 Found 79 hits with Last Name = 'harrison' and Initial = 'rj'
Found 79 hits with Last Name = 'harrison' and Initial = 'rj' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080842
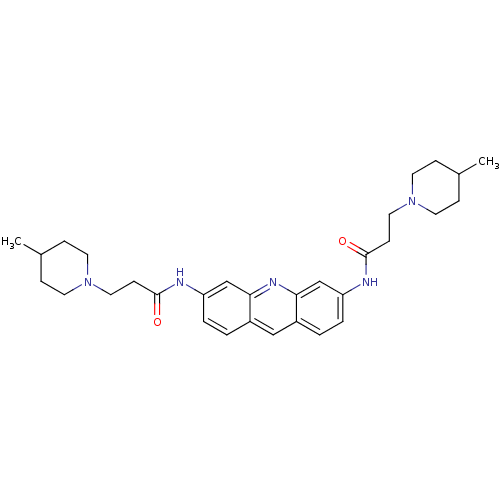
(3,6-Bis[3-(4-methylpiperidino)propionamido]acridin...)Show SMILES CC1CCN(CCC(=O)Nc2ccc3cc4ccc(NC(=O)CCN5CCC(C)CC5)cc4nc3c2)CC1 Show InChI InChI=1S/C31H41N5O2/c1-22-7-13-35(14-8-22)17-11-30(37)32-26-5-3-24-19-25-4-6-27(21-29(25)34-28(24)20-26)33-31(38)12-18-36-15-9-23(2)10-16-36/h3-6,19-23H,7-18H2,1-2H3,(H,32,37)(H,33,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 1.35E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080848
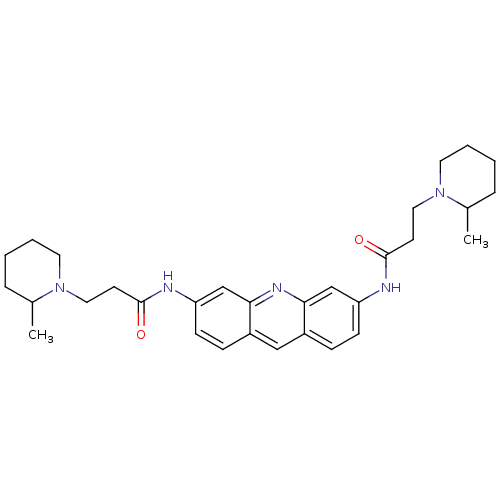
(3-(2-Methyl-piperidin-1-yl)-N-{6-[3-(2-methyl-pipe...)Show SMILES CC1CCCCN1CCC(=O)Nc1ccc2cc3ccc(NC(=O)CCN4CCCCC4C)cc3nc2c1 Show InChI InChI=1S/C31H41N5O2/c1-22-7-3-5-15-35(22)17-13-30(37)32-26-11-9-24-19-25-10-12-27(21-29(25)34-28(24)20-26)33-31(38)14-18-36-16-6-4-8-23(36)2/h9-12,19-23H,3-8,13-18H2,1-2H3,(H,32,37)(H,33,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080837

(3-(2-Ethyl-piperidin-1-yl)-N-{6-[3-(2-ethyl-piperi...)Show SMILES CCC1CCCCN1CCC(=O)Nc1ccc2cc3ccc(NC(=O)CCN4CCCCC4CC)cc3nc2c1 Show InChI InChI=1S/C33H45N5O2/c1-3-28-9-5-7-17-37(28)19-15-32(39)34-26-13-11-24-21-25-12-14-27(23-31(25)36-30(24)22-26)35-33(40)16-20-38-18-8-6-10-29(38)4-2/h11-14,21-23,28-29H,3-10,15-20H2,1-2H3,(H,34,39)(H,35,40) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080847

(3,6-Bis(3-piperidinopropionamido)acridine | 3-Pipe...)Show SMILES O=C(CCN1CCCCC1)Nc1ccc2cc3ccc(NC(=O)CCN4CCCCC4)cc3nc2c1 Show InChI InChI=1S/C29H37N5O2/c35-28(11-17-33-13-3-1-4-14-33)30-24-9-7-22-19-23-8-10-25(21-27(23)32-26(22)20-24)31-29(36)12-18-34-15-5-2-6-16-34/h7-10,19-21H,1-6,11-18H2,(H,30,35)(H,31,36) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080840
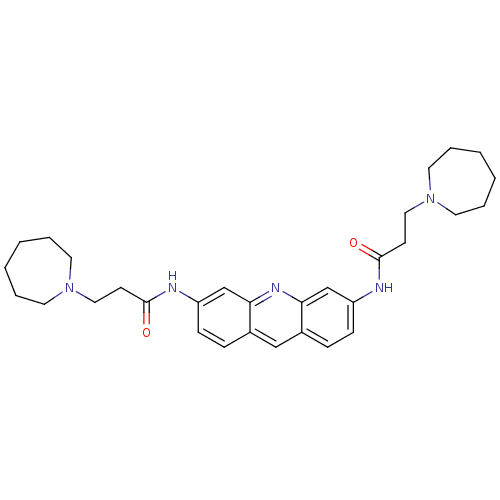
(3,6-Bis[3-(azepan-1-yl)propionamido]acridine | 3,6...)Show SMILES O=C(CCN1CCCCCC1)Nc1ccc2cc3ccc(NC(=O)CCN4CCCCCC4)cc3nc2c1 Show InChI InChI=1S/C31H41N5O2/c37-30(13-19-35-15-5-1-2-6-16-35)32-26-11-9-24-21-25-10-12-27(23-29(25)34-28(24)22-26)33-31(38)14-20-36-17-7-3-4-8-18-36/h9-12,21-23H,1-8,13-20H2,(H,32,37)(H,33,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 3.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080850

(3-[2-(2-Hydroxy-ethyl)-piperidin-1-yl]-N-(6-{3-[2-...)Show SMILES OCCC1CCCCN1CCC(=O)Nc1ccc2cc3ccc(NC(=O)CCN4CCCCC4CCO)cc3nc2c1 Show InChI InChI=1S/C33H45N5O4/c39-19-13-28-5-1-3-15-37(28)17-11-32(41)34-26-9-7-24-21-25-8-10-27(23-31(25)36-30(24)22-26)35-33(42)12-18-38-16-4-2-6-29(38)14-20-40/h7-10,21-23,28-29,39-40H,1-6,11-20H2,(H,34,41)(H,35,42) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080839

(3-(3-Methyl-piperidin-1-yl)-N-{6-[3-(3-methyl-pipe...)Show SMILES CC1CCCN(CCC(=O)Nc2ccc3cc4ccc(NC(=O)CCN5CCCC(C)C5)cc4nc3c2)C1 Show InChI InChI=1S/C31H41N5O2/c1-22-5-3-13-35(20-22)15-11-30(37)32-26-9-7-24-17-25-8-10-27(19-29(25)34-28(24)18-26)33-31(38)12-16-36-14-4-6-23(2)21-36/h7-10,17-19,22-23H,3-6,11-16,20-21H2,1-2H3,(H,32,37)(H,33,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 4.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080849
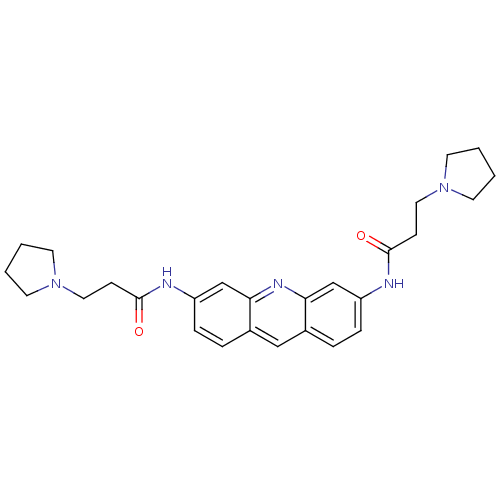
(3,6-Bis(3-pyrrolidinopropionamido)acridine | 3-PYR...)Show SMILES O=C(CCN1CCCC1)Nc1ccc2cc3ccc(NC(=O)CCN4CCCC4)cc3nc2c1 Show InChI InChI=1S/C27H33N5O2/c33-26(9-15-31-11-1-2-12-31)28-22-7-5-20-17-21-6-8-23(19-25(21)30-24(20)18-22)29-27(34)10-16-32-13-3-4-14-32/h5-8,17-19H,1-4,9-16H2,(H,28,33)(H,29,34) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 5.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080841

(3-(2-Hydroxymethyl-piperidin-1-yl)-N-{6-[3-(2-hydr...)Show SMILES OCC1CCCCN1CCC(=O)Nc1ccc2cc3ccc(NC(=O)CCN4CCCCC4CO)cc3nc2c1 Show InChI InChI=1S/C31H41N5O4/c37-20-26-5-1-3-13-35(26)15-11-30(39)32-24-9-7-22-17-23-8-10-25(19-29(23)34-28(22)18-24)33-31(40)12-16-36-14-4-2-6-27(36)21-38/h7-10,17-19,26-27,37-38H,1-6,11-16,20-21H2,(H,32,39)(H,33,40) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080844
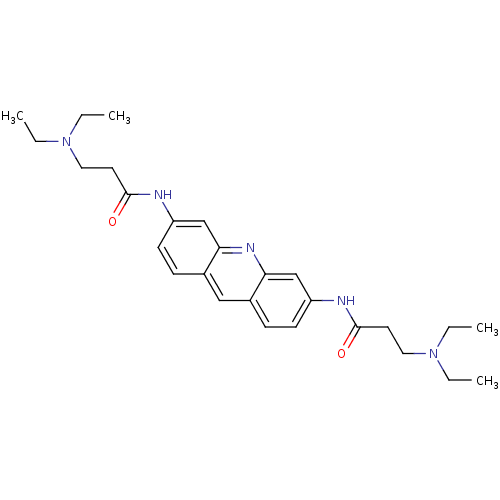
(3-Diethylamino-N-[6-(3-diethylamino-propionylamino...)Show SMILES CCN(CC)CCC(=O)Nc1ccc2cc3ccc(NC(=O)CCN(CC)CC)cc3nc2c1 Show InChI InChI=1S/C27H37N5O2/c1-5-31(6-2)15-13-26(33)28-22-11-9-20-17-21-10-12-23(19-25(21)30-24(20)18-22)29-27(34)14-16-32(7-3)8-4/h9-12,17-19H,5-8,13-16H2,1-4H3,(H,28,33)(H,29,34) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080845
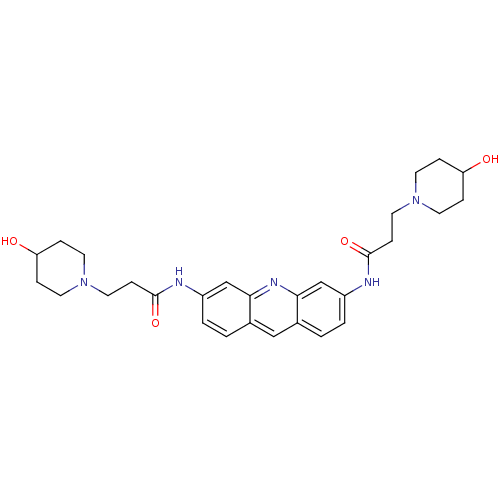
(3-(4-Hydroxy-piperidin-1-yl)-N-{6-[3-(4-hydroxy-pi...)Show SMILES OC1CCN(CCC(=O)Nc2ccc3cc4ccc(NC(=O)CCN5CCC(O)CC5)cc4nc3c2)CC1 Show InChI InChI=1S/C29H37N5O4/c35-24-5-11-33(12-6-24)15-9-28(37)30-22-3-1-20-17-21-2-4-23(19-27(21)32-26(20)18-22)31-29(38)10-16-34-13-7-25(36)8-14-34/h1-4,17-19,24-25,35-36H,5-16H2,(H,30,37)(H,31,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080851

(3-Dimethylamino-N-[6-(3-dimethylamino-propionylami...)Show SMILES CN(C)CCC(=O)Nc1ccc2cc3ccc(NC(=O)CCN(C)C)cc3nc2c1 Show InChI InChI=1S/C23H29N5O2/c1-27(2)11-9-22(29)24-18-7-5-16-13-17-6-8-19(15-21(17)26-20(16)14-18)25-23(30)10-12-28(3)4/h5-8,13-15H,9-12H2,1-4H3,(H,24,29)(H,25,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080852
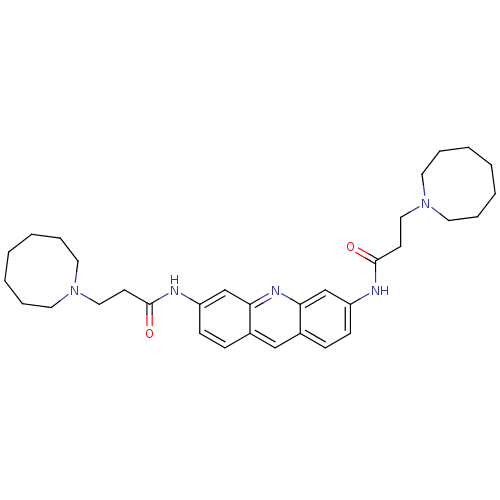
(3,6-Bis[3-(heptamethyleneimino)piperizinopropionam...)Show SMILES O=C(CCN1CCCCCCC1)Nc1ccc2cc3ccc(NC(=O)CCN4CCCCCCC4)cc3nc2c1 Show InChI InChI=1S/C33H45N5O2/c39-32(15-21-37-17-7-3-1-4-8-18-37)34-28-13-11-26-23-27-12-14-29(25-31(27)36-30(26)24-28)35-33(40)16-22-38-19-9-5-2-6-10-20-38/h11-14,23-25H,1-10,15-22H2,(H,34,39)(H,35,40) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080843

(3,6-Bis[(3-morpholinopropionamido)] acridine | 3-M...)Show SMILES O=C(CCN1CCOCC1)Nc1ccc2cc3ccc(NC(=O)CCN4CCOCC4)cc3nc2c1 Show InChI InChI=1S/C27H33N5O4/c33-26(5-7-31-9-13-35-14-10-31)28-22-3-1-20-17-21-2-4-23(19-25(21)30-24(20)18-22)29-27(34)6-8-32-11-15-36-16-12-32/h1-4,17-19H,5-16H2,(H,28,33)(H,29,34) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080838

(3-(4-Benzyl-piperidin-1-yl)-N-{6-[3-(4-benzyl-pipe...)Show SMILES O=C(CCN1CCC(Cc2ccccc2)CC1)Nc1ccc2cc3ccc(NC(=O)CCN4CCC(Cc5ccccc5)CC4)cc3nc2c1 Show InChI InChI=1S/C43H49N5O2/c49-42(19-25-47-21-15-34(16-22-47)27-32-7-3-1-4-8-32)44-38-13-11-36-29-37-12-14-39(31-41(37)46-40(36)30-38)45-43(50)20-26-48-23-17-35(18-24-48)28-33-9-5-2-6-10-33/h1-14,29-31,34-35H,15-28H2,(H,44,49)(H,45,50) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080846

(3,6-Bis[3-(1,3,3-trimethyl-6-azabicyclo[3.2.1]octa...)Show SMILES CC12CC(CC(C)(C)C1)N(CCC(=O)Nc1ccc3cc4ccc(NC(=O)CCN5CC6(C)CC5CC(C)(C)C6)cc4nc3c1)C2 |TLB:10:9:2:5.4.8,28:29:33:36.35.39| Show InChI InChI=1S/C39H53N5O2/c1-36(2)18-30-20-38(5,22-36)24-43(30)13-11-34(45)40-28-9-7-26-15-27-8-10-29(17-33(27)42-32(26)16-28)41-35(46)12-14-44-25-39(6)21-31(44)19-37(3,4)23-39/h7-10,15-17,30-31H,11-14,18-25H2,1-6H3,(H,40,45)(H,41,46) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Ability to inhibit human telomerase in a modified cell-free TRAP assay using extracts from A2780 cell line |
Bioorg Med Chem Lett 9: 2463-8 (1999)
BindingDB Entry DOI: 10.7270/Q2H41QN7 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50133999
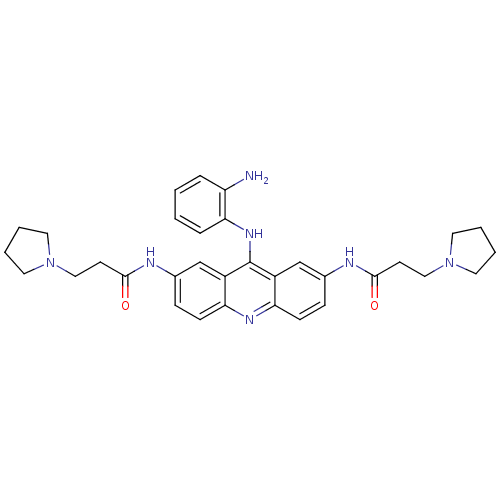
(CHEMBL345035 | N,N'-(9-(2-aminophenylamino)acridin...)Show SMILES Nc1ccccc1Nc1c2cc(NC(=O)CCN3CCCC3)ccc2nc2ccc(NC(=O)CCN3CCCC3)cc12 Show InChI InChI=1S/C33H39N7O2/c34-27-7-1-2-8-30(27)38-33-25-21-23(35-31(41)13-19-39-15-3-4-16-39)9-11-28(25)37-29-12-10-24(22-26(29)33)36-32(42)14-20-40-17-5-6-18-40/h1-2,7-12,21-22H,3-6,13-20,34H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 170 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134000
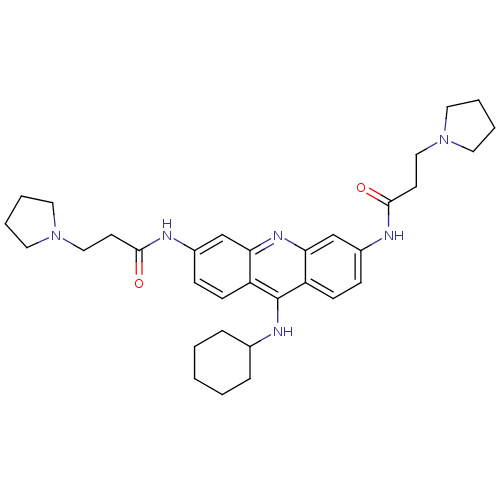
(CHEMBL139287 | N,N'-(9-(cyclohexylamino)acridine-3...)Show SMILES O=C(CCN1CCCC1)Nc1ccc2c(NC3CCCCC3)c3ccc(NC(=O)CCN4CCCC4)cc3nc2c1 Show InChI InChI=1S/C33H44N6O2/c40-31(14-20-38-16-4-5-17-38)34-25-10-12-27-29(22-25)37-30-23-26(35-32(41)15-21-39-18-6-7-19-39)11-13-28(30)33(27)36-24-8-2-1-3-9-24/h10-13,22-24H,1-9,14-21H2,(H,34,40)(H,35,41)(H,36,37) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 90 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134004
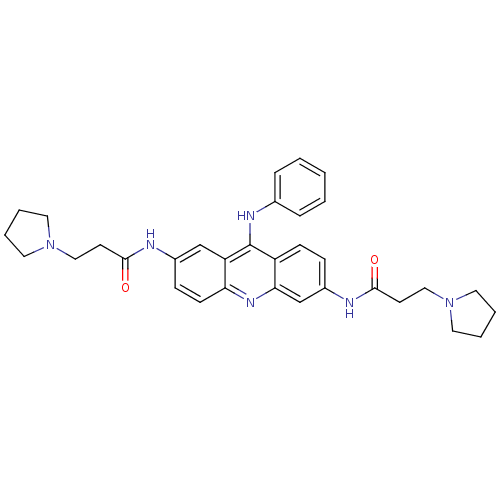
(CHEMBL140354 | N,N'-(9-(phenylamino)acridine-2,6-d...)Show SMILES O=C(CCN1CCCC1)Nc1ccc2c(Nc3ccccc3)c3cc(NC(=O)CCN4CCCC4)ccc3nc2c1 Show InChI InChI=1S/C33H38N6O2/c40-31(14-20-38-16-4-5-17-38)34-25-11-13-29-28(22-25)33(36-24-8-2-1-3-9-24)27-12-10-26(23-30(27)37-29)35-32(41)15-21-39-18-6-7-19-39/h1-3,8-13,22-23H,4-7,14-21H2,(H,34,40)(H,35,41)(H,36,37) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.33E+3 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134010
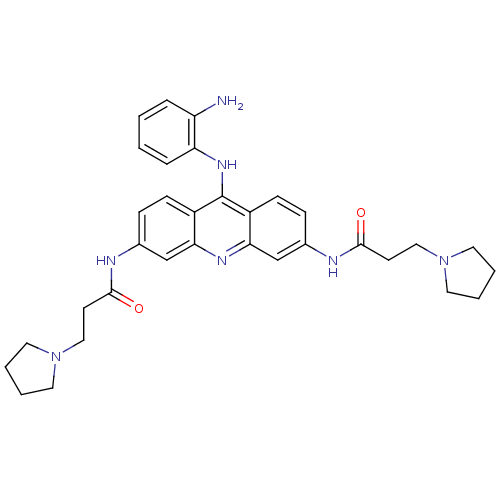
(CHEMBL139511 | N,N'-(9-(2-aminophenylamino)acridin...)Show SMILES Nc1ccccc1Nc1c2ccc(NC(=O)CCN3CCCC3)cc2nc2cc(NC(=O)CCN3CCCC3)ccc12 Show InChI InChI=1S/C33H39N7O2/c34-27-7-1-2-8-28(27)38-33-25-11-9-23(35-31(41)13-19-39-15-3-4-16-39)21-29(25)37-30-22-24(10-12-26(30)33)36-32(42)14-20-40-17-5-6-18-40/h1-2,7-12,21-22H,3-6,13-20,34H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 20 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134007
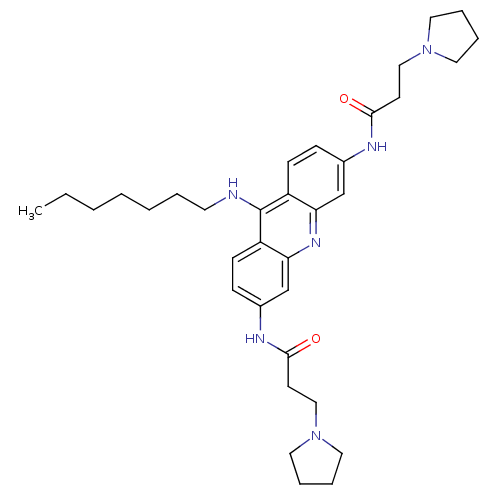
(CHEMBL337123 | N-[9-Heptylamino-6-(3-pyrrolidin-1-...)Show SMILES CCCCCCCNc1c2ccc(NC(=O)CCN3CCCC3)cc2nc2cc(NC(=O)CCN3CCCC3)ccc12 Show InChI InChI=1S/C34H48N6O2/c1-2-3-4-5-6-17-35-34-28-13-11-26(36-32(41)15-22-39-18-7-8-19-39)24-30(28)38-31-25-27(12-14-29(31)34)37-33(42)16-23-40-20-9-10-21-40/h11-14,24-25H,2-10,15-23H2,1H3,(H,35,38)(H,36,41)(H,37,42) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 210 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134009
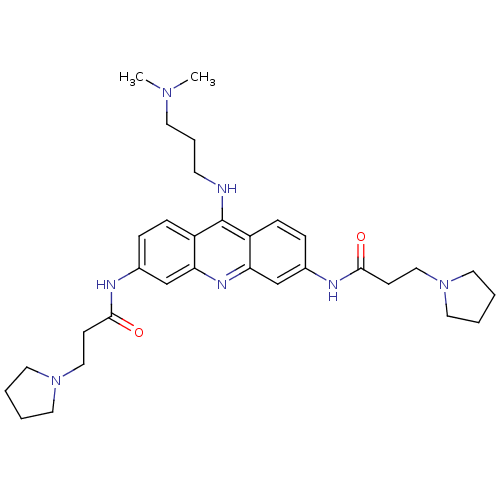
(CHEMBL335132 | N,N'-(9-(3-(dimethylamino)propylami...)Show SMILES CN(C)CCCNc1c2ccc(NC(=O)CCN3CCCC3)cc2nc2cc(NC(=O)CCN3CCCC3)ccc12 Show InChI InChI=1S/C32H45N7O2/c1-37(2)15-7-14-33-32-26-10-8-24(34-30(40)12-20-38-16-3-4-17-38)22-28(26)36-29-23-25(9-11-27(29)32)35-31(41)13-21-39-18-5-6-19-39/h8-11,22-23H,3-7,12-21H2,1-2H3,(H,33,36)(H,34,40)(H,35,41) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 60 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134011
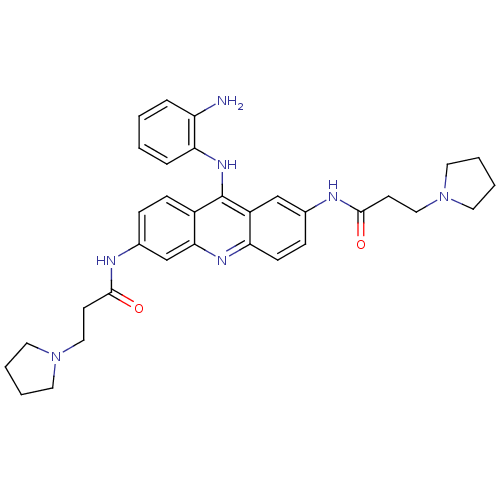
(CHEMBL343609 | N,N'-(9-(2-aminophenylamino)acridin...)Show SMILES Nc1ccccc1Nc1c2ccc(NC(=O)CCN3CCCC3)cc2nc2ccc(NC(=O)CCN3CCCC3)cc12 Show InChI InChI=1S/C33H39N7O2/c34-27-7-1-2-8-29(27)38-33-25-11-9-24(36-32(42)14-20-40-17-5-6-18-40)22-30(25)37-28-12-10-23(21-26(28)33)35-31(41)13-19-39-15-3-4-16-39/h1-2,7-12,21-22H,3-6,13-20,34H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 21 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134012
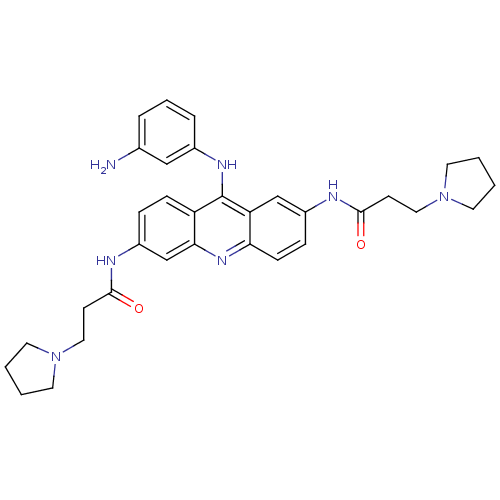
(CHEMBL342480 | N,N'-(9-(3-aminophenylamino)acridin...)Show SMILES Nc1cccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3ccc(NC(=O)CCN4CCCC4)cc23)c1 Show InChI InChI=1S/C33H39N7O2/c34-23-6-5-7-24(20-23)37-33-27-10-8-26(36-32(42)13-19-40-16-3-4-17-40)22-30(27)38-29-11-9-25(21-28(29)33)35-31(41)12-18-39-14-1-2-15-39/h5-11,20-22H,1-4,12-19,34H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 210 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134014

(CHEMBL343041 | N,N'-(9-(2-hydroxyphenylamino)acrid...)Show SMILES Oc1ccccc1Nc1c2cc(NC(=O)CCN3CCCC3)ccc2nc2ccc(NC(=O)CCN3CCCC3)cc12 Show InChI InChI=1S/C33H38N6O3/c40-30-8-2-1-7-29(30)37-33-25-21-23(34-31(41)13-19-38-15-3-4-16-38)9-11-27(25)36-28-12-10-24(22-26(28)33)35-32(42)14-20-39-17-5-6-18-39/h1-2,7-12,21-22,40H,3-6,13-20H2,(H,34,41)(H,35,42)(H,36,37) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.03E+3 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134018
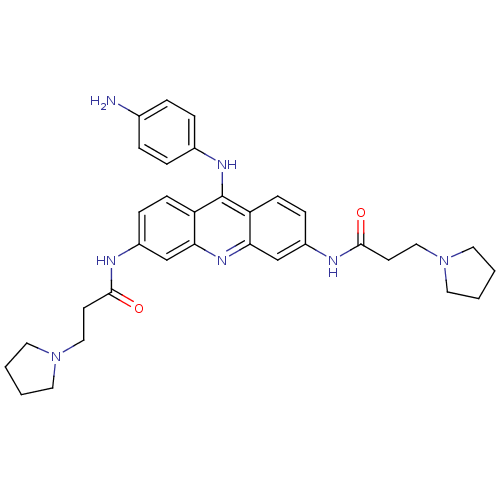
(CHEMBL335819 | N,N'-(9-(4-aminophenylamino)acridin...)Show SMILES Nc1ccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3cc(NC(=O)CCN4CCCC4)ccc23)cc1 Show InChI InChI=1S/C33H39N7O2/c34-23-5-7-24(8-6-23)37-33-27-11-9-25(35-31(41)13-19-39-15-1-2-16-39)21-29(27)38-30-22-26(10-12-28(30)33)36-32(42)14-20-40-17-3-4-18-40/h5-12,21-22H,1-4,13-20,34H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 74 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50133998

(CHEMBL137809 | N,N'-(9-(2-(methylthio)phenylamino)...)Show SMILES CSc1ccccc1Nc1c2ccc(NC(=O)CCN3CCCC3)cc2nc2cc(NC(=O)CCN3CCCC3)ccc12 Show InChI InChI=1S/C34H40N6O2S/c1-43-31-9-3-2-8-28(31)38-34-26-12-10-24(35-32(41)14-20-39-16-4-5-17-39)22-29(26)37-30-23-25(11-13-27(30)34)36-33(42)15-21-40-18-6-7-19-40/h2-3,8-13,22-23H,4-7,14-21H2,1H3,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 30 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134024

(CHEMBL139500 | N,N'-(9-(3-aminophenylamino)acridin...)Show SMILES Nc1cccc(Nc2c3cc(NC(=O)CCN4CCCC4)ccc3nc3ccc(NC(=O)CCN4CCCC4)cc23)c1 Show InChI InChI=1S/C33H39N7O2/c34-23-6-5-7-24(20-23)37-33-27-21-25(35-31(41)12-18-39-14-1-2-15-39)8-10-29(27)38-30-11-9-26(22-28(30)33)36-32(42)13-19-40-16-3-4-17-40/h5-11,20-22H,1-4,12-19,34H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.09E+3 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50080849

(3,6-Bis(3-pyrrolidinopropionamido)acridine | 3-PYR...)Show SMILES O=C(CCN1CCCC1)Nc1ccc2cc3ccc(NC(=O)CCN4CCCC4)cc3nc2c1 Show InChI InChI=1S/C27H33N5O2/c33-26(9-15-31-11-1-2-12-31)28-22-7-5-20-17-21-6-8-23(19-25(21)30-24(20)18-22)29-27(34)10-16-32-13-3-4-14-32/h5-8,17-19H,1-4,9-16H2,(H,28,33)(H,29,34) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.20E+3 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134025

(CHEMBL343795 | N,N'-(9-(4-fluorophenylamino)acridi...)Show SMILES Fc1ccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3cc(NC(=O)CCN4CCCC4)ccc23)cc1 Show InChI InChI=1S/C33H37FN6O2/c34-23-5-7-24(8-6-23)37-33-27-11-9-25(35-31(41)13-19-39-15-1-2-16-39)21-29(27)38-30-22-26(10-12-28(30)33)36-32(42)14-20-40-17-3-4-18-40/h5-12,21-22H,1-4,13-20H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 70 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134021
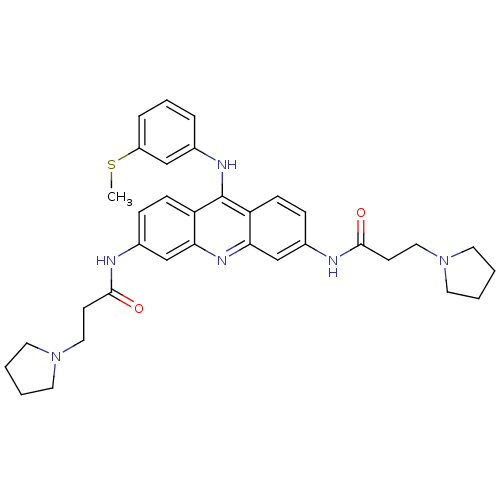
(CHEMBL138761 | N,N'-(9-(3-(methylthio)phenylamino)...)Show SMILES CSc1cccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3cc(NC(=O)CCN4CCCC4)ccc23)c1 Show InChI InChI=1S/C34H40N6O2S/c1-43-27-8-6-7-24(21-27)37-34-28-11-9-25(35-32(41)13-19-39-15-2-3-16-39)22-30(28)38-31-23-26(10-12-29(31)34)36-33(42)14-20-40-17-4-5-18-40/h6-12,21-23H,2-5,13-20H2,1H3,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 60 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134029
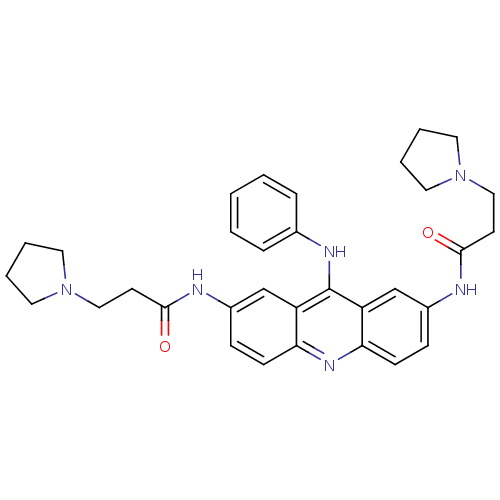
(CHEMBL138369 | N,N'-(9-(phenylamino)acridine-2,7-d...)Show SMILES O=C(CCN1CCCC1)Nc1ccc2nc3ccc(NC(=O)CCN4CCCC4)cc3c(Nc3ccccc3)c2c1 Show InChI InChI=1S/C33H38N6O2/c40-31(14-20-38-16-4-5-17-38)34-25-10-12-29-27(22-25)33(36-24-8-2-1-3-9-24)28-23-26(11-13-30(28)37-29)35-32(41)15-21-39-18-6-7-19-39/h1-3,8-13,22-23H,4-7,14-21H2,(H,34,40)(H,35,41)(H,36,37) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.29E+3 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134016
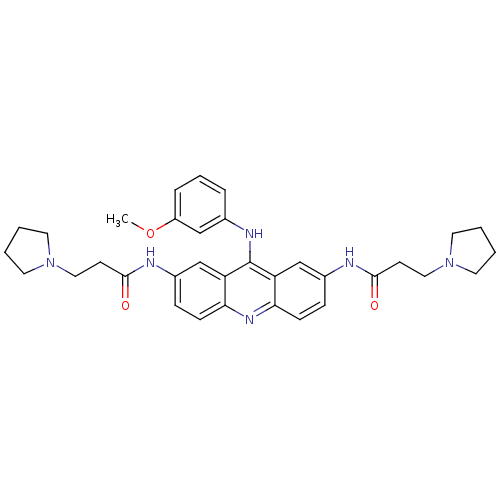
(CHEMBL342921 | N,N'-(9-(3-methoxyphenylamino)acrid...)Show SMILES COc1cccc(Nc2c3cc(NC(=O)CCN4CCCC4)ccc3nc3ccc(NC(=O)CCN4CCCC4)cc23)c1 Show InChI InChI=1S/C34H40N6O3/c1-43-27-8-6-7-24(21-27)37-34-28-22-25(35-32(41)13-19-39-15-2-3-16-39)9-11-30(28)38-31-12-10-26(23-29(31)34)36-33(42)14-20-40-17-4-5-18-40/h6-12,21-23H,2-5,13-20H2,1H3,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.73E+3 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134002
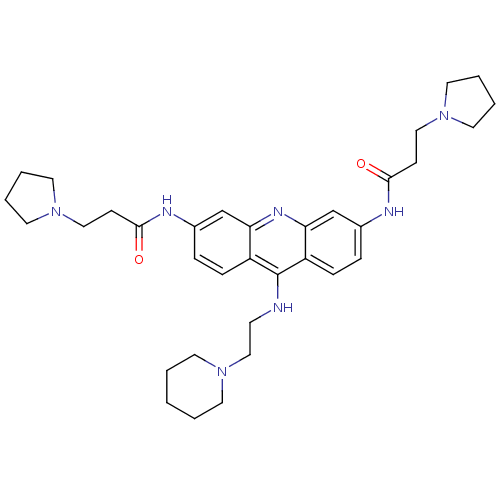
(CHEMBL141540 | N,N'-(9-(2-(piperidin-1-yl)ethylami...)Show SMILES O=C(CCN1CCCC1)Nc1ccc2c(NCCN3CCCCC3)c3ccc(NC(=O)CCN4CCCC4)cc3nc2c1 Show InChI InChI=1S/C34H47N7O2/c42-32(12-21-39-17-4-5-18-39)36-26-8-10-28-30(24-26)38-31-25-27(37-33(43)13-22-40-19-6-7-20-40)9-11-29(31)34(28)35-14-23-41-15-2-1-3-16-41/h8-11,24-25H,1-7,12-23H2,(H,35,38)(H,36,42)(H,37,43) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 500 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134028

(CHEMBL142036 | N,N'-(9-(4-aminophenylamino)acridin...)Show SMILES Nc1ccc(Nc2c3cc(NC(=O)CCN4CCCC4)ccc3nc3ccc(NC(=O)CCN4CCCC4)cc23)cc1 Show InChI InChI=1S/C33H39N7O2/c34-23-5-7-24(8-6-23)37-33-27-21-25(35-31(41)13-19-39-15-1-2-16-39)9-11-29(27)38-30-12-10-26(22-28(30)33)36-32(42)14-20-40-17-3-4-18-40/h5-12,21-22H,1-4,13-20,34H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 200 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134019

(CHEMBL336417 | N,N'-(9-(3-aminophenylamino)acridin...)Show SMILES Nc1cccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3cc(NC(=O)CCN4CCCC4)ccc23)c1 Show InChI InChI=1S/C33H39N7O2/c34-23-6-5-7-24(20-23)37-33-27-10-8-25(35-31(41)12-18-39-14-1-2-15-39)21-29(27)38-30-22-26(9-11-28(30)33)36-32(42)13-19-40-16-3-4-17-40/h5-11,20-22H,1-4,12-19,34H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 60 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134032

(CHEMBL138811 | N,N'-(9-(cyclopropylamino)acridine-...)Show SMILES O=C(CCN1CCCC1)Nc1ccc2c(NC3CC3)c3ccc(NC(=O)CCN4CCCC4)cc3nc2c1 Show InChI InChI=1S/C30H38N6O2/c37-28(11-17-35-13-1-2-14-35)31-22-7-9-24-26(19-22)34-27-20-23(8-10-25(27)30(24)33-21-5-6-21)32-29(38)12-18-36-15-3-4-16-36/h7-10,19-21H,1-6,11-18H2,(H,31,37)(H,32,38)(H,33,34) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 50 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134030
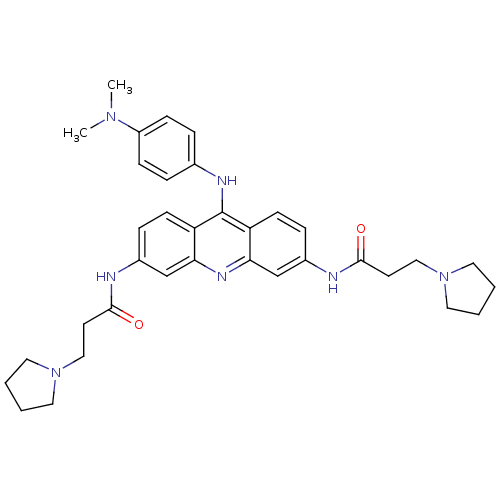
(9-[4-(N,N-dimethylamino)phenylamino]-3,6-bis(3-pyr...)Show SMILES CN(C)c1ccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3cc(NC(=O)CCN4CCCC4)ccc23)cc1 Show InChI InChI=1S/C35H43N7O2/c1-40(2)28-11-7-25(8-12-28)38-35-29-13-9-26(36-33(43)15-21-41-17-3-4-18-41)23-31(29)39-32-24-27(10-14-30(32)35)37-34(44)16-22-42-19-5-6-20-42/h7-14,23-24H,3-6,15-22H2,1-2H3,(H,36,43)(H,37,44)(H,38,39) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 115 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134031

(CHEMBL137973 | N,N'-(9-(cyclohexylamino)acridine-2...)Show SMILES O=C(CCN1CCCC1)Nc1ccc2c(NC3CCCCC3)c3cc(NC(=O)CCN4CCCC4)ccc3nc2c1 Show InChI InChI=1S/C33H44N6O2/c40-31(14-20-38-16-4-5-17-38)34-25-11-13-29-28(22-25)33(36-24-8-2-1-3-9-24)27-12-10-26(23-30(27)37-29)35-32(41)15-21-39-18-6-7-19-39/h10-13,22-24H,1-9,14-21H2,(H,34,40)(H,35,41)(H,36,37) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 210 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134013
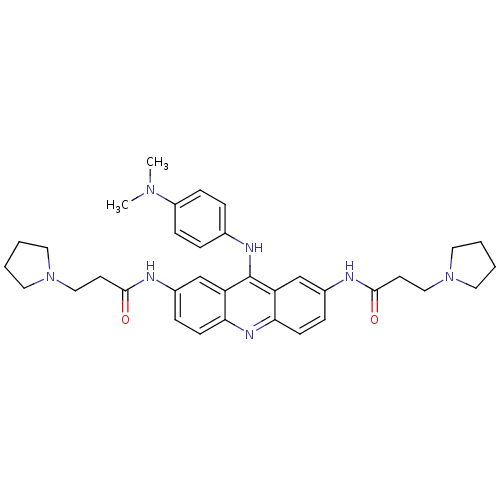
(CHEMBL341985 | N,N'-(9-(4-(dimethylamino)phenylami...)Show SMILES CN(C)c1ccc(Nc2c3cc(NC(=O)CCN4CCCC4)ccc3nc3ccc(NC(=O)CCN4CCCC4)cc23)cc1 Show InChI InChI=1S/C35H43N7O2/c1-40(2)28-11-7-25(8-12-28)38-35-29-23-26(36-33(43)15-21-41-17-3-4-18-41)9-13-31(29)39-32-14-10-27(24-30(32)35)37-34(44)16-22-42-19-5-6-20-42/h7-14,23-24H,3-6,15-22H2,1-2H3,(H,36,43)(H,37,44)(H,38,39) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 500 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134022
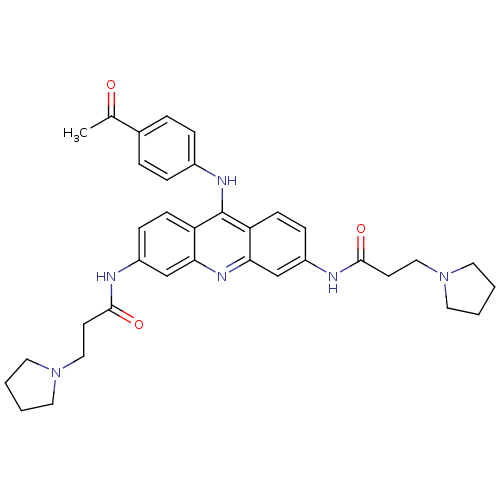
(CHEMBL336444 | N,N'-(9-(4-acetylphenylamino)acridi...)Show SMILES CC(=O)c1ccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3cc(NC(=O)CCN4CCCC4)ccc23)cc1 Show InChI InChI=1S/C35H40N6O3/c1-24(42)25-6-8-26(9-7-25)38-35-29-12-10-27(36-33(43)14-20-40-16-2-3-17-40)22-31(29)39-32-23-28(11-13-30(32)35)37-34(44)15-21-41-18-4-5-19-41/h6-13,22-23H,2-5,14-21H2,1H3,(H,36,43)(H,37,44)(H,38,39) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 40 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134017
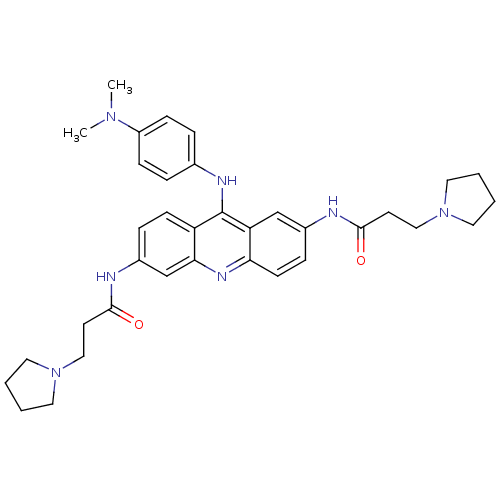
(CHEMBL138357 | N,N'-(9-(4-(dimethylamino)phenylami...)Show SMILES CN(C)c1ccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3ccc(NC(=O)CCN4CCCC4)cc23)cc1 Show InChI InChI=1S/C35H43N7O2/c1-40(2)28-11-7-25(8-12-28)38-35-29-13-9-27(37-34(44)16-22-42-19-5-6-20-42)24-32(29)39-31-14-10-26(23-30(31)35)36-33(43)15-21-41-17-3-4-18-41/h7-14,23-24H,3-6,15-22H2,1-2H3,(H,36,43)(H,37,44)(H,38,39) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 170 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134026
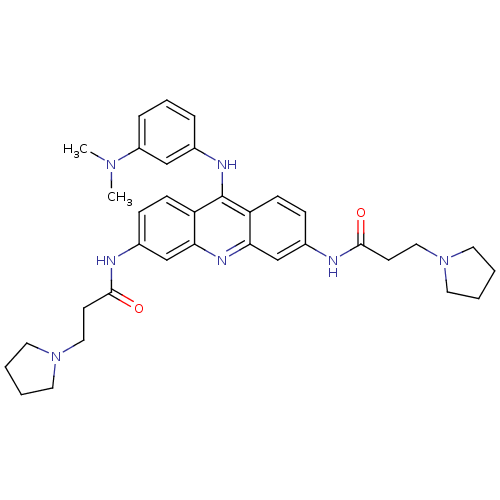
(CHEMBL138487 | N,N'-(9-(3-(dimethylamino)phenylami...)Show SMILES CN(C)c1cccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3cc(NC(=O)CCN4CCCC4)ccc23)c1 Show InChI InChI=1S/C35H43N7O2/c1-40(2)28-9-7-8-25(22-28)38-35-29-12-10-26(36-33(43)14-20-41-16-3-4-17-41)23-31(29)39-32-24-27(11-13-30(32)35)37-34(44)15-21-42-18-5-6-19-42/h7-13,22-24H,3-6,14-21H2,1-2H3,(H,36,43)(H,37,44)(H,38,39) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134033
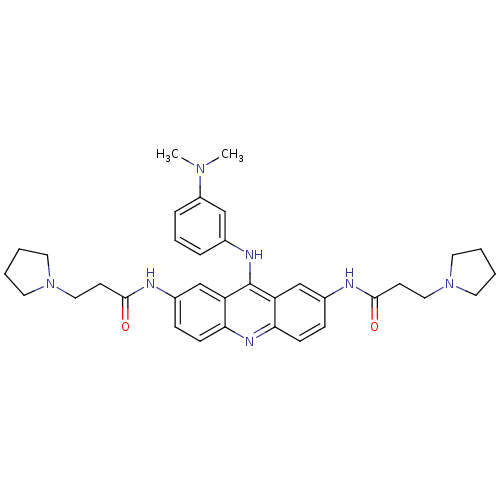
(CHEMBL139443 | N,N'-(9-(3-(dimethylamino)phenylami...)Show SMILES CN(C)c1cccc(Nc2c3cc(NC(=O)CCN4CCCC4)ccc3nc3ccc(NC(=O)CCN4CCCC4)cc23)c1 Show InChI InChI=1S/C35H43N7O2/c1-40(2)28-9-7-8-25(22-28)38-35-29-23-26(36-33(43)14-20-41-16-3-4-17-41)10-12-31(29)39-32-13-11-27(24-30(32)35)37-34(44)15-21-42-18-5-6-19-42/h7-13,22-24H,3-6,14-21H2,1-2H3,(H,36,43)(H,37,44)(H,38,39) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 600 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134015
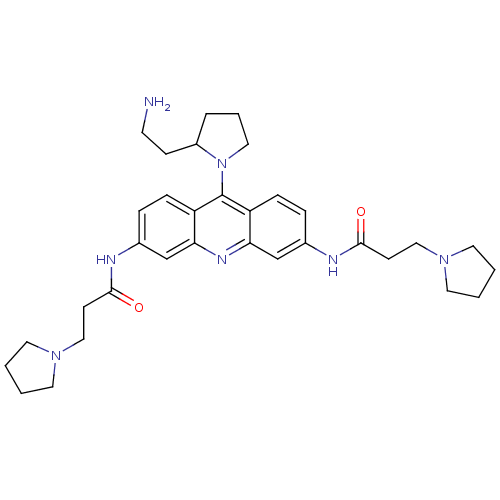
(CHEMBL44130 | N-[9-[2-(2-Amino-ethyl)-pyrrolidin-1...)Show SMILES NCCC1CCCN1c1c2ccc(NC(=O)CCN3CCCC3)cc2nc2cc(NC(=O)CCN3CCCC3)ccc12 Show InChI InChI=1S/C33H45N7O2/c34-14-11-26-6-5-19-40(26)33-27-9-7-24(35-31(41)12-20-38-15-1-2-16-38)22-29(27)37-30-23-25(8-10-28(30)33)36-32(42)13-21-39-17-3-4-18-39/h7-10,22-23,26H,1-6,11-21,34H2,(H,35,41)(H,36,42) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 18 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134008
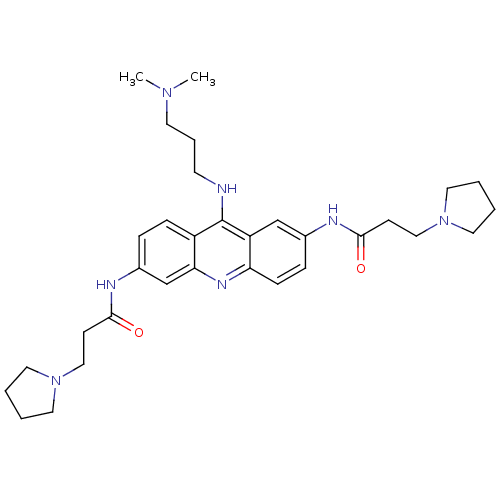
(CHEMBL138187 | N,N'-(9-(3-(dimethylamino)propylami...)Show SMILES CN(C)CCCNc1c2ccc(NC(=O)CCN3CCCC3)cc2nc2ccc(NC(=O)CCN3CCCC3)cc12 Show InChI InChI=1S/C32H45N7O2/c1-37(2)15-7-14-33-32-26-10-8-25(35-31(41)13-21-39-18-5-6-19-39)23-29(26)36-28-11-9-24(22-27(28)32)34-30(40)12-20-38-16-3-4-17-38/h8-11,22-23H,3-7,12-21H2,1-2H3,(H,33,36)(H,34,40)(H,35,41) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 80 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134023

(CHEMBL337762 | N,N'-(9-(4-aminophenylamino)acridin...)Show SMILES Nc1ccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3ccc(NC(=O)CCN4CCCC4)cc23)cc1 Show InChI InChI=1S/C33H39N7O2/c34-23-5-7-24(8-6-23)37-33-27-11-9-26(36-32(42)14-20-40-17-3-4-18-40)22-30(27)38-29-12-10-25(21-28(29)33)35-31(41)13-19-39-15-1-2-16-39/h5-12,21-22H,1-4,13-20,34H2,(H,35,41)(H,36,42)(H,37,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134006

(CHEMBL335564 | N,N'-(9-(2-methoxyethylamino)acridi...)Show SMILES COCCNc1c2ccc(NC(=O)CCN3CCCC3)cc2nc2cc(NC(=O)CCN3CCCC3)ccc12 Show InChI InChI=1S/C30H40N6O3/c1-39-19-12-31-30-24-8-6-22(32-28(37)10-17-35-13-2-3-14-35)20-26(24)34-27-21-23(7-9-25(27)30)33-29(38)11-18-36-15-4-5-16-36/h6-9,20-21H,2-5,10-19H2,1H3,(H,31,34)(H,32,37)(H,33,38) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 140 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134027
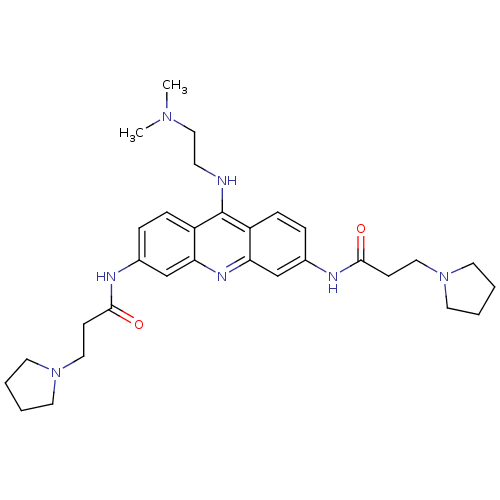
(CHEMBL140180 | N,N'-(9-(2-(dimethylamino)ethylamin...)Show SMILES CN(C)CCNc1c2ccc(NC(=O)CCN3CCCC3)cc2nc2cc(NC(=O)CCN3CCCC3)ccc12 Show InChI InChI=1S/C31H43N7O2/c1-36(2)20-13-32-31-25-9-7-23(33-29(39)11-18-37-14-3-4-15-37)21-27(25)35-28-22-24(8-10-26(28)31)34-30(40)12-19-38-16-5-6-17-38/h7-10,21-22H,3-6,11-20H2,1-2H3,(H,32,35)(H,33,39)(H,34,40) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 18 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |
Telomerase reverse transcriptase
(Homo sapiens (Human)) | BDBM50134020
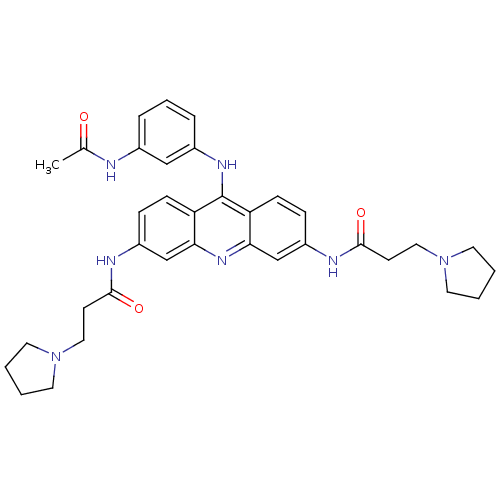
(CHEMBL141661 | N,N'-(9-(3-acetamidophenylamino)acr...)Show SMILES CC(=O)Nc1cccc(Nc2c3ccc(NC(=O)CCN4CCCC4)cc3nc3cc(NC(=O)CCN4CCCC4)ccc23)c1 Show InChI InChI=1S/C35H41N7O3/c1-24(43)36-25-7-6-8-26(21-25)39-35-29-11-9-27(37-33(44)13-19-41-15-2-3-16-41)22-31(29)40-32-23-28(10-12-30(32)35)38-34(45)14-20-42-17-4-5-18-42/h6-12,21-23H,2-5,13-20H2,1H3,(H,36,43)(H,37,44)(H,38,45)(H,39,40) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
University of London
Curated by ChEMBL
| Assay Description
Inhibitory activity against human telomerase |
J Med Chem 46: 4463-76 (2003)
Article DOI: 10.1021/jm0308693
BindingDB Entry DOI: 10.7270/Q2RR1XN3 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data