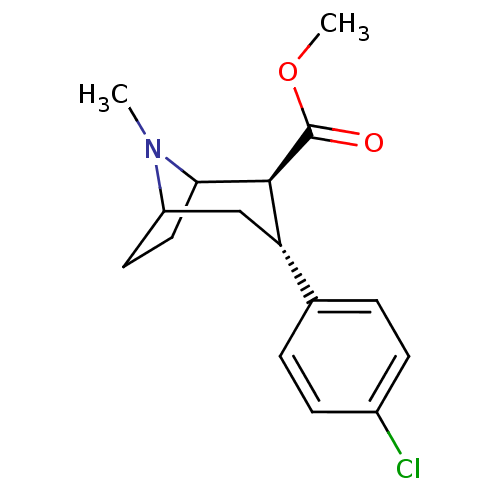
((2R,3S)-methyl 3-(4-chlorophenyl)-8-methyl-8-aza-b...)Show SMILES COC(=O)[C@@H]1C2CCC(C[C@H]1c1ccc(Cl)cc1)N2C |r,THB:2:4:18:6.7| Show InChI InChI=1S/C16H20ClNO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12?,13-,14?,15-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from NET |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192757
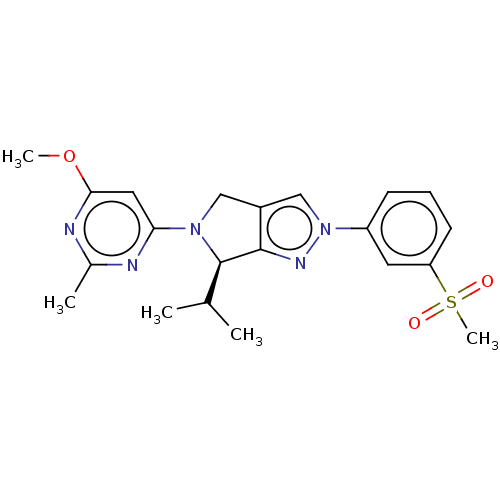
(CHEMBL3914727)Show SMILES COc1cc(nc(C)n1)N1Cc2cn(nc2[C@H]1C(C)C)-c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H25N5O3S/c1-13(2)21-20-15(11-25(21)18-10-19(29-4)23-14(3)22-18)12-26(24-20)16-7-6-8-17(9-16)30(5,27)28/h6-10,12-13,21H,11H2,1-5H3/t21-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177011
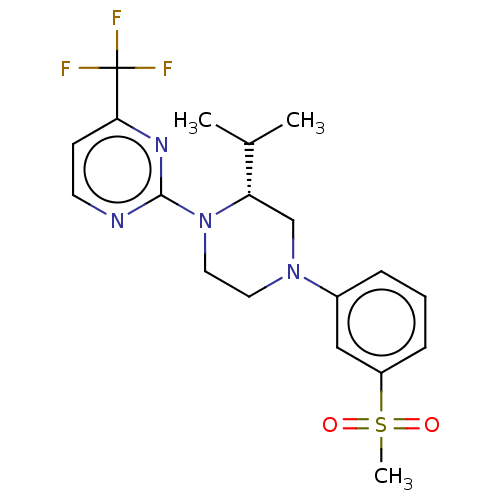
(CHEMBL3815014 | US10144715, Compound 7-13)Show SMILES CC(C)[C@@H]1CN(CCN1c1nccc(n1)C(F)(F)F)c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H23F3N4O2S/c1-13(2)16-12-25(14-5-4-6-15(11-14)29(3,27)28)9-10-26(16)18-23-8-7-17(24-18)19(20,21)22/h4-8,11,13,16H,9-10,12H2,1-3H3/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50035738

((R)-3-(4-Chloro-phenyl)-8-methyl-8-aza-bicyclo[3.2...)Show SMILES COC(=O)C1C2CCC(CC1c1ccc(Cl)cc1)N2C |TLB:11:10:18:6.7,THB:2:4:18:6.7| Show InChI InChI=1S/C16H20ClNO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]paroxetine from 5HTT |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50156915

((2R,3S)-methyl 3-(4-chlorophenyl)-8-methyl-8-aza-b...)Show SMILES COC(=O)[C@@H]1C2CCC(C[C@H]1c1ccc(Cl)cc1)N2C |r,THB:2:4:18:6.7| Show InChI InChI=1S/C16H20ClNO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12?,13-,14?,15-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]paroxetine from 5HTT |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192750
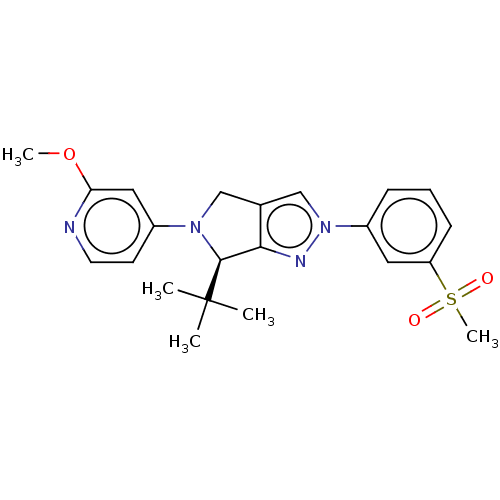
(CHEMBL3940521)Show SMILES COc1cc(ccn1)N1Cc2cn(nc2[C@H]1C(C)(C)C)-c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C22H26N4O3S/c1-22(2,3)21-20-15(13-25(21)16-9-10-23-19(12-16)29-4)14-26(24-20)17-7-6-8-18(11-17)30(5,27)28/h6-12,14,21H,13H2,1-5H3/t21-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50156917

((2R,3S)-methyl 3-(4-iodophenyl)-8-methyl-8-aza-bic...)Show SMILES COC(=O)[C@@H]1C2CCC(C[C@H]1c1ccc(I)cc1)N2C |r,THB:2:4:18:6.7| Show InChI InChI=1S/C16H20INO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12?,13-,14?,15-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]paroxetine from 5HTT |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192761
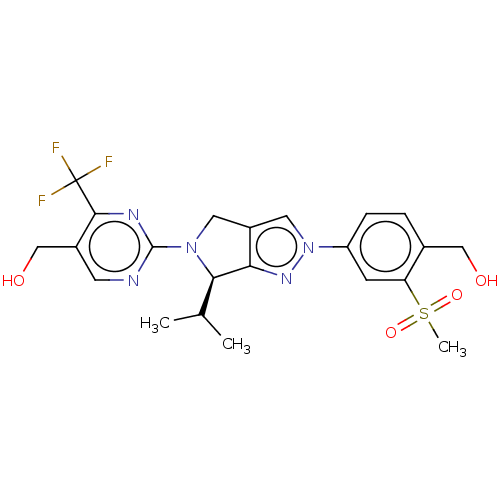
(CHEMBL3978980)Show SMILES CC(C)[C@H]1N(Cc2cn(nc12)-c1ccc(CO)c(c1)S(C)(=O)=O)c1ncc(CO)c(n1)C(F)(F)F |r| Show InChI InChI=1S/C22H24F3N5O4S/c1-12(2)19-18-15(8-29(19)21-26-7-14(11-32)20(27-21)22(23,24)25)9-30(28-18)16-5-4-13(10-31)17(6-16)35(3,33)34/h4-7,9,12,19,31-32H,8,10-11H2,1-3H3/t19-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192758
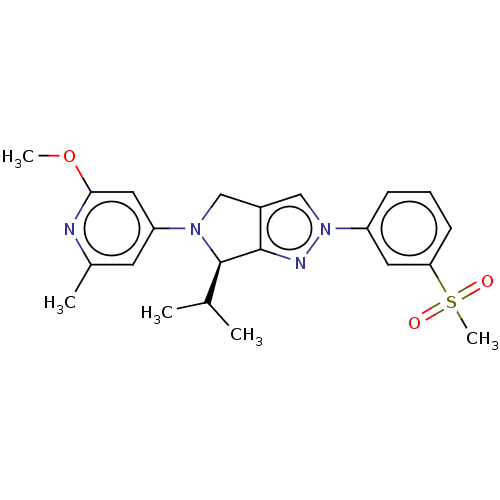
(CHEMBL3976470)Show SMILES COc1cc(cc(C)n1)N1Cc2cn(nc2[C@H]1C(C)C)-c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C22H26N4O3S/c1-14(2)22-21-16(12-25(22)18-9-15(3)23-20(11-18)29-4)13-26(24-21)17-7-6-8-19(10-17)30(5,27)28/h6-11,13-14,22H,12H2,1-5H3/t22-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192756
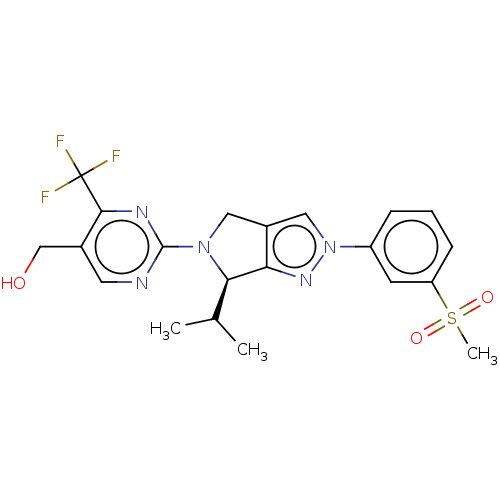
(CHEMBL3932169)Show SMILES CC(C)[C@H]1N(Cc2cn(nc12)-c1cccc(c1)S(C)(=O)=O)c1ncc(CO)c(n1)C(F)(F)F |r| Show InChI InChI=1S/C21H22F3N5O3S/c1-12(2)18-17-14(10-29(27-17)15-5-4-6-16(7-15)33(3,31)32)9-28(18)20-25-8-13(11-30)19(26-20)21(22,23)24/h4-8,10,12,18,30H,9,11H2,1-3H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192762
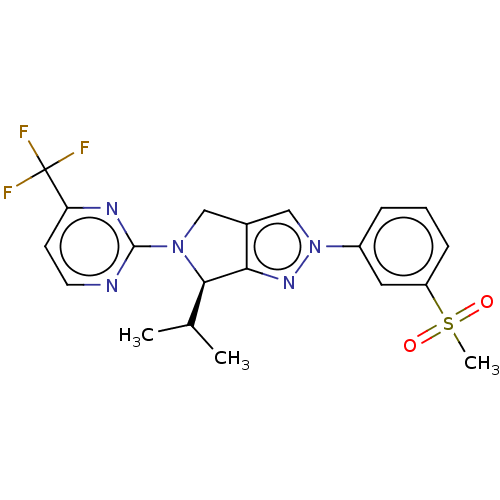
(CHEMBL3959681)Show SMILES CC(C)[C@H]1N(Cc2cn(nc12)-c1cccc(c1)S(C)(=O)=O)c1nccc(n1)C(F)(F)F |r| Show InChI InChI=1S/C20H20F3N5O2S/c1-12(2)18-17-13(10-27(18)19-24-8-7-16(25-19)20(21,22)23)11-28(26-17)14-5-4-6-15(9-14)31(3,29)30/h4-9,11-12,18H,10H2,1-3H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192759

(CHEMBL3960195)Show SMILES CC(C)[C@H]1N(Cc2cn(nc12)-c1cccc(c1)S(C)(=O)=O)c1cc(ncn1)C(F)(F)F |r| Show InChI InChI=1S/C20H20F3N5O2S/c1-12(2)19-18-13(9-27(19)17-8-16(20(21,22)23)24-11-25-17)10-28(26-18)14-5-4-6-15(7-14)31(3,29)30/h4-8,10-12,19H,9H2,1-3H3/t19-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50192752

(CHEMBL3905741)Show SMILES COc1ncnc(N2Cc3cn(nc3[C@H]2C(C)C)-c2cccc(c2)S(C)(=O)=O)c1F |r| Show InChI InChI=1S/C20H22FN5O3S/c1-12(2)18-17-13(9-25(18)19-16(21)20(29-3)23-11-22-19)10-26(24-17)14-6-5-7-15(8-14)30(4,27)28/h5-8,10-12,18H,9H2,1-4H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRalpha LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177018
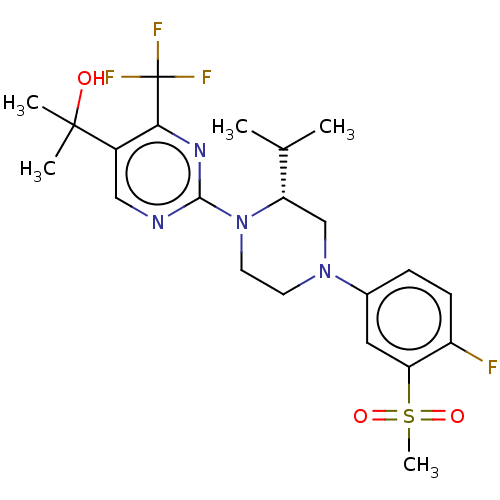
(CHEMBL3814478 | US10144715, Compound 14-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(c(n1)C(F)(F)F)C(C)(C)O)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C22H28F4N4O3S/c1-13(2)17-12-29(14-6-7-16(23)18(10-14)34(5,32)33)8-9-30(17)20-27-11-15(21(3,4)31)19(28-20)22(24,25)26/h6-7,10-11,13,17,31H,8-9,12H2,1-5H3/t17-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50138642
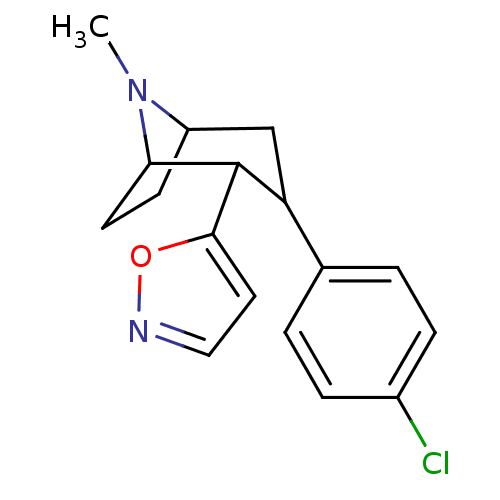
(3-(4-Chloro-phenyl)-2-isoxazol-5-yl-8-methyl-8-aza...)Show SMILES CN1C2CCC1C(C(C2)c1ccc(Cl)cc1)c1ccno1 |TLB:16:6:1:4.3,9:7:1:4.3| Show InChI InChI=1S/C17H19ClN2O/c1-20-13-6-7-15(20)17(16-8-9-19-21-16)14(10-13)11-2-4-12(18)5-3-11/h2-5,8-9,13-15,17H,6-7,10H2,1H3 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Binding affinity towards serotonin transporter 5-HTT was determined using [3H]paroxetine radioligand. |
J Med Chem 47: 296-302 (2004)
Article DOI: 10.1021/jm030453p
BindingDB Entry DOI: 10.7270/Q2VT1SVR |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177017
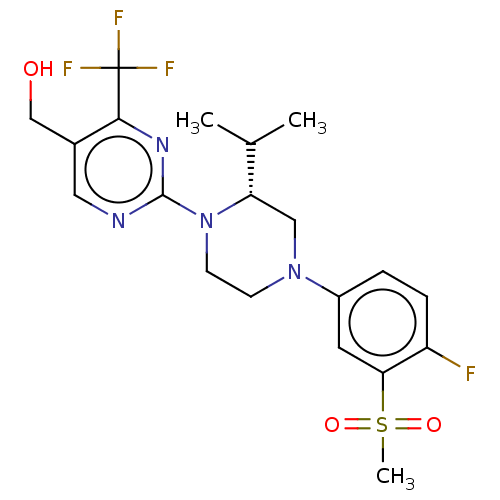
(CHEMBL3815001 | US10144715, Compound 11-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(CO)c(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C20H24F4N4O3S/c1-12(2)16-10-27(14-4-5-15(21)17(8-14)32(3,30)31)6-7-28(16)19-25-9-13(11-29)18(26-19)20(22,23)24/h4-5,8-9,12,16,29H,6-7,10-11H2,1-3H3/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192760
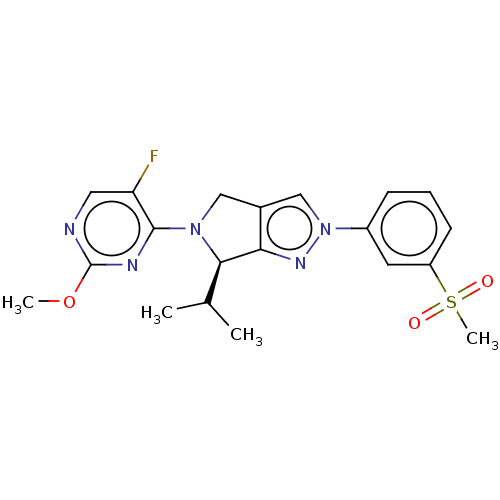
(CHEMBL3972392)Show SMILES COc1ncc(F)c(n1)N1Cc2cn(nc2[C@H]1C(C)C)-c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C20H22FN5O3S/c1-12(2)18-17-13(10-25(18)19-16(21)9-22-20(23-19)29-3)11-26(24-17)14-6-5-7-15(8-14)30(4,27)28/h5-9,11-12,18H,10H2,1-4H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192754
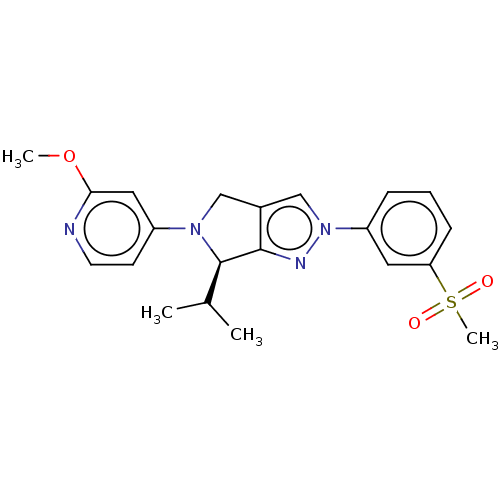
(CHEMBL3913348)Show SMILES COc1cc(ccn1)N1Cc2cn(nc2[C@H]1C(C)C)-c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H24N4O3S/c1-14(2)21-20-15(12-24(21)16-8-9-22-19(11-16)28-3)13-25(23-20)17-6-5-7-18(10-17)29(4,26)27/h5-11,13-14,21H,12H2,1-4H3/t21-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192763
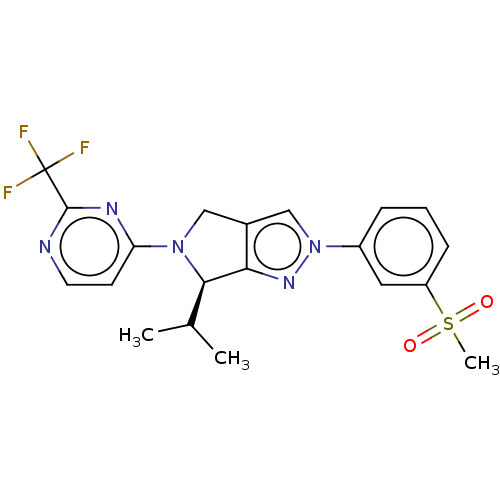
(CHEMBL3933543)Show SMILES CC(C)[C@H]1N(Cc2cn(nc12)-c1cccc(c1)S(C)(=O)=O)c1ccnc(n1)C(F)(F)F |r| Show InChI InChI=1S/C20H20F3N5O2S/c1-12(2)18-17-13(10-27(18)16-7-8-24-19(25-16)20(21,22)23)11-28(26-17)14-5-4-6-15(9-14)31(3,29)30/h4-9,11-12,18H,10H2,1-3H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50156918
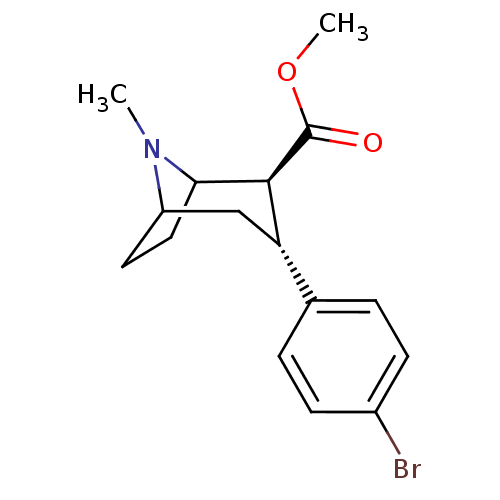
((2R,3S)-methyl 3-(4-bromophenyl)-8-methyl-8-aza-bi...)Show SMILES COC(=O)[C@@H]1C2CCC(C[C@H]1c1ccc(Br)cc1)N2C |r,THB:2:4:18:6.7| Show InChI InChI=1S/C16H20BrNO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12?,13-,14?,15-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 16.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from NET |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192755
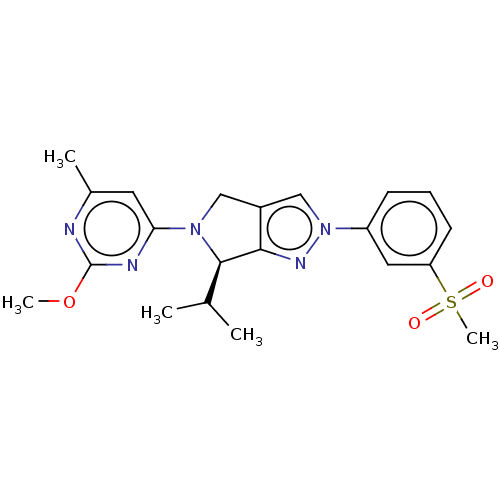
(CHEMBL3956923)Show SMILES COc1nc(C)cc(n1)N1Cc2cn(nc2[C@H]1C(C)C)-c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H25N5O3S/c1-13(2)20-19-15(11-25(20)18-9-14(3)22-21(23-18)29-4)12-26(24-19)16-7-6-8-17(10-16)30(5,27)28/h6-10,12-13,20H,11H2,1-5H3/t20-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM19993

(CHEMBL62136 | N-[4-(1,1,1,3,3,3-hexafluoro-2-hydro...)Show SMILES OC(c1ccc(cc1)N(CC(F)(F)F)S(=O)(=O)c1ccccc1)(C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C17H12F9NO3S/c18-14(19,20)10-27(31(29,30)13-4-2-1-3-5-13)12-8-6-11(7-9-12)15(28,16(21,22)23)17(24,25)26/h1-9,28H,10H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50192762

(CHEMBL3959681)Show SMILES CC(C)[C@H]1N(Cc2cn(nc12)-c1cccc(c1)S(C)(=O)=O)c1nccc(n1)C(F)(F)F |r| Show InChI InChI=1S/C20H20F3N5O2S/c1-12(2)18-17-13(10-27(18)19-24-8-7-16(25-19)20(21,22)23)11-28(26-17)14-5-4-6-15(9-14)31(3,29)30/h4-9,11-12,18H,10H2,1-3H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRalpha LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177010

(CHEMBL3814006)Show SMILES CS(=O)(=O)c1cc(ccc1CO)-c1ccc(CN(Cc2ccccc2)S(=O)(=O)c2ccccc2C(F)(F)F)s1 Show InChI InChI=1S/C27H24F3NO5S3/c1-38(33,34)26-15-20(11-12-21(26)18-32)24-14-13-22(37-24)17-31(16-19-7-3-2-4-8-19)39(35,36)25-10-6-5-9-23(25)27(28,29)30/h2-15,32H,16-18H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50156918

((2R,3S)-methyl 3-(4-bromophenyl)-8-methyl-8-aza-bi...)Show SMILES COC(=O)[C@@H]1C2CCC(C[C@H]1c1ccc(Br)cc1)N2C |r,THB:2:4:18:6.7| Show InChI InChI=1S/C16H20BrNO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12?,13-,14?,15-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 20.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]paroxetine from 5HTT |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50006774

((2S,3S)-methyl 3-(4-iodophenyl)-8-methyl-8-aza-bic...)Show SMILES COC(=O)[C@@H]1C2CC[C@H](C[C@@H]1c1ccc(I)cc1)N2C |TLB:19:18:4.10.9:6.7,THB:2:4:18:6.7,11:10:18:6.7| Show InChI InChI=1S/C16H20INO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12-,13-,14?,15+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from NET |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50035738

((R)-3-(4-Chloro-phenyl)-8-methyl-8-aza-bicyclo[3.2...)Show SMILES COC(=O)C1C2CCC(CC1c1ccc(Cl)cc1)N2C |TLB:11:10:18:6.7,THB:2:4:18:6.7| Show InChI InChI=1S/C16H20ClNO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from NET |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50192757

(CHEMBL3914727)Show SMILES COc1cc(nc(C)n1)N1Cc2cn(nc2[C@H]1C(C)C)-c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H25N5O3S/c1-13(2)21-20-15(11-25(21)18-10-19(29-4)23-14(3)22-18)12-26(24-20)16-7-6-8-17(9-16)30(5,27)28/h6-10,12-13,21H,11H2,1-5H3/t21-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRalpha LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50006781

((2S,3S)-methyl 8-methyl-3-p-tolyl-8-aza-bicyclo[3....)Show SMILES COC(=O)[C@@H]1C2CC[C@H](C[C@@H]1c1ccc(C)cc1)N2C |TLB:19:18:4.10.9:6.7,THB:2:4:18:6.7,11:10:18:6.7| Show InChI InChI=1S/C17H23NO2/c1-11-4-6-12(7-5-11)14-10-13-8-9-15(18(13)2)16(14)17(19)20-3/h4-7,13-16H,8-10H2,1-3H3/t13-,14-,15?,16+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]paroxetine from 5HTT |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50156916

((1R)-3beta-(4-bromophenyl)tropane-2beta-carboxylic...)Show SMILES COC(=O)[C@@H]1C2CCC(C[C@@H]1c1ccc(Br)cc1)N2C |TLB:11:10:18:7.6,THB:2:4:18:7.6| Show InChI InChI=1S/C16H20BrNO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12?,13-,14?,15+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from NET |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM19993

(CHEMBL62136 | N-[4-(1,1,1,3,3,3-hexafluoro-2-hydro...)Show SMILES OC(c1ccc(cc1)N(CC(F)(F)F)S(=O)(=O)c1ccccc1)(C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C17H12F9NO3S/c18-14(19,20)10-27(31(29,30)13-4-2-1-3-5-13)12-8-6-11(7-9-12)15(28,16(21,22)23)17(24,25)26/h1-9,28H,10H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRalpha LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177012

(CHEMBL3814153 | US10144715, Compound 7-32)Show SMILES CC(C)[C@@H]1CN(CCN1c1nccc(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H22F4N4O2S/c1-12(2)15-11-26(13-4-5-14(20)16(10-13)30(3,28)29)8-9-27(15)18-24-7-6-17(25-18)19(21,22)23/h4-7,10,12,15H,8-9,11H2,1-3H3/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Transporter
(Rattus norvegicus) | BDBM50138646
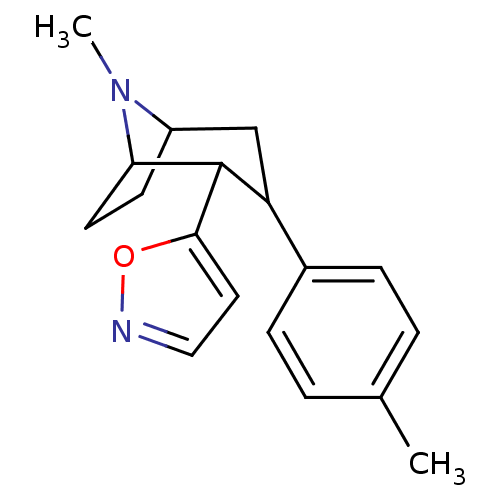
(2-Isoxazol-5-yl-8-methyl-3-p-tolyl-8-aza-bicyclo[3...)Show SMILES CN1C2CCC1C(C(C2)c1ccc(C)cc1)c1ccno1 |TLB:16:6:1:4.3,9:7:1:4.3| Show InChI InChI=1S/C18H22N2O/c1-12-3-5-13(6-4-12)15-11-14-7-8-16(20(14)2)18(15)17-9-10-19-21-17/h3-6,9-10,14-16,18H,7-8,11H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Binding affinity towards norepinephrine transporter was determined using [3H]nisoxetine radioligand. |
J Med Chem 47: 296-302 (2004)
Article DOI: 10.1021/jm030453p
BindingDB Entry DOI: 10.7270/Q2VT1SVR |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192751
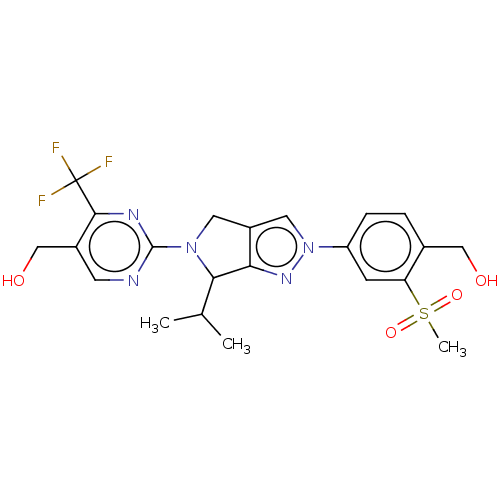
(CHEMBL3889601)Show SMILES CC(C)C1N(Cc2cn(nc12)-c1ccc(CO)c(c1)S(C)(=O)=O)c1ncc(CO)c(n1)C(F)(F)F Show InChI InChI=1S/C22H24F3N5O4S/c1-12(2)19-18-15(8-29(19)21-26-7-14(11-32)20(27-21)22(23,24)25)9-30(28-18)16-5-4-13(10-31)17(6-16)35(3,33)34/h4-7,9,12,19,31-32H,8,10-11H2,1-3H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50192751

(CHEMBL3889601)Show SMILES CC(C)C1N(Cc2cn(nc12)-c1ccc(CO)c(c1)S(C)(=O)=O)c1ncc(CO)c(n1)C(F)(F)F Show InChI InChI=1S/C22H24F3N5O4S/c1-12(2)19-18-15(8-29(19)21-26-7-14(11-32)20(27-21)22(23,24)25)9-30(28-18)16-5-4-13(10-31)17(6-16)35(3,33)34/h4-7,9,12,19,31-32H,8,10-11H2,1-3H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50156917

((2R,3S)-methyl 3-(4-iodophenyl)-8-methyl-8-aza-bic...)Show SMILES COC(=O)[C@@H]1C2CCC(C[C@H]1c1ccc(I)cc1)N2C |r,THB:2:4:18:6.7| Show InChI InChI=1S/C16H20INO2/c1-18-12-7-8-14(18)15(16(19)20-2)13(9-12)10-3-5-11(17)6-4-10/h3-6,12-15H,7-9H2,1-2H3/t12?,13-,14?,15-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from NET |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50192753

(CHEMBL3985591)Show SMILES CC(C)[C@H]1N(Cc2cn(nc12)-c1cccc(c1)S(C)(=O)=O)c1cc(nc(C)n1)C(F)(F)F |r| Show InChI InChI=1S/C21H22F3N5O2S/c1-12(2)20-19-14(10-28(20)18-9-17(21(22,23)24)25-13(3)26-18)11-29(27-19)15-6-5-7-16(8-15)32(4,30)31/h5-9,11-12,20H,10H2,1-4H3/t20-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled T0901317 from LXRalpha LBD (unknown origin) |
Bioorg Med Chem Lett 26: 5044-5050 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.089
BindingDB Entry DOI: 10.7270/Q28P62GC |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50006781

((2S,3S)-methyl 8-methyl-3-p-tolyl-8-aza-bicyclo[3....)Show SMILES COC(=O)[C@@H]1C2CC[C@H](C[C@@H]1c1ccc(C)cc1)N2C |TLB:19:18:4.10.9:6.7,THB:2:4:18:6.7,11:10:18:6.7| Show InChI InChI=1S/C17H23NO2/c1-11-4-6-12(7-5-11)14-10-13-8-9-15(18(13)2)16(14)17(19)20-3/h4-7,13-16H,8-10H2,1-3H3/t13-,14-,15?,16+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 36 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from NET |
J Med Chem 47: 6401-9 (2004)
Article DOI: 10.1021/jm0401311
BindingDB Entry DOI: 10.7270/Q2W37X4B |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50138646

(2-Isoxazol-5-yl-8-methyl-3-p-tolyl-8-aza-bicyclo[3...)Show SMILES CN1C2CCC1C(C(C2)c1ccc(C)cc1)c1ccno1 |TLB:16:6:1:4.3,9:7:1:4.3| Show InChI InChI=1S/C18H22N2O/c1-12-3-5-13(6-4-12)15-11-14-7-8-16(20(14)2)18(15)17-9-10-19-21-17/h3-6,9-10,14-16,18H,7-8,11H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Binding affinity towards serotonin transporter 5-HTT was determined using [3H]paroxetine radioligand. |
J Med Chem 47: 296-302 (2004)
Article DOI: 10.1021/jm030453p
BindingDB Entry DOI: 10.7270/Q2VT1SVR |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177011

(CHEMBL3815014 | US10144715, Compound 7-13)Show SMILES CC(C)[C@@H]1CN(CCN1c1nccc(n1)C(F)(F)F)c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H23F3N4O2S/c1-13(2)16-12-25(14-5-4-6-15(11-14)29(3,27)28)9-10-26(16)18-23-8-7-17(24-18)19(20,21)22/h4-8,11,13,16H,9-10,12H2,1-3H3/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data