 Found 71 hits with Last Name = 'garg' and Initial = 'v'
Found 71 hits with Last Name = 'garg' and Initial = 'v' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50150105
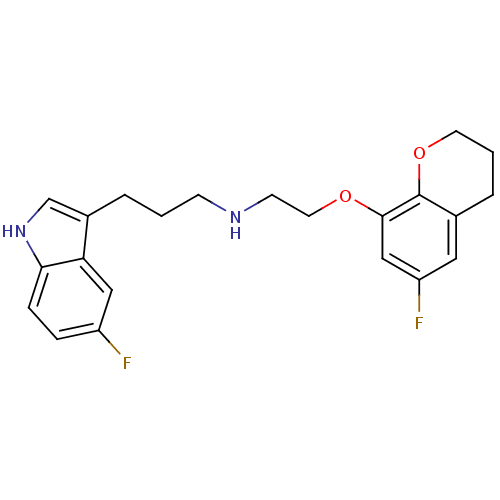
(CHEMBL124069 | [2-(6-Fluoro-chroman-8-yloxy)-ethyl...)Show SMILES Fc1cc2CCCOc2c(OCCNCCCc2c[nH]c3ccc(F)cc23)c1 Show InChI InChI=1S/C22H24F2N2O2/c23-17-5-6-20-19(12-17)16(14-26-20)3-1-7-25-8-10-27-21-13-18(24)11-15-4-2-9-28-22(15)21/h5-6,11-14,25-26H,1-4,7-10H2 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ISF College of Pharmacy
Curated by ChEMBL
| Assay Description
Inhibition of 5HT transporter (unknown origin) |
Eur J Med Chem 180: 562-612 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.019
BindingDB Entry DOI: 10.7270/Q2K93BWX |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50150093
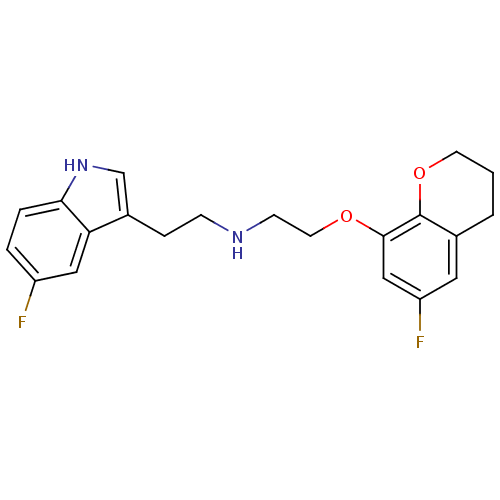
(CHEMBL340873 | [2-(6-Fluoro-chroman-8-yloxy)-ethyl...)Show InChI InChI=1S/C21H22F2N2O2/c22-16-3-4-19-18(11-16)15(13-25-19)5-6-24-7-9-26-20-12-17(23)10-14-2-1-8-27-21(14)20/h3-4,10-13,24-25H,1-2,5-9H2 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ISF College of Pharmacy
Curated by ChEMBL
| Assay Description
Inhibition of 5HT transporter (unknown origin) |
Eur J Med Chem 180: 562-612 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.019
BindingDB Entry DOI: 10.7270/Q2K93BWX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50150105

(CHEMBL124069 | [2-(6-Fluoro-chroman-8-yloxy)-ethyl...)Show SMILES Fc1cc2CCCOc2c(OCCNCCCc2c[nH]c3ccc(F)cc23)c1 Show InChI InChI=1S/C22H24F2N2O2/c23-17-5-6-20-19(12-17)16(14-26-20)3-1-7-25-8-10-27-21-13-18(24)11-15-4-2-9-28-22(15)21/h5-6,11-14,25-26H,1-4,7-10H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ISF College of Pharmacy
Curated by ChEMBL
| Assay Description
Agonist activity at 5HT1A receptor (unknown origin) |
Eur J Med Chem 180: 562-612 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.019
BindingDB Entry DOI: 10.7270/Q2K93BWX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50150093

(CHEMBL340873 | [2-(6-Fluoro-chroman-8-yloxy)-ethyl...)Show InChI InChI=1S/C21H22F2N2O2/c22-16-3-4-19-18(11-16)15(13-25-19)5-6-24-7-9-26-20-12-17(23)10-14-2-1-8-27-21(14)20/h3-4,10-13,24-25H,1-2,5-9H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ISF College of Pharmacy
Curated by ChEMBL
| Assay Description
Agonist activity at 5HT1A receptor (unknown origin) |
Eur J Med Chem 180: 562-612 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.019
BindingDB Entry DOI: 10.7270/Q2K93BWX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM79214
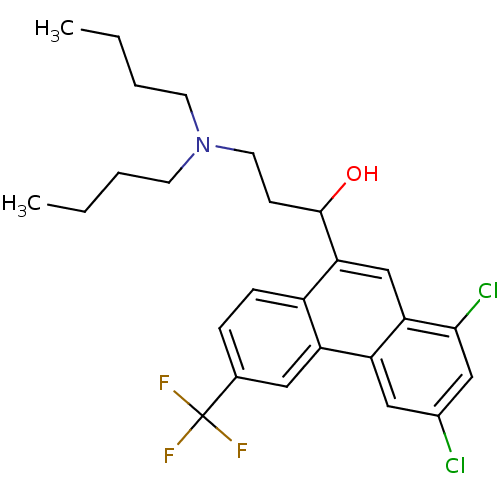
(1-[1,3-bis(chloranyl)-6-(trifluoromethyl)phenanthr...)Show SMILES CCCCN(CCCC)CCC(O)c1cc2c(Cl)cc(Cl)cc2c2cc(ccc12)C(F)(F)F Show InChI InChI=1S/C26H30Cl2F3NO/c1-3-5-10-32(11-6-4-2)12-9-25(33)23-16-22-21(14-18(27)15-24(22)28)20-13-17(26(29,30)31)7-8-19(20)23/h7-8,13-16,25,33H,3-6,9-12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human cloned ERG |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50247629
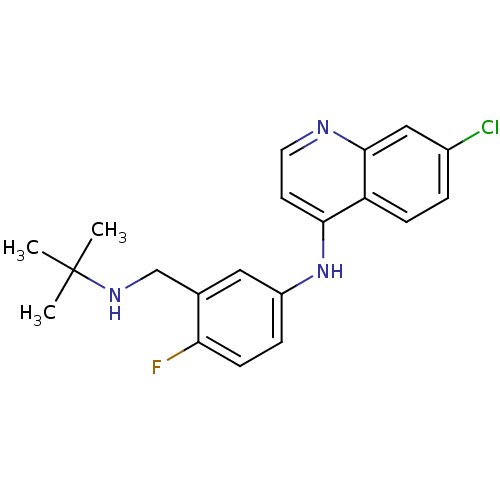
(CHEMBL453384 | N-(3-((tert-butylamino)methyl)-4-fl...)Show InChI InChI=1S/C20H21ClFN3/c1-20(2,3)24-12-13-10-15(5-7-17(13)22)25-18-8-9-23-19-11-14(21)4-6-16(18)19/h4-11,24H,12H2,1-3H3,(H,23,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2D6 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Reverse transcriptase
(Human immunodeficiency virus 1) | BDBM50516934

(CHEMBL4468611)Show SMILES CCCCC(O)(c1c(NC(=O)OCC)[nH]c2ccc(Br)cc12)C(F)(F)F Show InChI InChI=1S/C17H20BrF3N2O3/c1-3-5-8-16(25,17(19,20)21)13-11-9-10(18)6-7-12(11)22-14(13)23-15(24)26-4-2/h6-7,9,22,25H,3-5,8H2,1-2H3,(H,23,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.93E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ISF College of Pharmacy
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 3B Y181C mutant infected in human MT2 cells assessed as reduction in virus-induced cytopathogenicity after 3 days by BrightGlo luc... |
Eur J Med Chem 180: 562-612 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.019
BindingDB Entry DOI: 10.7270/Q2K93BWX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50041457

(4-[(7-chloroquinolin-4-yl)amino]-2-[(diethylamino)...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human cloned ERG |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM22985
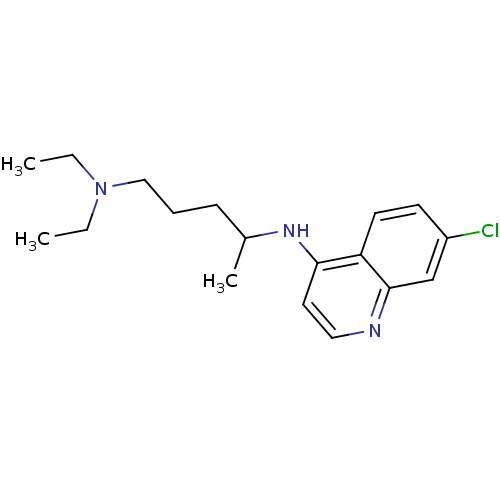
(Aralen | CHEMBL76 | CHLOROQUINE PHOSPHATE | Chloro...)Show InChI InChI=1S/C18H26ClN3/c1-4-22(5-2)12-6-7-14(3)21-17-10-11-20-18-13-15(19)8-9-16(17)18/h8-11,13-14H,4-7,12H2,1-3H3,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human cloned ERG |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50041457

(4-[(7-chloroquinolin-4-yl)amino]-2-[(diethylamino)...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2C8 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50041457

(4-[(7-chloroquinolin-4-yl)amino]-2-[(diethylamino)...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2C8 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50134936
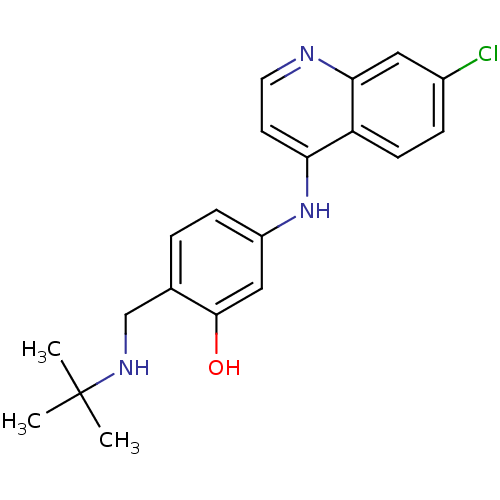
(2-(tert-Butylamino-methyl)-5-(7-chloro-quinolin-4-...)Show InChI InChI=1S/C20H22ClN3O/c1-20(2,3)23-12-13-4-6-15(11-19(13)25)24-17-8-9-22-18-10-14(21)5-7-16(17)18/h4-11,23,25H,12H2,1-3H3,(H,22,24) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2D6 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50134934
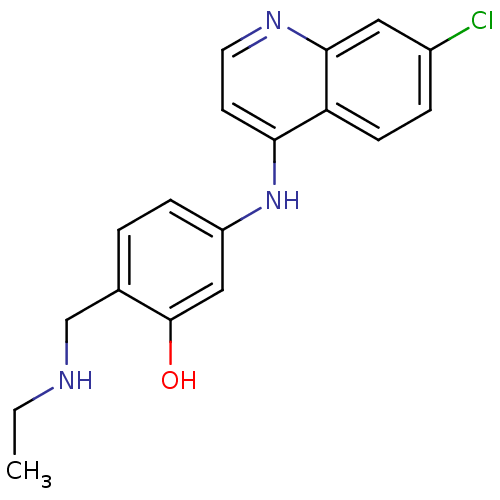
(5-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-3-5-14(10-18(12)23)22-16-7-8-21-17-9-13(19)4-6-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2D6 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50134931
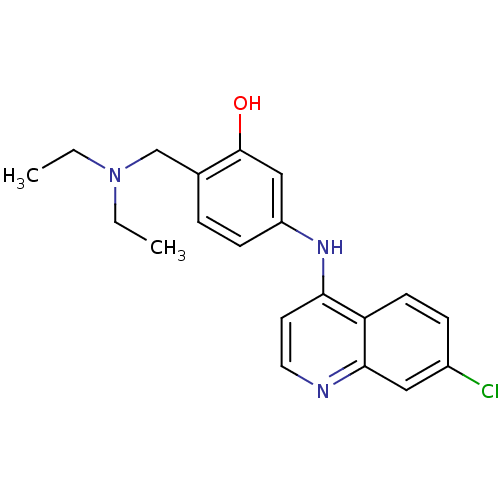
(5-(7-Chloro-quinolin-4-ylamino)-2-diethylaminometh...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-5-7-16(12-20(14)25)23-18-9-10-22-19-11-15(21)6-8-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human cloned ERG |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50041457

(4-[(7-chloroquinolin-4-yl)amino]-2-[(diethylamino)...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2D6 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50041457

(4-[(7-chloroquinolin-4-yl)amino]-2-[(diethylamino)...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2D6 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50516933

(CHEMBL4555803)Show InChI InChI=1S/C12H11NO/c1-3-5-9-8-13(2)12-10(9)6-4-7-11(12)14/h1,4,6-8,14H,5H2,2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.22E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ISF College of Pharmacy
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human COX2 assessed as decrease in PGE2 release after 10 mins by EIA |
Eur J Med Chem 180: 562-612 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.019
BindingDB Entry DOI: 10.7270/Q2K93BWX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50056190
(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2D6 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2D6 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50134931

(5-(7-Chloro-quinolin-4-ylamino)-2-diethylaminometh...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-5-7-16(12-20(14)25)23-18-9-10-22-19-11-15(21)6-8-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2D6 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50134931

(5-(7-Chloro-quinolin-4-ylamino)-2-diethylaminometh...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-5-7-16(12-20(14)25)23-18-9-10-22-19-11-15(21)6-8-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2D6 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50134936

(2-(tert-Butylamino-methyl)-5-(7-chloro-quinolin-4-...)Show InChI InChI=1S/C20H22ClN3O/c1-20(2,3)23-12-13-4-6-15(11-19(13)25)24-17-8-9-22-18-10-14(21)5-7-16(17)18/h4-11,23,25H,12H2,1-3H3,(H,22,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human cloned ERG |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50134934

(5-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-3-5-14(10-18(12)23)22-16-7-8-21-17-9-13(19)4-6-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP1A2 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50247629

(CHEMBL453384 | N-(3-((tert-butylamino)methyl)-4-fl...)Show InChI InChI=1S/C20H21ClFN3/c1-20(2,3)24-12-13-10-15(5-7-17(13)22)25-18-8-9-23-19-11-14(21)4-6-16(18)19/h4-11,24H,12H2,1-3H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP1A2 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human cloned ERG |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50247629

(CHEMBL453384 | N-(3-((tert-butylamino)methyl)-4-fl...)Show InChI InChI=1S/C20H21ClFN3/c1-20(2,3)24-12-13-10-15(5-7-17(13)22)25-18-8-9-23-19-11-14(21)4-6-16(18)19/h4-11,24H,12H2,1-3H3,(H,23,25) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2C8 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Reverse transcriptase
(Human immunodeficiency virus 1) | BDBM50516932

(CHEMBL1652111 | IDX899)Show SMILES COP(=O)(c1c([nH]c2ccc(Cl)cc12)C(N)=O)c1cc(C)cc(\C=C\C#N)c1 Show InChI InChI=1S/C20H17ClN3O3P/c1-12-8-13(4-3-7-22)10-15(9-12)28(26,27-2)19-16-11-14(21)5-6-17(16)24-18(19)20(23)25/h3-6,8-11,24H,1-2H3,(H2,23,25)/b4-3+ | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ISF College of Pharmacy
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 reverse transcriptase Y181C/K103N double mutant infected in human MT4 cells assessed as reduction in virus-induced cytopathogenici... |
Eur J Med Chem 180: 562-612 (2019)
Article DOI: 10.1016/j.ejmech.2019.07.019
BindingDB Entry DOI: 10.7270/Q2K93BWX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50134931

(5-(7-Chloro-quinolin-4-ylamino)-2-diethylaminometh...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-5-7-16(12-20(14)25)23-18-9-10-22-19-11-15(21)6-8-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2C8 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50134931

(5-(7-Chloro-quinolin-4-ylamino)-2-diethylaminometh...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-5-7-16(12-20(14)25)23-18-9-10-22-19-11-15(21)6-8-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2C8 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50041457

(4-[(7-chloroquinolin-4-yl)amino]-2-[(diethylamino)...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP1A2 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50041457

(4-[(7-chloroquinolin-4-yl)amino]-2-[(diethylamino)...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP1A2 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2C8 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2C8 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50134931

(5-(7-Chloro-quinolin-4-ylamino)-2-diethylaminometh...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-5-7-16(12-20(14)25)23-18-9-10-22-19-11-15(21)6-8-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP1A2 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50134931

(5-(7-Chloro-quinolin-4-ylamino)-2-diethylaminometh...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-5-7-16(12-20(14)25)23-18-9-10-22-19-11-15(21)6-8-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP1A2 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50134934

(5-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-3-5-14(10-18(12)23)22-16-7-8-21-17-9-13(19)4-6-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2C8 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50134936

(2-(tert-Butylamino-methyl)-5-(7-chloro-quinolin-4-...)Show InChI InChI=1S/C20H22ClN3O/c1-20(2,3)23-12-13-4-6-15(11-19(13)25)24-17-8-9-22-18-10-14(21)5-7-16(17)18/h4-11,23,25H,12H2,1-3H3,(H,22,24) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2C8 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP3A4 using DEF substrate |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP3A4 using N-N,diethyl-formamide as substrate |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50134936

(2-(tert-Butylamino-methyl)-5-(7-chloro-quinolin-4-...)Show InChI InChI=1S/C20H22ClN3O/c1-20(2,3)23-12-13-4-6-15(11-19(13)25)24-17-8-9-22-18-10-14(21)5-7-16(17)18/h4-11,23,25H,12H2,1-3H3,(H,22,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2C19 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP1A2 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50041457

(4-[(7-chloroquinolin-4-yl)amino]-2-[(diethylamino)...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP3A4 using DEF substrate |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP1A2 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50041457

(4-[(7-chloroquinolin-4-yl)amino]-2-[(diethylamino)...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP3A4 using N-N,diethyl-formamide as substrate |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50134936

(2-(tert-Butylamino-methyl)-5-(7-chloro-quinolin-4-...)Show InChI InChI=1S/C20H22ClN3O/c1-20(2,3)23-12-13-4-6-15(11-19(13)25)24-17-8-9-22-18-10-14(21)5-7-16(17)18/h4-11,23,25H,12H2,1-3H3,(H,22,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP1A2 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50134931

(5-(7-Chloro-quinolin-4-ylamino)-2-diethylaminometh...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-5-7-16(12-20(14)25)23-18-9-10-22-19-11-15(21)6-8-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2C19 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50134931

(5-(7-Chloro-quinolin-4-ylamino)-2-diethylaminometh...)Show InChI InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-5-7-16(12-20(14)25)23-18-9-10-22-19-11-15(21)6-8-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2C19 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50247629

(CHEMBL453384 | N-(3-((tert-butylamino)methyl)-4-fl...)Show InChI InChI=1S/C20H21ClFN3/c1-20(2,3)24-12-13-10-15(5-7-17(13)22)25-18-8-9-23-19-11-14(21)4-6-16(18)19/h4-11,24H,12H2,1-3H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2C9 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2C19 |
J Med Chem 52: 1828-44 (2009)
Article DOI: 10.1021/jm8012757
BindingDB Entry DOI: 10.7270/Q21Z45C3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50056190

(4-(7-Chloro-quinolin-4-ylamino)-2-ethylaminomethyl...)Show InChI InChI=1S/C18H18ClN3O/c1-2-20-11-12-9-14(4-6-18(12)23)22-16-7-8-21-17-10-13(19)3-5-15(16)17/h3-10,20,23H,2,11H2,1H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Liverpool
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CYP2C19 |
J Med Chem 52: 1408-15 (2010)
Article DOI: 10.1021/jm8012618
BindingDB Entry DOI: 10.7270/Q2348KCX |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data