 Found 43 hits with Last Name = 'chun' and Initial = 'ho'
Found 43 hits with Last Name = 'chun' and Initial = 'ho' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM14361
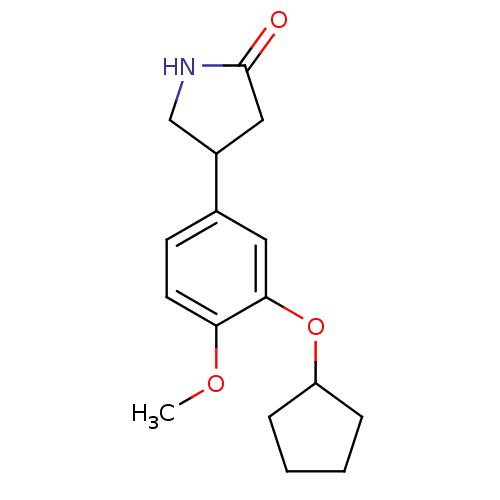
((R,S)-Rolipram | 4-(3-cyclopentyloxy-4-methoxy-phe...)Show InChI InChI=1S/C16H21NO3/c1-19-14-7-6-11(12-9-16(18)17-10-12)8-15(14)20-13-4-2-3-5-13/h6-8,12-13H,2-5,9-10H2,1H3,(H,17,18) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.00230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220995

(CHEMBL77788)Show InChI InChI=1S/C15H20N2O3/c1-11(18)17-16-10-12-7-8-14(19-2)15(9-12)20-13-5-3-4-6-13/h7-10,13H,3-6H2,1-2H3,(H,17,18)/b16-10+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221005
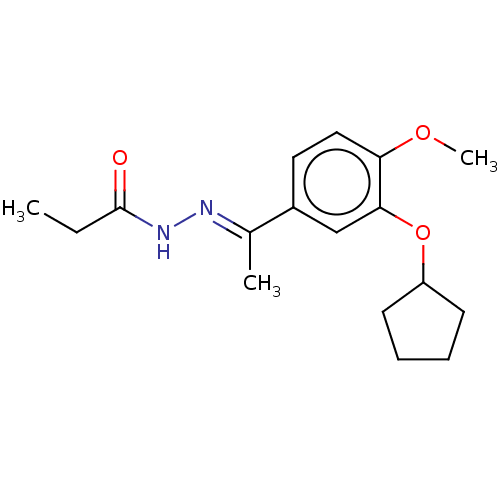
(CHEMBL75684)Show InChI InChI=1S/C17H24N2O3/c1-4-17(20)19-18-12(2)13-9-10-15(21-3)16(11-13)22-14-7-5-6-8-14/h9-11,14H,4-8H2,1-3H3,(H,19,20)/b18-12+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220998
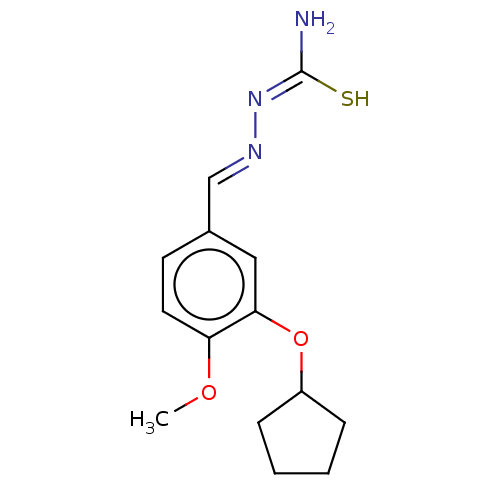
(CHEMBL76382)Show InChI InChI=1S/C14H19N3O2S/c1-18-12-7-6-10(9-16-17-14(15)20)8-13(12)19-11-4-2-3-5-11/h6-9,11H,2-5H2,1H3,(H3,15,17,20)/b16-9+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221003
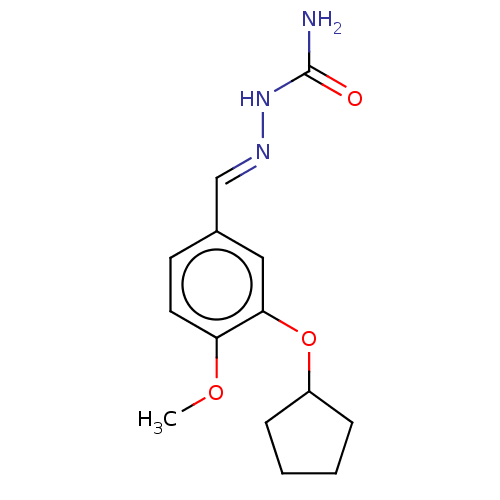
(CHEMBL432348)Show InChI InChI=1S/C14H19N3O3/c1-19-12-7-6-10(9-16-17-14(15)18)8-13(12)20-11-4-2-3-5-11/h6-9,11H,2-5H2,1H3,(H3,15,17,18)/b16-9+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0690 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220997

(CHEMBL78237)Show InChI InChI=1S/C16H22N2O3/c1-11(17-18-12(2)19)13-8-9-15(20-3)16(10-13)21-14-6-4-5-7-14/h8-10,14H,4-7H2,1-3H3,(H,18,19)/b17-11+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0920 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221006
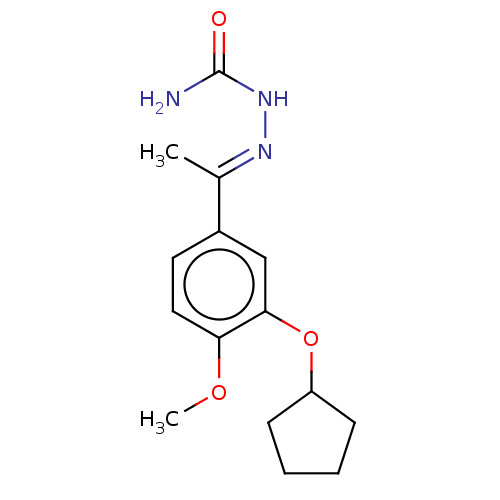
(CHEMBL77358)Show InChI InChI=1S/C15H21N3O3/c1-10(17-18-15(16)19)11-7-8-13(20-2)14(9-11)21-12-5-3-4-6-12/h7-9,12H,3-6H2,1-2H3,(H3,16,18,19)/b17-10+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.146 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220999

(CHEMBL77745)Show InChI InChI=1S/C15H21N3O2S/c1-10(17-18-15(16)21)11-7-8-13(19-2)14(9-11)20-12-5-3-4-6-12/h7-9,12H,3-6H2,1-2H3,(H3,16,18,21)/b17-10+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.336 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220996

(CHEMBL76635)Show InChI InChI=1S/C20H23N3O3/c1-25-17-12-11-15(13-18(17)26-16-9-5-6-10-16)19(22-23-20(21)24)14-7-3-2-4-8-14/h2-4,7-8,11-13,16H,5-6,9-10H2,1H3,(H3,21,23,24)/b22-19+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221004
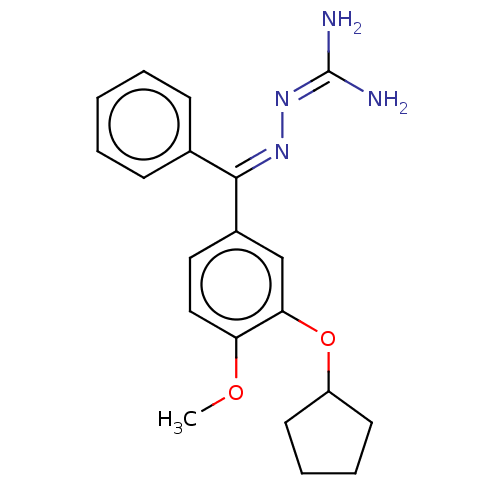
(CHEMBL77999)Show SMILES [#6]-[#8]-c1ccc(cc1-[#8]-[#6]-1-[#6]-[#6]-[#6]-[#6]-1)-[#6](=[#7]\[#7]=[#6](/[#7])-[#7])\c1ccccc1 Show InChI InChI=1S/C20H24N4O2/c1-25-17-12-11-15(13-18(17)26-16-9-5-6-10-16)19(23-24-20(21)22)14-7-3-2-4-8-14/h2-4,7-8,11-13,16H,5-6,9-10H2,1H3,(H4,21,22,24)/b23-19+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221001
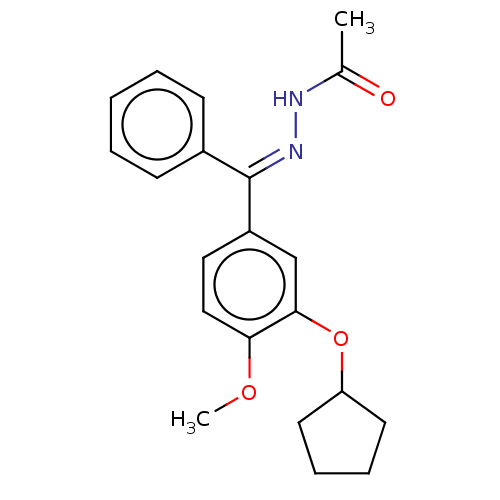
(CHEMBL76257)Show InChI InChI=1S/C21H24N2O3/c1-15(24)22-23-21(16-8-4-3-5-9-16)17-12-13-19(25-2)20(14-17)26-18-10-6-7-11-18/h3-5,8-9,12-14,18H,6-7,10-11H2,1-2H3,(H,22,24)/b23-21+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220993

(CHEMBL78238)Show SMILES COc1ccc(cc1OC1CCCC1)C(=N\N=C(\N)S)\c1ccccc1 Show InChI InChI=1S/C20H23N3O2S/c1-24-17-12-11-15(13-18(17)25-16-9-5-6-10-16)19(22-23-20(21)26)14-7-3-2-4-8-14/h2-4,7-8,11-13,16H,5-6,9-10H2,1H3,(H3,21,23,26)/b22-19+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221007
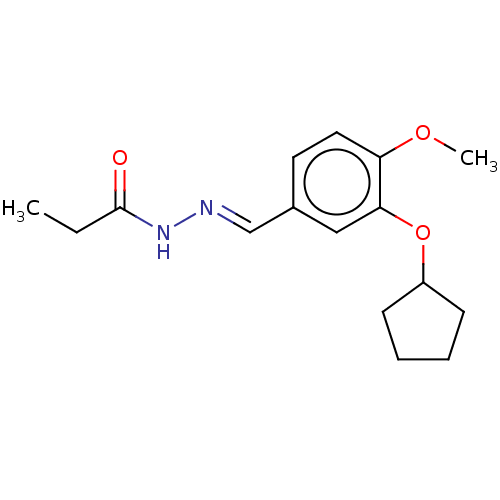
(CHEMBL80258)Show InChI InChI=1S/C16H22N2O3/c1-3-16(19)18-17-11-12-8-9-14(20-2)15(10-12)21-13-6-4-5-7-13/h8-11,13H,3-7H2,1-2H3,(H,18,19)/b17-11+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221002

(CHEMBL306320)Show SMILES [#6]-[#8]-c1ccc(cc1-[#8]-[#6]-1-[#6]-[#6]-[#6]-[#6]-1)-[#6](\[#6])=[#7]\[#7]=[#6](/[#7])-[#7] Show InChI InChI=1S/C15H22N4O2/c1-10(18-19-15(16)17)11-7-8-13(20-2)14(9-11)21-12-5-3-4-6-12/h7-9,12H,3-6H2,1-2H3,(H4,16,17,19)/b18-10+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220994
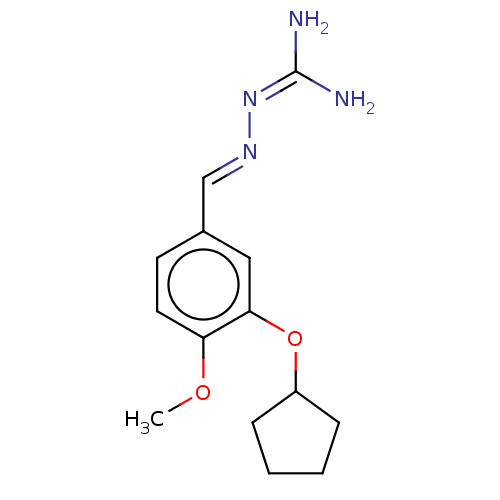
(CHEMBL77177)Show SMILES [#6]-[#8]-c1ccc(\[#6]=[#7]\[#7]=[#6](/[#7])-[#7])cc1-[#8]-[#6]-1-[#6]-[#6]-[#6]-[#6]-1 Show InChI InChI=1S/C14H20N4O2/c1-19-12-7-6-10(9-17-18-14(15)16)8-13(12)20-11-4-2-3-5-11/h6-9,11H,2-5H2,1H3,(H4,15,16,18)/b17-9+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221000
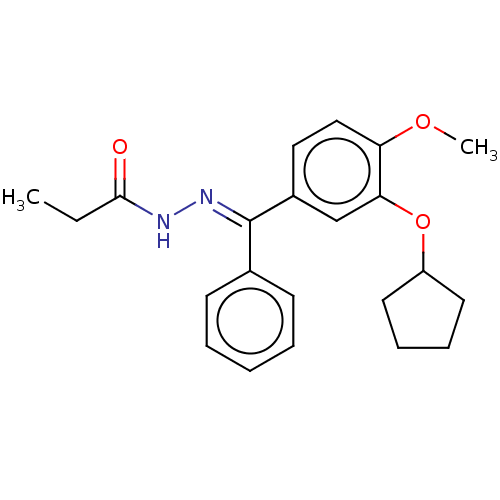
(CHEMBL431962)Show SMILES CCC(=O)N\N=C(/c1ccccc1)c1ccc(OC)c(OC2CCCC2)c1 Show InChI InChI=1S/C22H26N2O3/c1-3-21(25)23-24-22(16-9-5-4-6-10-16)17-13-14-19(26-2)20(15-17)27-18-11-7-8-12-18/h4-6,9-10,13-15,18H,3,7-8,11-12H2,1-2H3,(H,23,25)/b24-22+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50116711
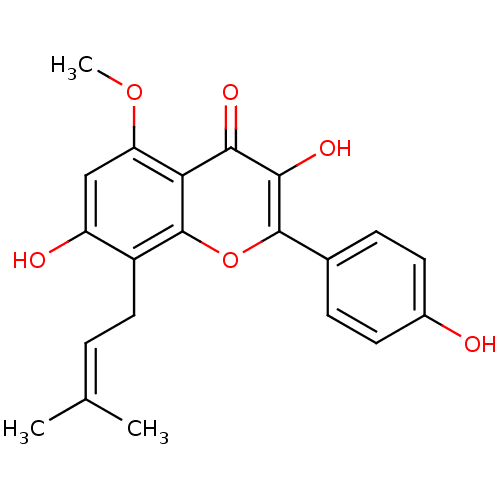
(3,7-Dihydroxy-2-(4-hydroxy-phenyl)-5-methoxy-8-(3-...)Show SMILES [#6]-[#8]-c1cc(-[#8])c(-[#6]\[#6]=[#6](\[#6])-[#6])c2oc(c(-[#8])c(=O)c12)-c1ccc(-[#8])cc1 Show InChI InChI=1S/C21H20O6/c1-11(2)4-9-14-15(23)10-16(26-3)17-18(24)19(25)20(27-21(14)17)12-5-7-13(22)8-6-12/h4-8,10,22-23,25H,9H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 5 from rat diaphragm |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM14390

(5-[2-ethoxy-5-(4-methyl-1-piperazinylsulfonyl)phen...)Show SMILES CCCc1nn(C)c2c1nc([nH]c2=O)-c1cc(ccc1OCC)S(=O)(=O)N1CCN(C)CC1 Show InChI InChI=1S/C22H30N6O4S/c1-5-7-17-19-20(27(4)25-17)22(29)24-21(23-19)16-14-15(8-9-18(16)32-6-2)33(30,31)28-12-10-26(3)11-13-28/h8-9,14H,5-7,10-13H2,1-4H3,(H,23,24,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 5 from rat diaphragm |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM14361

((R,S)-Rolipram | 4-(3-cyclopentyloxy-4-methoxy-phe...)Show InChI InChI=1S/C16H21NO3/c1-19-14-7-6-11(12-9-16(18)17-10-12)8-15(14)20-13-4-2-3-5-13/h6-8,12-13H,2-5,9-10H2,1H3,(H,17,18) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220996

(CHEMBL76635)Show InChI InChI=1S/C20H23N3O3/c1-25-17-12-11-15(13-18(17)26-16-9-5-6-10-16)19(22-23-20(21)24)14-7-3-2-4-8-14/h2-4,7-8,11-13,16H,5-6,9-10H2,1H3,(H3,21,23,24)/b22-19+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.580 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221003

(CHEMBL432348)Show InChI InChI=1S/C14H19N3O3/c1-19-12-7-6-10(9-16-17-14(15)18)8-13(12)20-11-4-2-3-5-11/h6-9,11H,2-5H2,1H3,(H3,15,17,18)/b16-9+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220998

(CHEMBL76382)Show InChI InChI=1S/C14H19N3O2S/c1-18-12-7-6-10(9-16-17-14(15)20)8-13(12)19-11-4-2-3-5-11/h6-9,11H,2-5H2,1H3,(H3,15,17,20)/b16-9+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221004

(CHEMBL77999)Show SMILES [#6]-[#8]-c1ccc(cc1-[#8]-[#6]-1-[#6]-[#6]-[#6]-[#6]-1)-[#6](=[#7]\[#7]=[#6](/[#7])-[#7])\c1ccccc1 Show InChI InChI=1S/C20H24N4O2/c1-25-17-12-11-15(13-18(17)26-16-9-5-6-10-16)19(23-24-20(21)22)14-7-3-2-4-8-14/h2-4,7-8,11-13,16H,5-6,9-10H2,1H3,(H4,21,22,24)/b23-19+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220993

(CHEMBL78238)Show SMILES COc1ccc(cc1OC1CCCC1)C(=N\N=C(\N)S)\c1ccccc1 Show InChI InChI=1S/C20H23N3O2S/c1-24-17-12-11-15(13-18(17)25-16-9-5-6-10-16)19(22-23-20(21)26)14-7-3-2-4-8-14/h2-4,7-8,11-13,16H,5-6,9-10H2,1H3,(H3,21,23,26)/b22-19+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220995

(CHEMBL77788)Show InChI InChI=1S/C15H20N2O3/c1-11(18)17-16-10-12-7-8-14(19-2)15(9-12)20-13-5-3-4-6-13/h7-10,13H,3-6H2,1-2H3,(H,17,18)/b16-10+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221001

(CHEMBL76257)Show InChI InChI=1S/C21H24N2O3/c1-15(24)22-23-21(16-8-4-3-5-9-16)17-12-13-19(25-2)20(14-17)26-18-10-6-7-11-18/h3-5,8-9,12-14,18H,6-7,10-11H2,1-2H3,(H,22,24)/b23-21+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220999

(CHEMBL77745)Show InChI InChI=1S/C15H21N3O2S/c1-10(17-18-15(16)21)11-7-8-13(19-2)14(9-11)20-12-5-3-4-6-12/h7-9,12H,3-6H2,1-2H3,(H3,16,18,21)/b17-10+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221000

(CHEMBL431962)Show SMILES CCC(=O)N\N=C(/c1ccccc1)c1ccc(OC)c(OC2CCCC2)c1 Show InChI InChI=1S/C22H26N2O3/c1-3-21(25)23-24-22(16-9-5-4-6-10-16)17-13-14-19(26-2)20(15-17)27-18-11-7-8-12-18/h4-6,9-10,13-15,18H,3,7-8,11-12H2,1-2H3,(H,23,25)/b24-22+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220994

(CHEMBL77177)Show SMILES [#6]-[#8]-c1ccc(\[#6]=[#7]\[#7]=[#6](/[#7])-[#7])cc1-[#8]-[#6]-1-[#6]-[#6]-[#6]-[#6]-1 Show InChI InChI=1S/C14H20N4O2/c1-19-12-7-6-10(9-17-18-14(15)16)8-13(12)20-11-4-2-3-5-11/h6-9,11H,2-5H2,1H3,(H4,15,16,18)/b17-9+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >10 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221002

(CHEMBL306320)Show SMILES [#6]-[#8]-c1ccc(cc1-[#8]-[#6]-1-[#6]-[#6]-[#6]-[#6]-1)-[#6](\[#6])=[#7]\[#7]=[#6](/[#7])-[#7] Show InChI InChI=1S/C15H22N4O2/c1-10(18-19-15(16)17)11-7-8-13(20-2)14(9-11)21-12-5-3-4-6-12/h7-9,12H,3-6H2,1-2H3,(H4,16,17,19)/b18-10+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >10 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221006

(CHEMBL77358)Show InChI InChI=1S/C15H21N3O3/c1-10(17-18-15(16)19)11-7-8-13(20-2)14(9-11)21-12-5-3-4-6-12/h7-9,12H,3-6H2,1-2H3,(H3,16,18,19)/b17-10+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >10 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221005

(CHEMBL75684)Show InChI InChI=1S/C17H24N2O3/c1-4-17(20)19-18-12(2)13-9-10-15(21-3)16(11-13)22-14-7-5-6-8-14/h9-11,14H,4-8H2,1-3H3,(H,19,20)/b18-12+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >10 | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Inhibitory activity against purified rat liver phosphodiesterase 4 |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50116711

(3,7-Dihydroxy-2-(4-hydroxy-phenyl)-5-methoxy-8-(3-...)Show SMILES [#6]-[#8]-c1cc(-[#8])c(-[#6]\[#6]=[#6](\[#6])-[#6])c2oc(c(-[#8])c(=O)c12)-c1ccc(-[#8])cc1 Show InChI InChI=1S/C21H20O6/c1-11(2)4-9-14-15(23)10-16(26-3)17-18(24)19(25)20(27-21(14)17)12-5-7-13(22)8-6-12/h4-8,10,22-23,25H,9H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 5 from rat diaphragm |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50366787
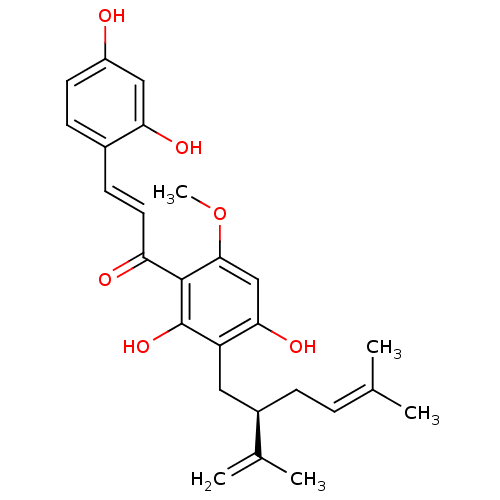
(KURAIDIN)Show SMILES [#6]-[#8]-c1cc(-[#8])c(-[#6]-[#6@@H](-[#6]\[#6]=[#6](\[#6])-[#6])-[#6](-[#6])=[#6])c(-[#8])c1-[#6](=O)\[#6]=[#6]\c1ccc(-[#8])cc1-[#8] Show InChI InChI=1S/C26H30O6/c1-15(2)6-7-18(16(3)4)12-20-23(30)14-24(32-5)25(26(20)31)21(28)11-9-17-8-10-19(27)13-22(17)29/h6,8-11,13-14,18,27,29-31H,3,7,12H2,1-2,4-5H3/b11-9+/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 640 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 5 from rat diaphragm |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A/4B/4C/4D
(RAT-Rattus norvegicus) | BDBM14361

((R,S)-Rolipram | 4-(3-cyclopentyloxy-4-methoxy-phe...)Show InChI InChI=1S/C16H21NO3/c1-19-14-7-6-11(12-9-16(18)17-10-12)8-15(14)20-13-4-2-3-5-13/h6-8,12-13H,2-5,9-10H2,1H3,(H,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| n/a | n/a | 1.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 4 from rat liver |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A/4B/4C/4D
(RAT-Rattus norvegicus) | BDBM50116711

(3,7-Dihydroxy-2-(4-hydroxy-phenyl)-5-methoxy-8-(3-...)Show SMILES [#6]-[#8]-c1cc(-[#8])c(-[#6]\[#6]=[#6](\[#6])-[#6])c2oc(c(-[#8])c(=O)c12)-c1ccc(-[#8])cc1 Show InChI InChI=1S/C21H20O6/c1-11(2)4-9-14-15(23)10-16(26-3)17-18(24)19(25)20(27-21(14)17)12-5-7-13(22)8-6-12/h4-8,10,22-23,25H,9H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.55E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 4 from rat liver |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50116713

((2R,3R)-2-(2,4-Dihydroxy-phenyl)-3,7-dihydroxy-8-(...)Show SMILES COc1cc(O)c(CC(CCC(C)(C)O)C(C)=C)c2O[C@@H]([C@@H](O)C(=O)c12)c1ccc(O)cc1O Show InChI InChI=1S/C26H32O8/c1-13(2)14(8-9-26(3,4)32)10-17-19(29)12-20(33-5)21-22(30)23(31)25(34-24(17)21)16-7-6-15(27)11-18(16)28/h6-7,11-12,14,23,25,27-29,31-32H,1,8-10H2,2-5H3/t14?,23-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.75E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 5 from rat diaphragm |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50366788
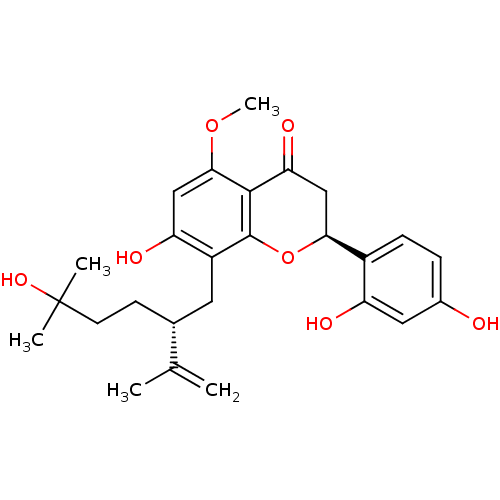
(KURARINOL)Show SMILES COc1cc(O)c(C[C@@H](CCC(C)(C)O)C(C)=C)c2O[C@@H](CC(=O)c12)c1ccc(O)cc1O |r| Show InChI InChI=1S/C26H32O7/c1-14(2)15(8-9-26(3,4)31)10-18-20(29)12-23(32-5)24-21(30)13-22(33-25(18)24)17-7-6-16(27)11-19(17)28/h6-7,11-12,15,22,27-29,31H,1,8-10,13H2,2-5H3/t15-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 5 from rat diaphragm |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A/4B/4C/4D
(RAT-Rattus norvegicus) | BDBM50366788

(KURARINOL)Show SMILES COc1cc(O)c(C[C@@H](CCC(C)(C)O)C(C)=C)c2O[C@@H](CC(=O)c12)c1ccc(O)cc1O |r| Show InChI InChI=1S/C26H32O7/c1-14(2)15(8-9-26(3,4)31)10-18-20(29)12-23(32-5)24-21(30)13-22(33-25(18)24)17-7-6-16(27)11-19(17)28/h6-7,11-12,15,22,27-29,31H,1,8-10,13H2,2-5H3/t15-,22+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6.59E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 4 from rat liver |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A/4B/4C/4D
(RAT-Rattus norvegicus) | BDBM50366787

(KURAIDIN)Show SMILES [#6]-[#8]-c1cc(-[#8])c(-[#6]-[#6@@H](-[#6]\[#6]=[#6](\[#6])-[#6])-[#6](-[#6])=[#6])c(-[#8])c1-[#6](=O)\[#6]=[#6]\c1ccc(-[#8])cc1-[#8] Show InChI InChI=1S/C26H30O6/c1-15(2)6-7-18(16(3)4)12-20-23(30)14-24(32-5)25(26(20)31)21(28)11-9-17-8-10-19(27)13-22(17)29/h6,8-11,13-14,18,27,29-31H,3,7,12H2,1-2,4-5H3/b11-9+/t18-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 5 from rat diaphragm |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50116710
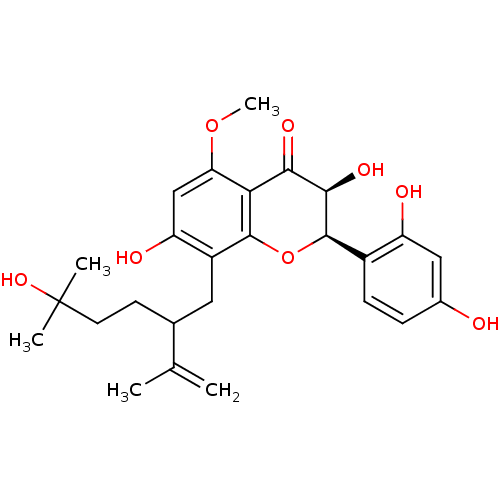
((2R,3S)-2-(2,4-Dihydroxy-phenyl)-3,7-dihydroxy-8-(...)Show SMILES COc1cc(O)c(CC(CCC(C)(C)O)C(C)=C)c2O[C@@H]([C@H](O)C(=O)c12)c1ccc(O)cc1O Show InChI InChI=1S/C26H32O8/c1-13(2)14(8-9-26(3,4)32)10-17-19(29)12-20(33-5)21-22(30)23(31)25(34-24(17)21)16-7-6-15(27)11-18(16)28/h6-7,11-12,14,23,25,27-29,31-32H,1,8-10H2,2-5H3/t14?,23-,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.06E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 5 from rat diaphragm |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A/4B/4C/4D
(RAT-Rattus norvegicus) | BDBM50116713

((2R,3R)-2-(2,4-Dihydroxy-phenyl)-3,7-dihydroxy-8-(...)Show SMILES COc1cc(O)c(CC(CCC(C)(C)O)C(C)=C)c2O[C@@H]([C@@H](O)C(=O)c12)c1ccc(O)cc1O Show InChI InChI=1S/C26H32O8/c1-13(2)14(8-9-26(3,4)32)10-17-19(29)12-20(33-5)21-22(30)23(31)25(34-24(17)21)16-7-6-15(27)11-18(16)28/h6-7,11-12,14,23,25,27-29,31-32H,1,8-10H2,2-5H3/t14?,23-,25+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.87E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 4 from rat liver |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4A/4B/4C/4D
(RAT-Rattus norvegicus) | BDBM50116710

((2R,3S)-2-(2,4-Dihydroxy-phenyl)-3,7-dihydroxy-8-(...)Show SMILES COc1cc(O)c(CC(CCC(C)(C)O)C(C)=C)c2O[C@@H]([C@H](O)C(=O)c12)c1ccc(O)cc1O Show InChI InChI=1S/C26H32O8/c1-13(2)14(8-9-26(3,4)32)10-17-19(29)12-20(33-5)21-22(30)23(31)25(34-24(17)21)16-7-6-15(27)11-18(16)28/h6-7,11-12,14,23,25,27-29,31-32H,1,8-10H2,2-5H3/t14?,23-,25-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.38E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science& Technology
Curated by ChEMBL
| Assay Description
Inhibition of Phosphodiesterase 4 from rat liver |
Bioorg Med Chem Lett 12: 2313-6 (2002)
BindingDB Entry DOI: 10.7270/Q21R6R2S |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data