 Found 133 hits with Last Name = 'bhattachar' and Initial = 'sn'
Found 133 hits with Last Name = 'bhattachar' and Initial = 'sn' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50028854
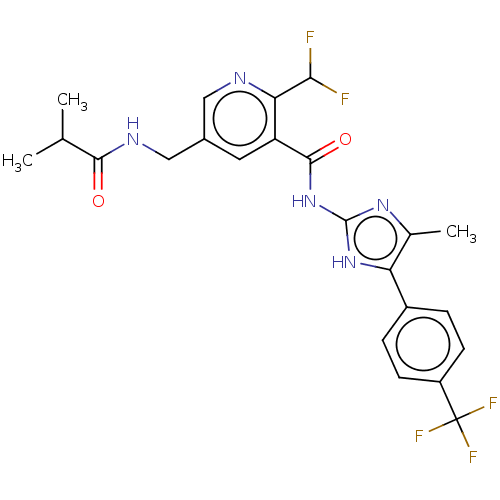
(CHEMBL3342693)Show SMILES CC(C)C(=O)NCc1cnc(C(F)F)c(c1)C(=O)Nc1nc(C)c([nH]1)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C23H22F5N5O2/c1-11(2)20(34)30-10-13-8-16(18(19(24)25)29-9-13)21(35)33-22-31-12(3)17(32-22)14-4-6-15(7-5-14)23(26,27)28/h4-9,11,19H,10H2,1-3H3,(H,30,34)(H2,31,32,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.13E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50028854

(CHEMBL3342693)Show SMILES CC(C)C(=O)NCc1cnc(C(F)F)c(c1)C(=O)Nc1nc(C)c([nH]1)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C23H22F5N5O2/c1-11(2)20(34)30-10-13-8-16(18(19(24)25)29-9-13)21(35)33-22-31-12(3)17(32-22)14-4-6-15(7-5-14)23(26,27)28/h4-9,11,19H,10H2,1-3H3,(H,30,34)(H2,31,32,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human microsomal PGES1 expressed in 293E cells by LC/MS/MS analysis |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233283

(CHEMBL4102262)Show SMILES OC(=O)c1ccc(Cl)cc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C27H30ClF3N2O3/c28-21-8-11-23(26(35)36)19(16-21)5-4-17-2-1-3-24(17)32-25(34)18-12-14-33(15-13-18)22-9-6-20(7-10-22)27(29,30)31/h6-11,16-18,24H,1-5,12-15H2,(H,32,34)(H,35,36)/t17-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233283

(CHEMBL4102262)Show SMILES OC(=O)c1ccc(Cl)cc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C27H30ClF3N2O3/c28-21-8-11-23(26(35)36)19(16-21)5-4-17-2-1-3-24(17)32-25(34)18-12-14-33(15-13-18)22-9-6-20(7-10-22)27(29,30)31/h6-11,16-18,24H,1-5,12-15H2,(H,32,34)(H,35,36)/t17-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233285

(CHEMBL4101413)Show SMILES Cc1cccc2ccc(nc12)N1CCC(CC1)C(=O)N[C@H]1CCC[C@H]1CCc1ccccc1C(O)=O |r| Show InChI InChI=1S/C30H35N3O3/c1-20-6-4-9-23-14-15-27(32-28(20)23)33-18-16-24(17-19-33)29(34)31-26-11-5-8-22(26)13-12-21-7-2-3-10-25(21)30(35)36/h2-4,6-7,9-10,14-15,22,24,26H,5,8,11-13,16-19H2,1H3,(H,31,34)(H,35,36)/t22-,26-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233281

(CHEMBL4084995)Show SMILES OC(=O)c1ccccc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(OC(F)(F)F)cc1 |r| Show InChI InChI=1S/C27H31F3N2O4/c28-27(29,30)36-22-12-10-21(11-13-22)32-16-14-20(15-17-32)25(33)31-24-7-3-5-19(24)9-8-18-4-1-2-6-23(18)26(34)35/h1-2,4,6,10-13,19-20,24H,3,5,7-9,14-17H2,(H,31,33)(H,34,35)/t19-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233278

(CHEMBL4064335)Show SMILES OC(=O)c1ccc(F)cc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C27H30F4N2O3/c28-21-8-11-23(26(35)36)19(16-21)5-4-17-2-1-3-24(17)32-25(34)18-12-14-33(15-13-18)22-9-6-20(7-10-22)27(29,30)31/h6-11,16-18,24H,1-5,12-15H2,(H,32,34)(H,35,36)/t17-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233277

(CHEMBL4085873)Show SMILES Cc1ccc(C(O)=O)c(CC[C@@H]2CCC[C@@H]2NC(=O)C2CCN(CC2)c2ccc(cc2)C(F)(F)F)c1 |r| Show InChI InChI=1S/C28H33F3N2O3/c1-18-5-12-24(27(35)36)21(17-18)7-6-19-3-2-4-25(19)32-26(34)20-13-15-33(16-14-20)23-10-8-22(9-11-23)28(29,30)31/h5,8-12,17,19-20,25H,2-4,6-7,13-16H2,1H3,(H,32,34)(H,35,36)/t19-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194190
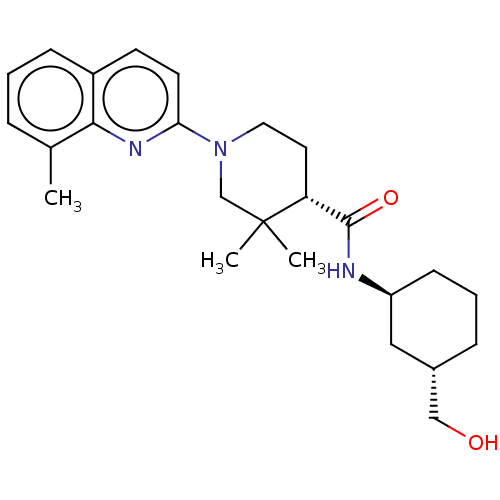
(CHEMBL3956184)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@H]2CCC[C@H](CO)C2)C(C)(C)C1 |r| Show InChI InChI=1S/C25H35N3O2/c1-17-6-4-8-19-10-11-22(27-23(17)19)28-13-12-21(25(2,3)16-28)24(30)26-20-9-5-7-18(14-20)15-29/h4,6,8,10-11,18,20-21,29H,5,7,9,12-16H2,1-3H3,(H,26,30)/t18-,20-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194190

(CHEMBL3956184)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@H]2CCC[C@H](CO)C2)C(C)(C)C1 |r| Show InChI InChI=1S/C25H35N3O2/c1-17-6-4-8-19-10-11-22(27-23(17)19)28-13-12-21(25(2,3)16-28)24(30)26-20-9-5-7-18(14-20)15-29/h4,6,8,10-11,18,20-21,29H,5,7,9,12-16H2,1-3H3,(H,26,30)/t18-,20-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES-1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS stimulation ... |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194141

(CHEMBL3938686)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@@H]2CC[C@H](CO)CC2)C(C)(C)C1 |r,wU:14.16,18.19,21.23,(40.97,-19.87,;39.64,-20.64,;39.65,-22.18,;38.31,-22.96,;36.97,-22.19,;36.97,-20.65,;35.63,-19.88,;35.63,-18.35,;36.97,-17.58,;38.3,-18.34,;38.3,-19.88,;36.97,-16.05,;38.3,-15.28,;38.3,-13.74,;36.97,-12.97,;36.97,-11.43,;38.3,-10.66,;35.63,-10.66,;35.63,-9.12,;36.97,-8.34,;36.98,-6.81,;35.65,-6.03,;35.65,-4.49,;36.99,-3.72,;34.31,-6.8,;34.3,-8.34,;35.64,-13.74,;34.14,-14.14,;34.55,-12.65,;35.64,-15.28,)| Show InChI InChI=1S/C25H35N3O2/c1-17-5-4-6-19-9-12-22(27-23(17)19)28-14-13-21(25(2,3)16-28)24(30)26-20-10-7-18(15-29)8-11-20/h4-6,9,12,18,20-21,29H,7-8,10-11,13-16H2,1-3H3,(H,26,30)/t18-,20+,21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233284
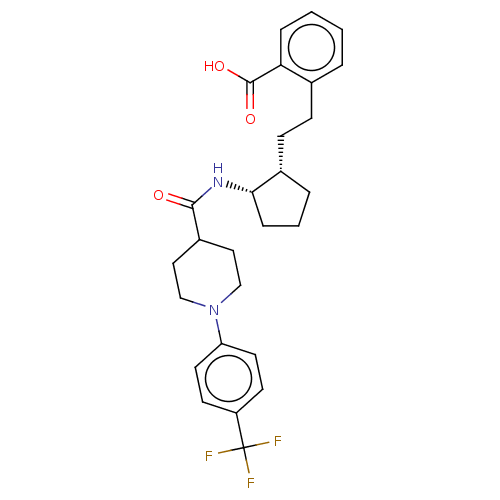
(CHEMBL4071976)Show SMILES OC(=O)c1ccccc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C27H31F3N2O3/c28-27(29,30)21-10-12-22(13-11-21)32-16-14-20(15-17-32)25(33)31-24-7-3-5-19(24)9-8-18-4-1-2-6-23(18)26(34)35/h1-2,4,6,10-13,19-20,24H,3,5,7-9,14-17H2,(H,31,33)(H,34,35)/t19-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233280

(CHEMBL4063350)Show SMILES OC(=O)c1ccccc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cn1)C(F)(F)F |r| Show InChI InChI=1S/C26H30F3N3O3/c27-26(28,29)20-10-11-23(30-16-20)32-14-12-19(13-15-32)24(33)31-22-7-3-5-18(22)9-8-17-4-1-2-6-21(17)25(34)35/h1-2,4,6,10-11,16,18-19,22H,3,5,7-9,12-15H2,(H,31,33)(H,34,35)/t18-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194141

(CHEMBL3938686)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@@H]2CC[C@H](CO)CC2)C(C)(C)C1 |r,wU:14.16,18.19,21.23,(40.97,-19.87,;39.64,-20.64,;39.65,-22.18,;38.31,-22.96,;36.97,-22.19,;36.97,-20.65,;35.63,-19.88,;35.63,-18.35,;36.97,-17.58,;38.3,-18.34,;38.3,-19.88,;36.97,-16.05,;38.3,-15.28,;38.3,-13.74,;36.97,-12.97,;36.97,-11.43,;38.3,-10.66,;35.63,-10.66,;35.63,-9.12,;36.97,-8.34,;36.98,-6.81,;35.65,-6.03,;35.65,-4.49,;36.99,-3.72,;34.31,-6.8,;34.3,-8.34,;35.64,-13.74,;34.14,-14.14,;34.55,-12.65,;35.64,-15.28,)| Show InChI InChI=1S/C25H35N3O2/c1-17-5-4-6-19-9-12-22(27-23(17)19)28-14-13-21(25(2,3)16-28)24(30)26-20-10-7-18(15-29)8-11-20/h4-6,9,12,18,20-21,29H,7-8,10-11,13-16H2,1-3H3,(H,26,30)/t18-,20+,21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES-1 in human A549 cells assessed as reduction in recombinant human interleukin-1 beta-induced PGE2 production preincubated for 30 m... |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194138
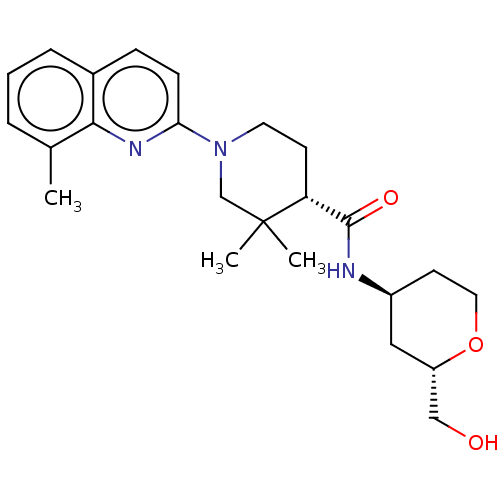
(CHEMBL3928608)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@H]2CCO[C@H](CO)C2)C(C)(C)C1 |r| Show InChI InChI=1S/C24H33N3O3/c1-16-5-4-6-17-7-8-21(26-22(16)17)27-11-9-20(24(2,3)15-27)23(29)25-18-10-12-30-19(13-18)14-28/h4-8,18-20,28H,9-15H2,1-3H3,(H,25,29)/t18-,19-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233276

(CHEMBL4078000)Show SMILES OC(=O)c1ccc(Cl)cc1OC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C26H28ClF3N2O4/c27-19-6-9-21(25(34)35)23(14-19)36-15-17-2-1-3-22(17)31-24(33)16-10-12-32(13-11-16)20-7-4-18(5-8-20)26(28,29)30/h4-9,14,16-17,22H,1-3,10-13,15H2,(H,31,33)(H,34,35)/t17-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233274
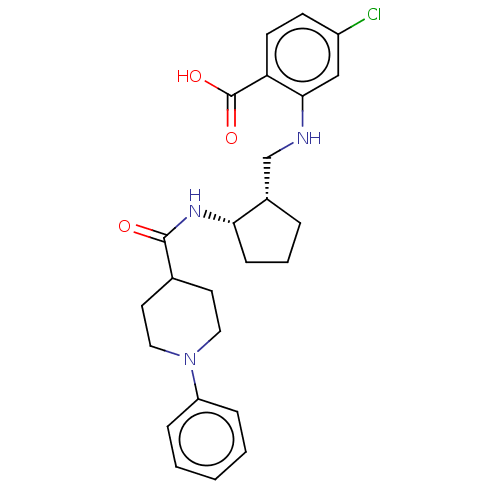
(CHEMBL4067045)Show SMILES OC(=O)c1ccc(Cl)cc1NC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccccc1 |r| Show InChI InChI=1S/C25H30ClN3O3/c26-19-9-10-21(25(31)32)23(15-19)27-16-18-5-4-8-22(18)28-24(30)17-11-13-29(14-12-17)20-6-2-1-3-7-20/h1-3,6-7,9-10,15,17-18,22,27H,4-5,8,11-14,16H2,(H,28,30)(H,31,32)/t18-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194141

(CHEMBL3938686)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@@H]2CC[C@H](CO)CC2)C(C)(C)C1 |r,wU:14.16,18.19,21.23,(40.97,-19.87,;39.64,-20.64,;39.65,-22.18,;38.31,-22.96,;36.97,-22.19,;36.97,-20.65,;35.63,-19.88,;35.63,-18.35,;36.97,-17.58,;38.3,-18.34,;38.3,-19.88,;36.97,-16.05,;38.3,-15.28,;38.3,-13.74,;36.97,-12.97,;36.97,-11.43,;38.3,-10.66,;35.63,-10.66,;35.63,-9.12,;36.97,-8.34,;36.98,-6.81,;35.65,-6.03,;35.65,-4.49,;36.99,-3.72,;34.31,-6.8,;34.3,-8.34,;35.64,-13.74,;34.14,-14.14,;34.55,-12.65,;35.64,-15.28,)| Show InChI InChI=1S/C25H35N3O2/c1-17-5-4-6-19-9-12-22(27-23(17)19)28-14-13-21(25(2,3)16-28)24(30)26-20-10-7-18(15-29)8-11-20/h4-6,9,12,18,20-21,29H,7-8,10-11,13-16H2,1-3H3,(H,26,30)/t18-,20+,21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES-1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS stimulation ... |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233277

(CHEMBL4085873)Show SMILES Cc1ccc(C(O)=O)c(CC[C@@H]2CCC[C@@H]2NC(=O)C2CCN(CC2)c2ccc(cc2)C(F)(F)F)c1 |r| Show InChI InChI=1S/C28H33F3N2O3/c1-18-5-12-24(27(35)36)21(17-18)7-6-19-3-2-4-25(19)32-26(34)20-13-15-33(16-14-20)23-10-8-22(9-11-23)28(29,30)31/h5,8-12,17,19-20,25H,2-4,6-7,13-16H2,1H3,(H,32,34)(H,35,36)/t19-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194190

(CHEMBL3956184)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@H]2CCC[C@H](CO)C2)C(C)(C)C1 |r| Show InChI InChI=1S/C25H35N3O2/c1-17-6-4-8-19-10-11-22(27-23(17)19)28-13-12-21(25(2,3)16-28)24(30)26-20-9-5-7-18(14-20)15-29/h4,6,8,10-11,18,20-21,29H,5,7,9,12-16H2,1-3H3,(H,26,30)/t18-,20-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES-1 in human A549 cells assessed as reduction in recombinant human interleukin-1 beta-induced PGE2 production preincubated for 30 m... |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194140
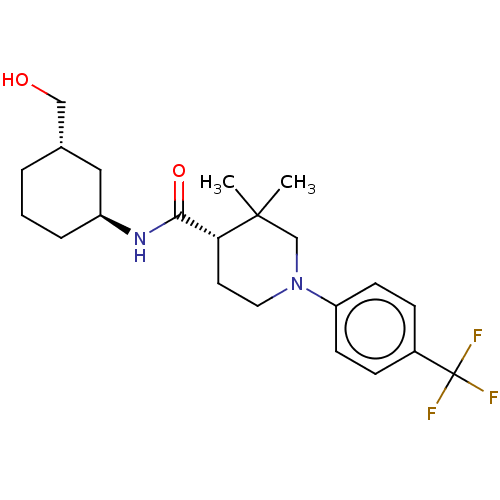
(CHEMBL3947494)Show SMILES CC1(C)CN(CC[C@@H]1C(=O)N[C@H]1CCC[C@H](CO)C1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C22H31F3N2O2/c1-21(2)14-27(18-8-6-16(7-9-18)22(23,24)25)11-10-19(21)20(29)26-17-5-3-4-15(12-17)13-28/h6-9,15,17,19,28H,3-5,10-14H2,1-2H3,(H,26,29)/t15-,17-,19+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50142252

(CHEMBL3758953)Show SMILES CC(C)C(=O)NCc1ccc(C(F)F)c(c1)C(=O)Nc1ncc([nH]1)-c1ccc(Cl)cc1 Show InChI InChI=1S/C22H21ClF2N4O2/c1-12(2)20(30)26-10-13-3-8-16(19(24)25)17(9-13)21(31)29-22-27-11-18(28-22)14-4-6-15(23)7-5-14/h3-9,11-12,19H,10H2,1-2H3,(H,26,30)(H2,27,28,29,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human microsomal PGES1 expressed in 293E cells by LC/MS/MS analysis |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50142253
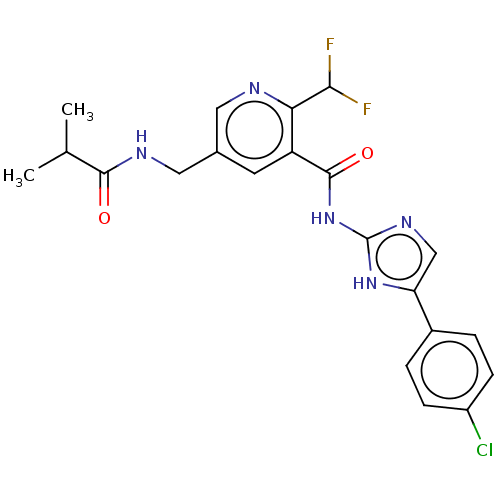
(CHEMBL3758924)Show SMILES CC(C)C(=O)NCc1cnc(C(F)F)c(c1)C(=O)Nc1ncc([nH]1)-c1ccc(Cl)cc1 Show InChI InChI=1S/C21H20ClF2N5O2/c1-11(2)19(30)26-9-12-7-15(17(18(23)24)25-8-12)20(31)29-21-27-10-16(28-21)13-3-5-14(22)6-4-13/h3-8,10-11,18H,9H2,1-2H3,(H,26,30)(H2,27,28,29,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human microsomal PGES1 expressed in 293E cells by LC/MS/MS analysis |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50142254
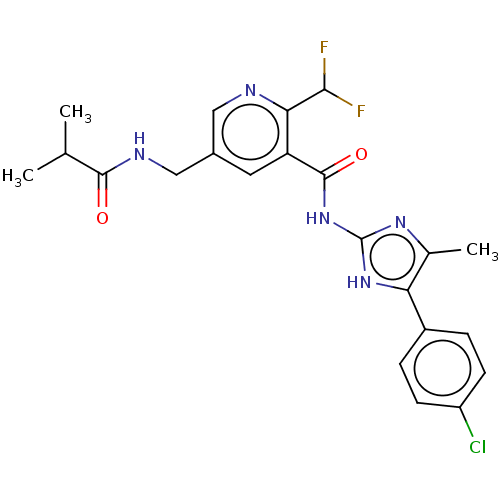
(CHEMBL3759288)Show SMILES CC(C)C(=O)NCc1cnc(C(F)F)c(c1)C(=O)Nc1nc(C)c([nH]1)-c1ccc(Cl)cc1 Show InChI InChI=1S/C22H22ClF2N5O2/c1-11(2)20(31)27-10-13-8-16(18(19(24)25)26-9-13)21(32)30-22-28-12(3)17(29-22)14-4-6-15(23)7-5-14/h4-9,11,19H,10H2,1-3H3,(H,27,31)(H2,28,29,30,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human microsomal PGES1 expressed in 293E cells by LC/MS/MS analysis |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233275
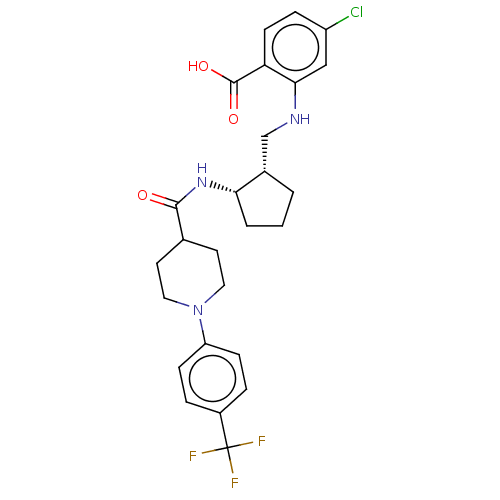
(CHEMBL4094518)Show SMILES OC(=O)c1ccc(Cl)cc1NC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C26H29ClF3N3O3/c27-19-6-9-21(25(35)36)23(14-19)31-15-17-2-1-3-22(17)32-24(34)16-10-12-33(13-11-16)20-7-4-18(5-8-20)26(28,29)30/h4-9,14,16-17,22,31H,1-3,10-13,15H2,(H,32,34)(H,35,36)/t17-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233275

(CHEMBL4094518)Show SMILES OC(=O)c1ccc(Cl)cc1NC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C26H29ClF3N3O3/c27-19-6-9-21(25(35)36)23(14-19)31-15-17-2-1-3-22(17)32-24(34)16-10-12-33(13-11-16)20-7-4-18(5-8-20)26(28,29)30/h4-9,14,16-17,22,31H,1-3,10-13,15H2,(H,32,34)(H,35,36)/t17-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in human A549 cells assessed as reduction in IL-1beta-induced PGE2 production preincubated for 30 mins followed by IL-1beta addi... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194137

(CHEMBL3922684)Show SMILES CC1(C)CN(CC[C@@H]1C(=O)N[C@@H]1CC[C@H](CO)CC1)c1ccc2cccc(Cl)c2n1 |r,wU:7.8,11.11,14.15,(.86,-13.35,;2.35,-12.95,;1.26,-11.86,;2.35,-14.49,;3.68,-15.25,;5,-14.49,;5,-12.95,;3.68,-12.17,;3.68,-10.63,;5.01,-9.87,;2.34,-9.87,;2.34,-8.33,;3.68,-7.55,;3.69,-6.02,;2.36,-5.24,;2.36,-3.7,;1.03,-2.93,;1.02,-6.01,;1.02,-7.55,;3.68,-16.79,;2.34,-17.56,;2.34,-19.1,;3.67,-19.87,;3.68,-21.41,;5.02,-22.18,;6.36,-21.39,;6.35,-19.85,;7.67,-19.07,;5.01,-19.09,;5,-17.55,)| Show InChI InChI=1S/C24H32ClN3O2/c1-24(2)15-28(21-11-8-17-4-3-5-20(25)22(17)27-21)13-12-19(24)23(30)26-18-9-6-16(14-29)7-10-18/h3-5,8,11,16,18-19,29H,6-7,9-10,12-15H2,1-2H3,(H,26,30)/t16-,18+,19-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233275

(CHEMBL4094518)Show SMILES OC(=O)c1ccc(Cl)cc1NC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C26H29ClF3N3O3/c27-19-6-9-21(25(35)36)23(14-19)31-15-17-2-1-3-22(17)32-24(34)16-10-12-33(13-11-16)20-7-4-18(5-8-20)26(28,29)30/h4-9,14,16-17,22,31H,1-3,10-13,15H2,(H,32,34)(H,35,36)/t17-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233284

(CHEMBL4071976)Show SMILES OC(=O)c1ccccc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C27H31F3N2O3/c28-27(29,30)21-10-12-22(13-11-21)32-16-14-20(15-17-32)25(33)31-24-7-3-5-19(24)9-8-18-4-1-2-6-23(18)26(34)35/h1-2,4,6,10-13,19-20,24H,3,5,7-9,14-17H2,(H,31,33)(H,34,35)/t19-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194138

(CHEMBL3928608)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@H]2CCO[C@H](CO)C2)C(C)(C)C1 |r| Show InChI InChI=1S/C24H33N3O3/c1-16-5-4-6-17-7-8-21(26-22(16)17)27-11-9-20(24(2,3)15-27)23(29)25-18-10-12-30-19(13-18)14-28/h4-8,18-20,28H,9-15H2,1-3H3,(H,25,29)/t18-,19-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES-1 in human A549 cells assessed as reduction in recombinant human interleukin-1 beta-induced PGE2 production preincubated for 30 m... |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194140

(CHEMBL3947494)Show SMILES CC1(C)CN(CC[C@@H]1C(=O)N[C@H]1CCC[C@H](CO)C1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C22H31F3N2O2/c1-21(2)14-27(18-8-6-16(7-9-18)22(23,24)25)11-10-19(21)20(29)26-17-5-3-4-15(12-17)13-28/h6-9,15,17,19,28H,3-5,10-14H2,1-2H3,(H,26,29)/t15-,17-,19+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES-1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS stimulation ... |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50227631
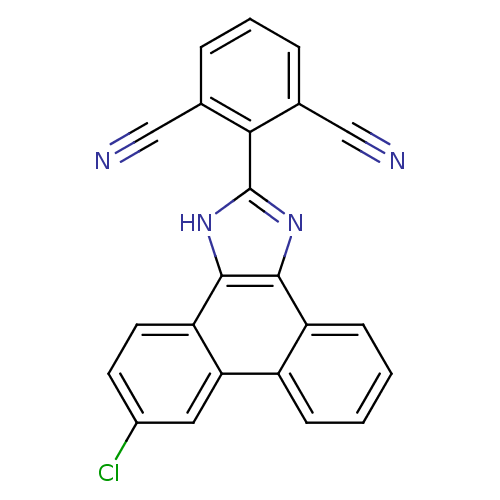
(2-(6-chloro-1H-phenanthro[9,10-d]imidazol-2-yl)iso...)Show SMILES Clc1ccc2c3[nH]c(nc3c3ccccc3c2c1)-c1c(cccc1C#N)C#N Show InChI InChI=1S/C23H11ClN4/c24-15-8-9-18-19(10-15)16-6-1-2-7-17(16)21-22(18)28-23(27-21)20-13(11-25)4-3-5-14(20)12-26/h1-10H,(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human microsomal PGES1 expressed in 293E cells by LC/MS/MS analysis |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233285

(CHEMBL4101413)Show SMILES Cc1cccc2ccc(nc12)N1CCC(CC1)C(=O)N[C@H]1CCC[C@H]1CCc1ccccc1C(O)=O |r| Show InChI InChI=1S/C30H35N3O3/c1-20-6-4-9-23-14-15-27(32-28(20)23)33-18-16-24(17-19-33)29(34)31-26-11-5-8-22(26)13-12-21-7-2-3-10-25(21)30(35)36/h2-4,6-7,9-10,14-15,22,24,26H,5,8,11-13,16-19H2,1H3,(H,31,34)(H,35,36)/t22-,26-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233278

(CHEMBL4064335)Show SMILES OC(=O)c1ccc(F)cc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C27H30F4N2O3/c28-21-8-11-23(26(35)36)19(16-21)5-4-17-2-1-3-24(17)32-25(34)18-12-14-33(15-13-18)22-9-6-20(7-10-22)27(29,30)31/h6-11,16-18,24H,1-5,12-15H2,(H,32,34)(H,35,36)/t17-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233279

(CHEMBL4092750)Show SMILES OC(=O)c1cccnc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C26H30F3N3O3/c27-26(28,29)19-7-9-20(10-8-19)32-15-12-18(13-16-32)24(33)31-22-5-1-3-17(22)6-11-23-21(25(34)35)4-2-14-30-23/h2,4,7-10,14,17-18,22H,1,3,5-6,11-13,15-16H2,(H,31,33)(H,34,35)/t17-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194193
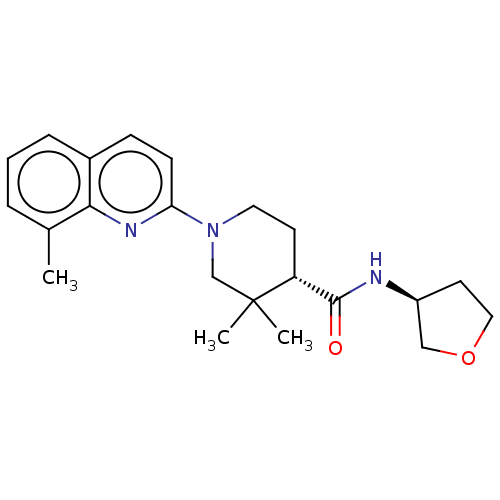
(CHEMBL3926051)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@H]2CCOC2)C(C)(C)C1 |r| Show InChI InChI=1S/C22H29N3O2/c1-15-5-4-6-16-7-8-19(24-20(15)16)25-11-9-18(22(2,3)14-25)21(26)23-17-10-12-27-13-17/h4-8,17-18H,9-14H2,1-3H3,(H,23,26)/t17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
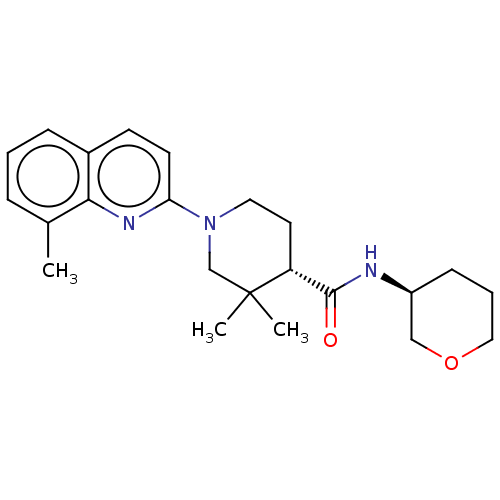
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194192
(CHEMBL3910746)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@H]2CCCOC2)C(C)(C)C1 |r| Show InChI InChI=1S/C23H31N3O2/c1-16-6-4-7-17-9-10-20(25-21(16)17)26-12-11-19(23(2,3)15-26)22(27)24-18-8-5-13-28-14-18/h4,6-7,9-10,18-19H,5,8,11-15H2,1-3H3,(H,24,27)/t18-,19+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194138

(CHEMBL3928608)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@H]2CCO[C@H](CO)C2)C(C)(C)C1 |r| Show InChI InChI=1S/C24H33N3O3/c1-16-5-4-6-17-7-8-21(26-22(16)17)27-11-9-20(24(2,3)15-27)23(29)25-18-10-12-30-19(13-18)14-28/h4-8,18-20,28H,9-15H2,1-3H3,(H,25,29)/t18-,19-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES-1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS stimulation ... |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Fatty-acid amide hydrolase 1
(Homo sapiens (Human)) | BDBM50335377

(CHEMBL1651534 | N-Pyridazin-3-yl-4-(3-{[5-(trifluo...)Show SMILES FC(F)(F)c1ccc(-[#8]-c2cccc(\[#6]=[#6]-3\[#6]-[#6]-[#7](-[#6]-[#6]-3)-[#6](=O)-[#7]-c3cccnn3)c2)nc1 Show InChI InChI=1S/C23H20F3N5O2/c24-23(25,26)18-6-7-21(27-15-18)33-19-4-1-3-17(14-19)13-16-8-11-31(12-9-16)22(32)29-20-5-2-10-28-30-20/h1-7,10,13-15H,8-9,11-12H2,(H,29,30,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of His-tagged human FAAH N-terminal transmembrane-deleted truncated form expressed in Escherichia coli preincubated for 60 mins before ole... |
ACS Med Chem Lett 2: 91-96 (2011)
Article DOI: 10.1021/ml100190t
BindingDB Entry DOI: 10.7270/Q25X29WM |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50335377

(CHEMBL1651534 | N-Pyridazin-3-yl-4-(3-{[5-(trifluo...)Show SMILES FC(F)(F)c1ccc(-[#8]-c2cccc(\[#6]=[#6]-3\[#6]-[#6]-[#7](-[#6]-[#6]-3)-[#6](=O)-[#7]-c3cccnn3)c2)nc1 Show InChI InChI=1S/C23H20F3N5O2/c24-23(25,26)18-6-7-21(27-15-18)33-19-4-1-3-17(14-19)13-16-8-11-31(12-9-16)22(32)29-20-5-2-10-28-30-20/h1-7,10,13-15H,8-9,11-12H2,(H,29,30,32) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Irreversible inhibition of His-tagged rat FAAH N-terminal transmembrane-deleted truncated form expressed in Escherichia coli preincubated for 60 mins... |
ACS Med Chem Lett 2: 91-96 (2011)
Article DOI: 10.1021/ml100190t
BindingDB Entry DOI: 10.7270/Q25X29WM |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233276

(CHEMBL4078000)Show SMILES OC(=O)c1ccc(Cl)cc1OC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C26H28ClF3N2O4/c27-19-6-9-21(25(34)35)23(14-19)36-15-17-2-1-3-22(17)31-24(33)16-10-12-32(13-11-16)20-7-4-18(5-8-20)26(28,29)30/h4-9,14,16-17,22H,1-3,10-13,15H2,(H,31,33)(H,34,35)/t17-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50233281

(CHEMBL4084995)Show SMILES OC(=O)c1ccccc1CC[C@@H]1CCC[C@@H]1NC(=O)C1CCN(CC1)c1ccc(OC(F)(F)F)cc1 |r| Show InChI InChI=1S/C27H31F3N2O4/c28-27(29,30)36-22-12-10-21(11-13-22)32-16-14-20(15-17-32)25(33)31-24-7-3-5-19(24)9-8-18-4-1-2-6-23(18)26(34)35/h1-2,4,6,10-13,19-20,24H,3,5,7-9,14-17H2,(H,31,33)(H,34,35)/t19-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... |
Bioorg Med Chem Lett 27: 1478-1483 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.011
BindingDB Entry DOI: 10.7270/Q28P62RZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50142255
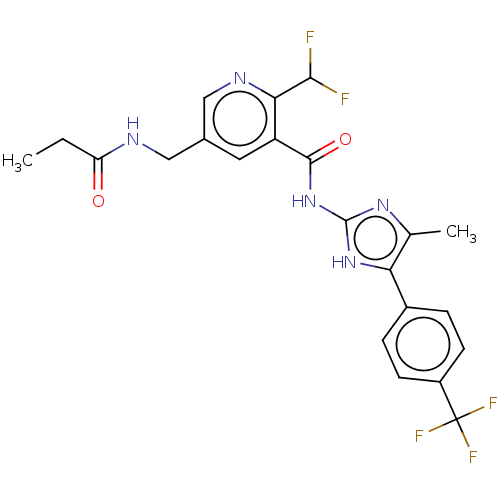
(CHEMBL3758222)Show SMILES CCC(=O)NCc1cnc(C(F)F)c(c1)C(=O)Nc1nc(C)c([nH]1)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C22H20F5N5O2/c1-3-16(33)28-9-12-8-15(18(19(23)24)29-10-12)20(34)32-21-30-11(2)17(31-21)13-4-6-14(7-5-13)22(25,26)27/h4-8,10,19H,3,9H2,1-2H3,(H,28,33)(H2,30,31,32,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human microsomal PGES1 expressed in 293E cells by LC/MS/MS analysis |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194140

(CHEMBL3947494)Show SMILES CC1(C)CN(CC[C@@H]1C(=O)N[C@H]1CCC[C@H](CO)C1)c1ccc(cc1)C(F)(F)F |r| Show InChI InChI=1S/C22H31F3N2O2/c1-21(2)14-27(18-8-6-16(7-9-18)22(23,24)25)11-10-19(21)20(29)26-17-5-3-4-15(12-17)13-28/h6-9,15,17,19,28H,3-5,10-14H2,1-2H3,(H,26,29)/t15-,17-,19+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES-1 in human A549 cells assessed as reduction in recombinant human interleukin-1 beta-induced PGE2 production preincubated for 30 m... |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Canis familiaris) | BDBM50194138

(CHEMBL3928608)Show SMILES Cc1cccc2ccc(nc12)N1CC[C@H](C(=O)N[C@H]2CCO[C@H](CO)C2)C(C)(C)C1 |r| Show InChI InChI=1S/C24H33N3O3/c1-16-5-4-6-17-7-8-21(26-22(16)17)27-11-9-20(24(2,3)15-27)23(29)25-18-10-12-30-19(13-18)14-28/h4-8,18-20,28H,9-15H2,1-3H3,(H,25,29)/t18-,19-,20+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of dog mPGES-1 |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50028854

(CHEMBL3342693)Show SMILES CC(C)C(=O)NCc1cnc(C(F)F)c(c1)C(=O)Nc1nc(C)c([nH]1)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C23H22F5N5O2/c1-11(2)20(34)30-10-13-8-16(18(19(24)25)29-9-13)21(35)33-22-31-12(3)17(32-22)14-4-6-15(7-5-14)23(26,27)28/h4-9,11,19H,10H2,1-3H3,(H,30,34)(H2,31,32,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in LPS-induced human whole blood assessed as suppression of PGE2 response after 20 to 24 hrs by LC-MS/MS analysis |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50028854

(CHEMBL3342693)Show SMILES CC(C)C(=O)NCc1cnc(C(F)F)c(c1)C(=O)Nc1nc(C)c([nH]1)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C23H22F5N5O2/c1-11(2)20(34)30-10-13-8-16(18(19(24)25)29-9-13)21(35)33-22-31-12(3)17(32-22)14-4-6-15(7-5-14)23(26,27)28/h4-9,11,19H,10H2,1-3H3,(H,30,34)(H2,31,32,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in rhIL-1beta-stimulated human A549 cells assessed as PGE2 level treated for 18 hrs after 30 mins pre-incubation with rhIL-1beta... |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50194142
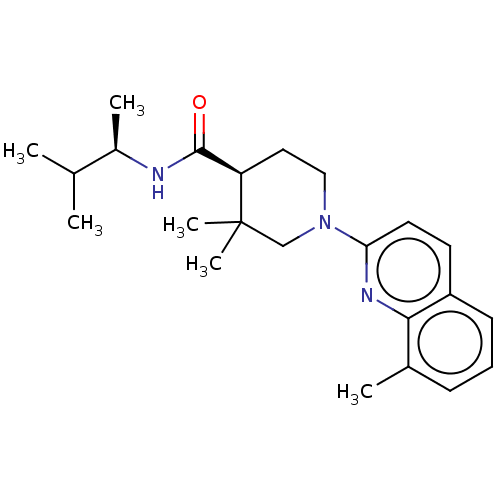
(CHEMBL3947843)Show SMILES CC(C)[C@@H](C)NC(=O)[C@H]1CCN(CC1(C)C)c1ccc2cccc(C)c2n1 |r| Show InChI InChI=1S/C23H33N3O/c1-15(2)17(4)24-22(27)19-12-13-26(14-23(19,5)6)20-11-10-18-9-7-8-16(3)21(18)25-20/h7-11,15,17,19H,12-14H2,1-6H3,(H,24,27)/t17-,19-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis |
Bioorg Med Chem Lett 26: 4824-4828 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.023
BindingDB Entry DOI: 10.7270/Q2125VMX |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50142252

(CHEMBL3758953)Show SMILES CC(C)C(=O)NCc1ccc(C(F)F)c(c1)C(=O)Nc1ncc([nH]1)-c1ccc(Cl)cc1 Show InChI InChI=1S/C22H21ClF2N4O2/c1-12(2)20(30)26-10-13-3-8-16(19(24)25)17(9-13)21(31)29-22-27-11-18(28-22)14-4-6-15(23)7-5-14/h3-9,11-12,19H,10H2,1-2H3,(H,26,30)(H2,27,28,29,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in rhIL-1beta-stimulated human A549 cells assessed as PGE2 level treated for 18 hrs after 30 mins pre-incubation with rhIL-1beta... |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50142251
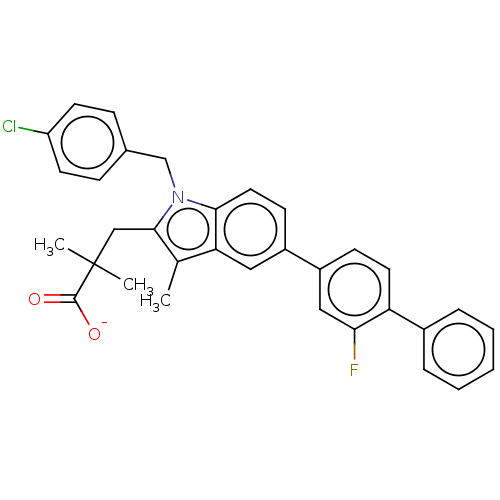
(CHEMBL3758663)Show SMILES [Na+].Cc1c(CC(C)(C)C([O-])=O)n(Cc2ccc(Cl)cc2)c2ccc(cc12)-c1ccc(c(F)c1)-c1ccccc1 Show InChI InChI=1S/C33H29ClFNO2.Na/c1-21-28-17-24(25-11-15-27(29(35)18-25)23-7-5-4-6-8-23)12-16-30(28)36(20-22-9-13-26(34)14-10-22)31(21)19-33(2,3)32(37)38;/h4-18H,19-20H2,1-3H3,(H,37,38);/q;+1/p-1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human microsomal PGES1 expressed in 293E cells by LC/MS/MS analysis |
J Med Chem 59: 194-205 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01249
BindingDB Entry DOI: 10.7270/Q2474CQR |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data